Abstract
PDE4 is a potential target for treating mental diseases including depression, Alzheimer’s disease and schizophrenia involving in the hydrolysis of cAMP. Curcumin is well-known for its potential as a neuroprotective compound against depression. It was able to inhibit PDE4 with IC50 value in the range of 10 to 20 μM with low selectivity. However, curcumin itself cannot be used clinically because of its poor drug-like properties. Herein, a series of new curcumin derivatives were designed and synthesized. Their PDE4 inhibitory activities and antidepressant effects in vivo were also evaluated. Consequently, most of these curcumin derivatives demonstrated good inhibitory activities against PDE4. Furthermore, compound

A series of novel curcumin derivatives were synthesized and subjected to pharmacological evaluation as PDE4 inhibitors. Consequently, compound
Introduction
Depression is a severe disorder by high rates of suicide and its clinical manifestations include mainly anhedonia, lack of sleep and motivation, or mental torture.1,2 Statistics shows that the mortality rate of depression ranks among the top in various diseases, and it also is the second largest contributor to medical burden all over the world.3 –5 The unknown etiopathology of depression partly cause to its complexity in pharmacotherapy. Presently, clinical antidepressants are developed based on the theoretical hypothesis of serotonin and noradrenaline function in the brain. However, only a small portion of patients could be relieved in response to these drugs. 6 Novel antidepressants with different mechanisms may provide a novel perspective for new drug discovery. Therefore, the development of anti-depression agents with novel mechanism is still urgent clinically.
As a second messenger for mediating cellular responses to neurotransmitters in the brain, cAMP has been shown to be a vital target for the treatment of mental diseases.7 –9 Phosphodiesterase 4 (PDE4), an enzyme catalyzing specifically the hydrolysis of cAMP, can regulate the cAMP/PKA/CREB pathway. Besides, PDE4 also plays a key role in modulating cAMP-mediated dopamine signaling in the postsynaptic region. 10 In fact, some PDE4 inhibitors including rolipram and roflumilast have been explored for treating neuropsychiatric disorders.11 –16 An early developed PDE4 inhibitor, rolipram, could increase cAMP signaling and exhibit antidepressant- and anxiolytic-like effects in vivo. 17
It has been known that natural compounds are promising resources for depression therapy. 18 Curcumin (Figure 1) is the active chemical constituent in turmeric (Curcuma longa), has been used as a common traditional Chinese medicine and food additive. It also has been well known for its various biological activities such as anti-inflammatory and antioxidant properties. 19 Previous studies demonstrated that curcumin exhibits significant effects in depression-like behaviors induced by stress, but for non-induced depression, its effect is unknown.20 –22 Interestingly, curcumin was proved to be a nonselective PDE inhibitor, inhibiting PDE1-4 with IC50 values between 10 and 20 μM, and PDE5 with an IC50 value of 35 μM. 23 For example, curcumin exhibited anti-angiogenic antitumour activities in vitro and in vivo through PDE2/4 inhibition. Abusnina et al. have reported that curcumin could inhibit melanoma cell proliferation by targeting PDE1. However, currently, there are no reports about curcumin derivatives as PDE4 inhibitors and their antidepressant effects.24,25 Furthermore, the clinical application of curcumin is limited because of its poor drug-like properties such as low water-solubility and bioavailability. In order to overcome this, many of its derivatives have been reported until now, of which pyrazole derivatives of curcumin demonstrated many interesting bioactivity such as enhanced memory, antioxidant, anti-mycobacterial activities.26 –28 Therefore, we wonder if pyrazole derivatives of curcumin can be attractive leading compounds as PDE4 inhibitors for treating depression. Herein, we constructed a series of curcumin analogue by introducing a pyrazole group in its structure and evaluated their antidepressant activities in animal models.

The structures of curcumin.
Results and discussion
The preparation of curcumin derivatives

Synthesis of
All of the curcumin derivatives were evaluated for the activities of inhibiting PDE4, and the results are shown in Table 1. Curcumin and rolipram (a commercially available PDE4 inhibitor) were applied as standard compounds for comparison. PDE4 screening and other PDE evaluations were carried out using an activity assay based on the hydrolysis of cyclic AMP and cyclic GMP. We initially focused on introducing various substituted pyrazol rings in the middle of curcumin molecule while other structural parts keep unchanged. The results showed that most of this series of compounds demonstrated better inhibitory activities than curcumin except for
PDE4 inhibitory activities of curcumin derivatives.

Results are expressed as the mean of at least three experiments.
PDE selectivity of compound

Results are expressed as the mean of at least three experiments.
Compound

Binding modes of compound
Encouraged by its potent PDE4 inhibition activity, compound
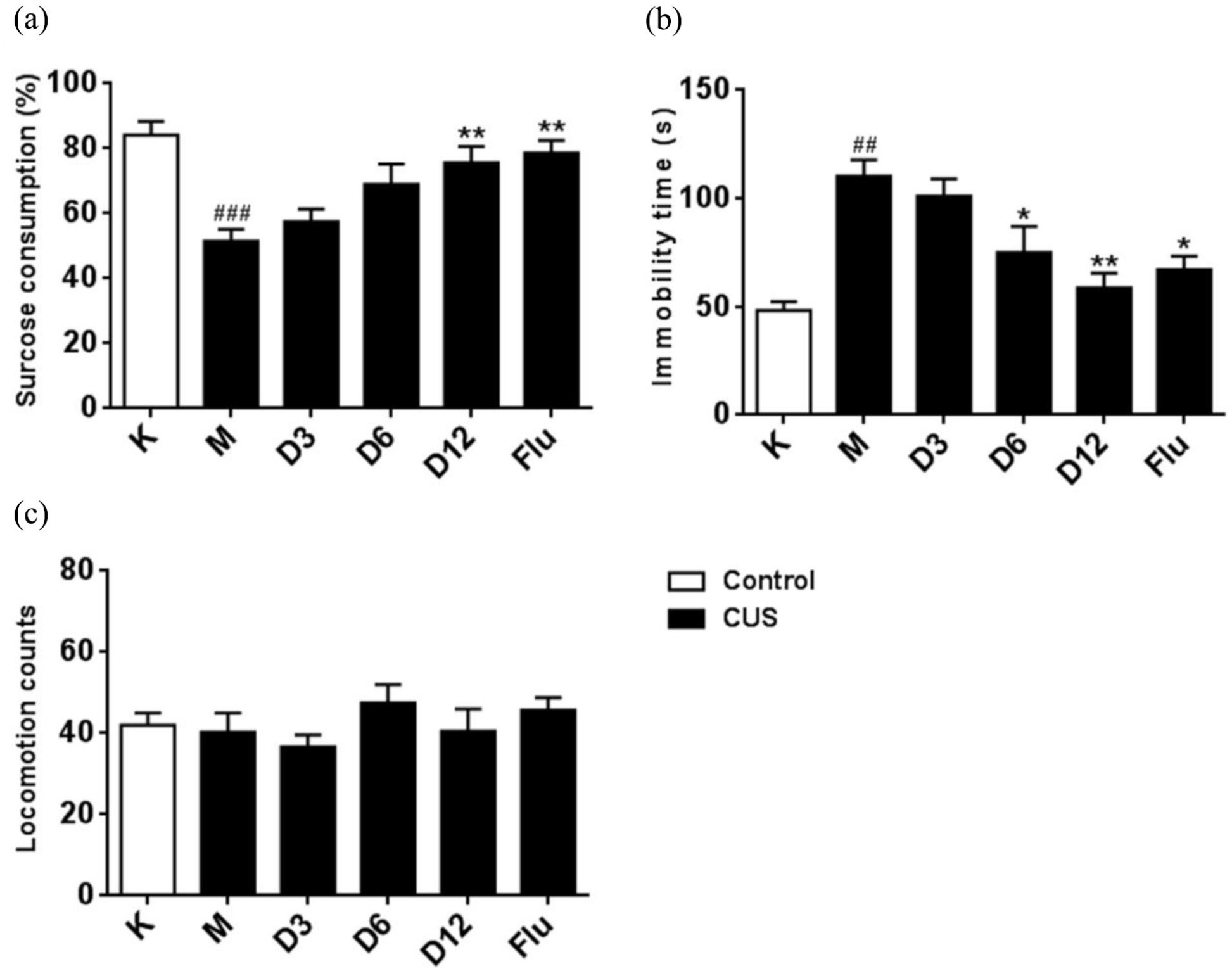
The antidepressant-like effects of compound
In summary, a series of curcumin-based PDE4 inhibitors have been designed and synthesized. The in vitro study showed that most of these compounds demonstrated better inhibitory activities than curcumin itself. Of all the curcumin derivatives, compound
Experimental
Materials and reagents
All chemicals and reagents used (Shanghai Aladdin Bio-Chem Technology Co., Ltd) were of analytical grade. Thin-layer and column chromatography was performed using silica gel (230-400 mesh). 1H and 13C NMR spectra were conducted on Bruker spectrometers and chemical shifts were reported in ppm (δ). MS measurements were performed on a Thermo Velos Pro Orbitrap Elite Hybrid Mass spectrometer (Thermo Fisher Scientific, Bremen, Germany). Measurement of melting points was carried out by a micro apparatus (Boetius).
Synthesis of 3,5-diarylpyrazoles 3a–j
1,7-bis(4-hydroxy-3-methoxyphenyl)heptane-3,5-dione (
1 H NMR (400 MHz, DMSO-d6): δ = 9.97 (s, 1H), 9.46 (s, 1H), 6.85 (m,2H), 6.67 (m, 4H), 5.53 (br s, 2H), 5.42 (s, 1H), 3.71 (s, 6H), 2.85-2.73 (m, 4H), 2.55 (m, 4H). 13C NMR (100 MHz, DMSO-d6): δ = 202.47, 194.22, 146.31, 143.56, 132.46, 120.71, 113.97, 110.43, 109.72, 99.81, 57.32, 55.46, 45.37, 41.25, 31.53, 29.12. HRMS (ESI): m/z [M + H]+ calcd for C21H25O6: 373.1651; found: 373.1669.
Synthesis of compounds
4,4'-((1-phenyl-1H-pyrazole-3,5-diyl)bis(ethane-2,1-diyl))bis(2-methoxyphenol) (
1-(3,5-bis(4-hydroxy-3-methoxyphenethyl)-1H-pyrazol-1-yl)ethan-1-one (
4,4'-((1-(2-hydroxyethyl)-1H-pyrazole-3,5-diyl)bis(ethane-2,1-diyl))bis(2-methoxyphenol) (
Ethyl 3,5-bis(4-hydroxy-3-methoxyphenethyl)-1H-pyrazole-1-carboxylate (
4,4'-((1-(4-methoxyphenyl)-1H-pyrazole-3,5-diyl)bis(ethane-2,1-diyl))bis(2-methoxyphenol) (
4,4'-((1-(3-methoxyphenyl)-1H-pyrazole-3,5-diyl)bis(ethane-2,1-diyl))bis(2-methoxyphenol) (
4,4'-((1-(4-chlorophenyl)-1H-pyrazole-3,5-diyl)bis(ethane-2,1-diyl))bis(2-methoxyphenol) (
4,4'-((1-(3-chlorophenyl)-1H-pyrazole-3,5-diyl)bis(ethane-2,1-diyl))bis(2-methoxyphenol) (
4,4'-((1-(4-(trifluoromethyl)phenyl)-1H-pyrazole-3,5-diyl)bis(ethane-2,1-diyl))bis(2-methoxyphenol) (
Synthesis of compounds
4,4'-((1E,1'E)-(1-phenyl-1H-pyrazole-3,5-diyl)bis(ethene-2,1-diyl))bis(2-methoxyphenol) (
1-(3,5-Bis((E)-4-hydroxy-3-methoxystyryl)-1H-pyrazol-1-yl)ethan-1-one (
4,4'-((1E,1'E)-(1-(2-hydroxyethyl)-1H-pyrazole-3,5-diyl)bis(ethene-2,1-diyl))bis(2-methoxyphenol) (
Ethyl 3,5-bis((E)-4-hydroxy-3-methoxystyryl)-1H-pyrazole-1-carboxylate (
4,4'-((1E,1'E)-(1-(4-methoxyphenyl)-1H-pyrazole-3,5-diyl)bis(ethene-2,1-diyl))bis(2-methoxyphenol) (
4,4'-((1E,1'E)-(1-(3-methoxyphenyl)-1H-pyrazole-3,5-diyl)bis(ethene-2,1-diyl))bis(2-methoxyphenol) (
4,4'-((1E,1'E)-(1-(4-chlorophenyl)-1H-pyrazole-3,5-diyl)bis(ethene-2,1-diyl))bis(2-methoxyphenol) (
4,4'-((1E,1'E)-(1-(3-chlorophenyl)-1H-pyrazole-3,5-diyl)bis(ethene-2,1-diyl))bis(2-methoxyphenol) (
4,4'-((1E,1'E)-(1-(4-(trifluoromethyl)phenyl)-1H-pyrazole-3,5-diyl)bis(ethene-2,1-diyl))bis(2-methoxyphenol) (
Enzymatic assay
The substrate 3H-cGMP and 3H-cAMP were used to assay PDE enzymatic activities by the phosphodiesterase scintillation proximity assay (SPA). First, PDEs were incubated with a solution of Tris–HCl (50 mmol/L, pH 7.8), MgCl2 (10 mmol/L), and 3H-cAMP or 3H-cGMP (40 000 cpm/assay) at 24 °C for 30 minutes. After complete hydrolysis, the reaction was terminated by adding ZnSO4 and then adding the solution of 0.2 N Ba(OH)2 to precipitate the product 3H-GMP. Then, the radioactivity of unreacted 3H-cGMP in the supernatant was measured, and the IC50 values were measured.
Animals and treatment
Male ICR mice, 22-25 g, were purchased from Nanjing University of Traditional Chinese Medicine. The animal tests were approved by the Institutional Animal Care and Use Committee of Changzhou University. Saline containing 5% DMSO was used to dissolve all of the test compounds, but for fluoxetine, the saline did not contain DMSO. Mice were administrated intragastrically (compound
Behavioral tests
SPT and locomotor activity (LA) tests were conducted according to the previous description.30,31 For FST, mice were induced individually into a glass cylinder (20 cm diameter, 40 cm depth). And the cylinder was filled with water of 25 cm at 25 °C for 10 min pre-swimming. Then, the mice were placed in the cylinder for 6 min after 24 h. During the last 4 min, the total immobility time was recorded. And those necessary posture for the mouse to keep its head above water were excluded.
Docking simulations
Molecular docking was carried out on Discovery Studio 2019 using the CDOCKER method with the crystal structure of PDE4 (PDB number: 4WCU), obtained from protein data bank (RSC).
Footnotes
Availability of data and materials
The data sets used and analyzed during this study are available from the corresponding author upon request.
Consent for publication
Written informed consent for publication was obtained from all authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was sponsored by the Postgraduate Research & Practice Innovation Program of Jiangsu Province (SJCX22_1314).
