Abstract
PDE4 inhibitors exhibit anti-stress and antidepressant-like abilities by catalyzing the hydrolysis of cAMP, a primary regulator for intracellular communication in the brain. Herein, novel diarylpyrazole derivatives are synthesized and investigated for their ability to inhibit PDE4. In vitro studies indicate that most of the synthesized compounds show significant potency for the inhibition of PDE4. Specifically, N-(4-(3-(3,5-dimethoxyphenyl)-1H-pyrazol-5-yl)benzyl)-2-morpholinoethan-1-amine exhibits the most potent PDE4 inhibition, with an IC50 value of ca. 0.09 μM. It also produces antidepressant-like activities in sugar water consumption and in forced swimming tests in vivo.
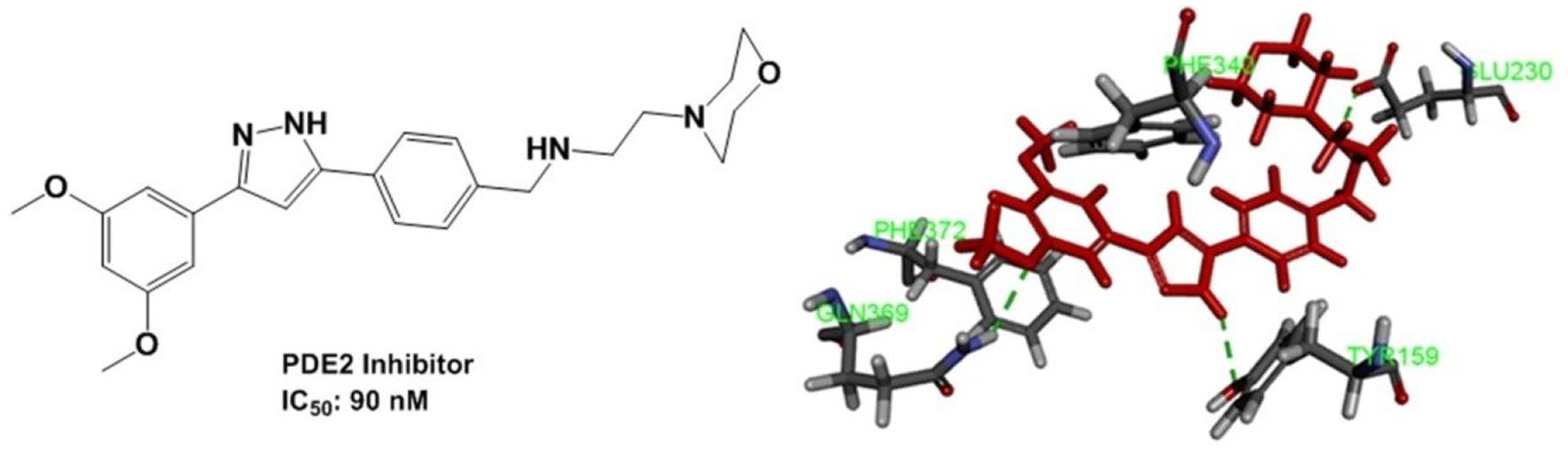
A series of novel resveratrol derivatives were synthesized and evaluated as PDE4 inhibitors against depression. Among the synthesized compounds,
Introduction
Depression is a serious psychiatric illness characterized by anxiety, irritable moods, cognitive sleep disorders, and so on.1,2 According to the World Health Organization, it is one of the leading causes of disability and the second largest contributor to the global burden of disease.3 –5 However, the etiopathology of depression presently remains unknown due to the complexity of its pharmacotherapy.6,7 Based on classic serotonin or noradrenaline hypotheses, several antidepressants such as chlorbipram hydrochloride, fluoxetine (Flu), and venlafaxine have been developed for the treatment of depression. However, all these drugs are slower to take effect and symptoms are only improved after several weeks of treatment.8 –12 Furthermore, according to previous reports, at least 40% of patients do not respond to these drugs. Therefore, the development of new and effective agents with fewer adverse effects is still needed. It has been shown that cAMP, a primary regulator of intracellular communication, plays an important role in the modulation of moods.13 –15 Its elevation can protect dopaminergic neurons from the toxic effects of MPP+ (1-methyl-4-phenylpyridinium) in vitro. 16 PDE4, a cAMP-specific hydrolyzing enzyme, is a potential target for the treatment of depression.17 –19 In fact, several PDE4 inhibitors, such as roflumilast, rolipram, and MK-0952, display antidepressant-like effects and cognitive enhancement.20 –22 However, there are still no PDE4 inhibitors available for the treatment of depression, although rolipram has been evaluated in a number of preclinical and clinical studies.
Resveratrol (Figure 1), a natural polyphenol in many plant-based foods, can alleviate depression-like symptoms induced by stress in rats by functioning as a PDE4 inhibitor. Furthermore, resveratrol can increase cAMP levels both in vitro and in vivo.23,24 Considering its chemical instability and low bioavailability, many resveratrol derivatives have been reported to overcome these problems.25 –27 Recently, we reported a novel resveratrol analogue (RES003) (Figure 1) via the introduction of a pyrazole group. It functions as a PDE4 inhibitor and ameliorates chronic-stress-induced depression-like behaviors. 28 Encouraged by these results, herein we report our continued studies on the synthesis of such resveratrol derivatives and the evaluation of their antidepressant potential both in vitro and in vivo.

The structures of resveratrol and RES003.
Results and discussion
The synthesis of compounds
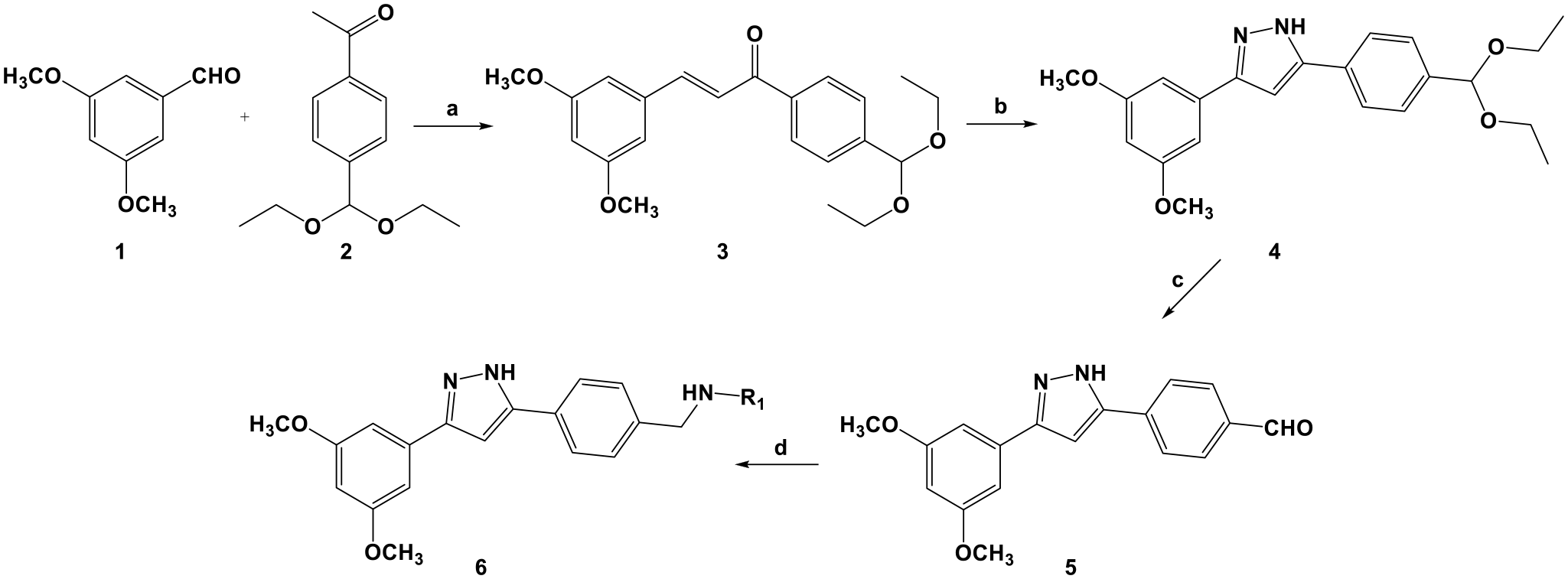
Synthesis of compounds

Synthesis of compounds
All the compounds were initially screened for their binding potency against PDE4 using rolipram as a positive control. The inhibitory activities are presented as IC50 (μM) values and the results are shown in Table 1. All of the derivatives showed better inhibitory activities against PDE4 than resveratrol. Our exploration was focused on introducing different substituents at the 4′-position of one benzene ring while keeping the remaining structures fixed. The results showed that the introduction of a substituent at the 4′-position had a significant influence on the inhibitory activity. According to the screening data, we found that compounds with linear or branched-chain substitution showed poor inhibitory activity against PDE4. For example, compounds
Structures and inhibitory activities toward PDE4 by the prepared resveratrol derivatives.

Results are expressed as the mean of at least three experiments.
PDE selectivity of compound

Results are expressed as the mean of at least three experiments.
In addition, a molecular docking simulation was performed to explore the interaction mode of compound

Binding mode of compound
Considering the importance of behavioral studies, two behavioral models, that is, the forced swim test (FST) and the sucrose preference test (SPT), were used to evaluate the antidepressant activities of compound
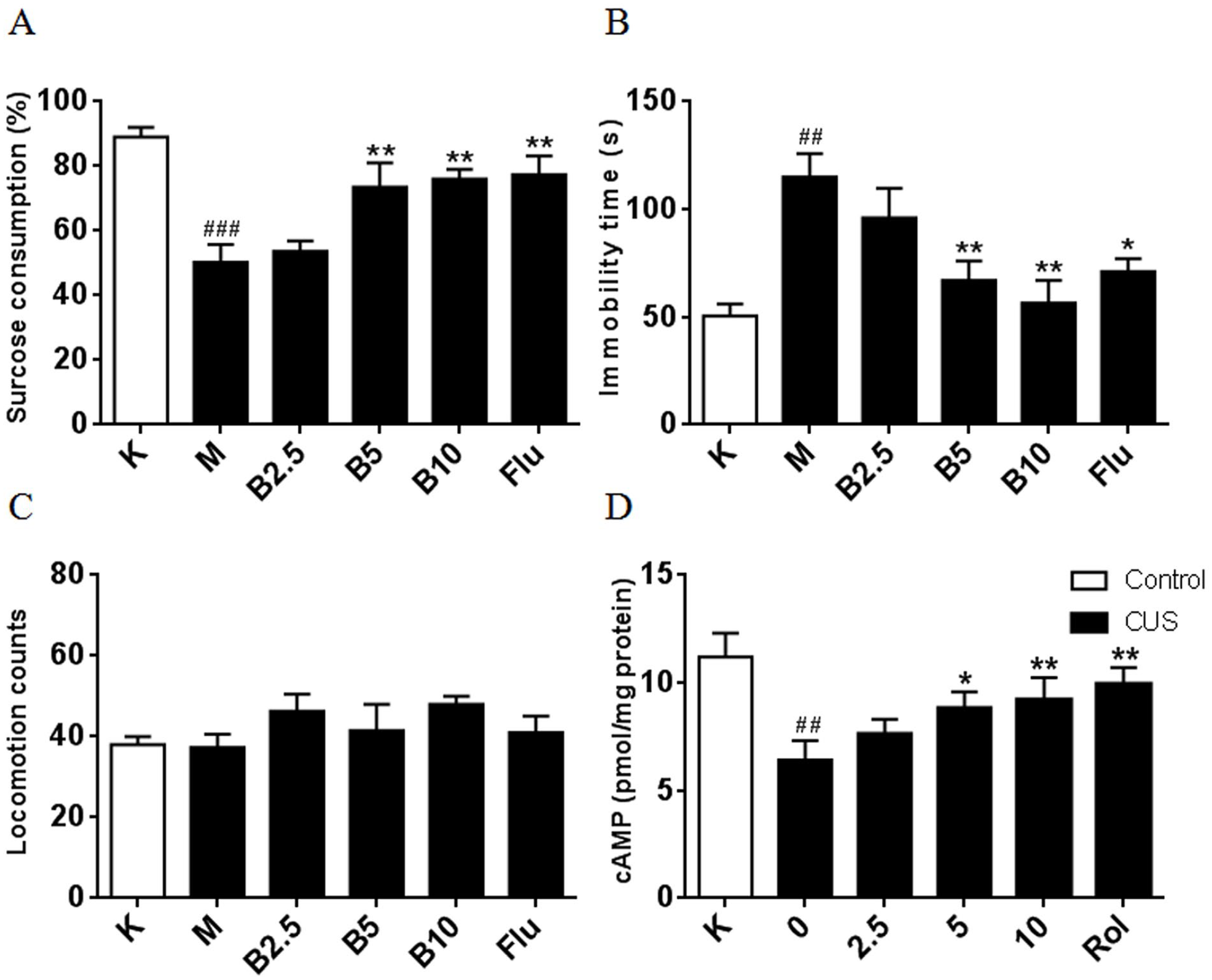
The antidepressant-like effects of compound
Conclusion
In summary, a series of resveratrol derivatives have been synthesized, with most of the products being potent inhibitors of PDE4. Compound
Experimental
Materials and reagents
All chemical reagents (analytical grade) were purchased from Shanghai Aladdin Bio-Chem Technology Co., LTD (Shanghai, China) and were used as received without any purification. Reactions were monitored by flash column chromatography on silica gel (Merck Kieselgel 60, 230-400 mesh).
1 H NMR and 13C NMR spectra were recorded at 400 MHz and 100 MHz, respectively, on a Bruker spectrometer in CDCl3 or DMSO-d6. MS spectra were acquired on a Thermo Scientific LTQ Orbitrap XL mass spectrometer. Melting points were obtained using a Boetius micro melting point apparatus.
Synthesis of 3,5-diarylpyrazoles 6a–f
(E)-1-(4-(Diethoxymethyl)phenyl)-3-(3,5-dimethoxyphenyl)prop-2-en-1-one (3 )
3,5-Dimethoxybenzaldehyde (
1 H NMR (400 MHz, CDCl3): δ = 7.53 (d, J = 8.6 Hz, 2H), 7.36 (m, 3H), 6.93–6.82 (m, 3H), 6.78 (m, 1H), 6.11 (s, 1H), 3.92 (s, 6H), 3.81 (q, J = 7.2 Hz, 4H), 1.26 (t, J = 7.2 Hz, 6H). 13C NMR (100 MHz, DMSO-d6): δ = 188.16, 159.87 (2C), 148.23, 142.65, 139.43, 135.36, 129.72 (2C), 125.37 (2C), 122.89, 106.67 (2C), 109.32, 100.27, 61.13 (2C), 56.35 (2C), 19.57 (2C). HRMS (ESI): m/z [M + H]+ calcd for C22H27O5: 371.1858; found: 371.1863.
5-(4-(Diethoxymethyl)phenyl)-3-(3,5-dimethoxyphenyl)-1H-pyrazole (4 )
A solution of compound
1 H NMR (400 MHz, DMSO-d6): δ = 7.55 (d, J = 8.7 Hz, 2H), 7.23 (m, 2H), 7.13 (d, J = 8.7 Hz, 2H), 6.92 (m, 1H), 6.78 (m, 1H), 6.12 (s, 1H), 3.91 (s, 6H), 3.83 (q, J = 7.2 Hz, 4H), 1.25 (t, J = 7.2 Hz, 6H). 13C NMR (100 MHz, DMSO-d6): δ = 158.97 (2C), 147.65, 145.71, 137.25, 135.13, 132.96, 129.12, 127.71 (2C), 106.82 (2C), 109.35, 100.68, 99.29, 58.63, 53.49 (2C), 37.86 (2C), 21.71 (2C). HRMS (ESI): m/z [M + H]+ calcd for C22H27N2O4: 383.1971; found: 383.1987.
4-(3-(3,5-Dimethoxyphenyl)-1H-pyrazol-5-yl)benzaldehyde (5 )
Compound
1 H NMR (400 MHz, DMSO-d6): δ = 9.92 (s, 1H), 8.07 (d, J = 8.5 Hz, 2H), 7.62 (d, J = 8.5 Hz, 2H), 7.13-7.11 (m, 2H), 6.97 (m, 1H), 6.85 (m, 1H), 3.89 (s, 6H). 13C NMR (100 MHz, DMSO-d6): δ = 191.32, 158.68 (2C), 147.12, 145.39, 139.16, 137.56, 135.13, 130.92 (2C), 127.13 (2C), 106.65 (2C), 100.68, 99.32, 53.16 (2C). HRMS (ESI): m/z [M + H]+ calcd for C18H17N2O3: 309.1239; found: 309.1226.
Synthesis of compounds 6a –f
Compound
N-(4-(3-(3,5-dimethoxyphenyl)-1H-pyrazol-5-yl)benzyl)cyclopentanamine (
N-(4-(3-(3,5-dimethoxyphenyl)-1H-pyrazol-5-yl)benzyl)cyclopentanamine (
4-(2-((4-(3-(3,5-Dimethoxyphenyl)-1H-pyrazol-5-yl)benzyl)amino)ethyl)phenol (
N-benzyl-1-(4-(3-(3,5-dimethoxyphenyl)-1H-pyrazol-5-yl)phenyl)methanamine (
N-(4-(3-(3,5-dimethoxyphenyl)-1H-pyrazol-5-yl)benzyl)-2-morpholinoethan-1-amine (
N-(4-(3-(3,5-dimethoxyphenyl)-1H-pyrazol-5-yl)benzyl)-1-(furan-2-yl)methanamine (
Synthesis of compounds 10a–g
Synthesis of intermediates 8a –g
A solution of p-hydroxyacetophenone (1.36 g, 10 mmol), the corresponding bromide (R2Br) (15 mmol), and K2CO3 (0.69 g, 5 mmol) in 50 mL of dry DMF was stirred at 80 °C for 5–6 h. After completion of reaction, the solution was added to a mixture of ice and H2O, and the resulting yellow solution was extracted with EtOAc (3 × 10 mL). The combined organic layers were washed with H2O, dried (Na2SO4), filtered, evaporated, and recrystallized from EtOAc.
1-(4-Ethoxyphenyl)ethan-1-one (
1-(4-(1-Hydroxy-2-methylpropoxy)phenyl)ethan-1-one (
1-(4-(2-Methoxyethoxy)phenyl)ethan-1-one (
1-(4-(4-Methoxybutoxy)phenyl)ethan-1-one (
1-(4-(Pent-4-en-1-yloxy)phenyl)ethan-1-one (
2-(4-Acetylphenoxy)-N-benzylacetamide (
1-(4-((3,5-Difluorobenzyl)oxy)phenyl)ethan-1-one (
Synthesis of chalcones 9
Compound
Synthesis of pyrazoles 10a –g
A solution of compound
3-(3,5-Dimethoxyphenyl)-5-(4-ethoxyphenyl)-1H-pyrazole (
1-(4-(3-(3,5-Dimethoxyphenyl)-1H-pyrazol-5-yl)phenoxy)-2-methylpropan-1-ol (
3-(3,5-Dimethoxyphenyl)-5-(4-(2-methoxyethoxy)phenyl)-1H-pyrazole (
3-(3,5-Dimethoxyphenyl)-5-(4-(4-methoxybutoxy)phenyl)-1H-pyrazole (
3-(3,5-Dimethoxyphenyl)-5-(4-(pent-4-en-1-yloxy)phenyl)-1H-pyrazole (
N-Benzyl-2-(4-(3-(3,5-dimethoxyphenyl)-1H-pyrazol-5-yl)phenoxy)acetamide (
5-(4-((3,5-Difluorobenzyl)oxy)phenyl)-3-(3,5-dimethoxyphenyl)-1H-pyrazole (
Enzymatic assay
All the enzymatic activities of PDEs were measured using 3H-cGMP or 3H-cAMP as the substrate. PDEs were first incubated with a buffer containing 50 mmol L−1 of Tris-HCl, pH 7.8, 10 mmol L−1 MgCl2, and 3H-cAMP or 3H-cGMP (40,000 cpm assay−1) at 24 °C for 30 min. The reaction was subsequently terminated by the addition of ZnSO4 and Ba(OH)2. The activity of the test compound was assessed by measuring the amount of 3H-cGMP or 3H-cAMP resulting from enzyme cleavage. The reaction product 3H-AMP or 3H-GMP was precipitated by adding BaSO4, while unreacted 3H-cAMP or 3H-cGMP remained in the supernatant. Radioactivity in the supernatant was measured in 2.5 mL of Ultima Gold liquid scintillation. The IC50 value of each inhibitor was measured at eight or more different concentrations of the inhibitor in the presence of 3H-cAMP or 3H-cGMP. Each measurement was repeated at least three times. The IC50 values were calculated by a nonlinear regression method using GraphPad Prism 5.0 Software.
Animals and treatment
Male ICR mice, weighing between 22 and 25 g, were obtained from the Animal Center at Changzhou University. All experiments were carried out according to the National Institute of Health Guide for Care and Use of Laboratory Animals (1996) and were approved by the Institutional Animal Care and Use Committee of Changzhou University (NO. Y20220057). Fluoxetine and rolipram were purchased from Sigma Chemical Co. (USA). The tested compounds and rolipram were dissolved in saline containing 5% DMSO, 5% solutol, and 90% saline. The positive control drug fluoxetine was dissolved in saline. Mice were subjected to intragastric administration (compound
The chronic unpredictable stress procedure (CUS) was carried out as described previously with minor modifications. 36 First, mice were housed individually for one day and subjected to chronic unpredictable stress for 10 days. Then, the animals were exposed to two different stressors each day including 2-h restraint, damp bedding, tilted cage, 10-min forced swimming and overnight illumination. All procedures were performed in isolated rooms adjacent to the housing room. Control mice were individually housed for the same period of time and were handled daily for 30 s in the housing room without being stressed.
After the CUS procedure, the sucrose preference test was assessed on the following day and then a forced swimming test was carried out the next day. Mice were treated with vehicle, compound
Behavioral tests
Sucrose preference test
The sucrose preference test was performed according to the previous description with minor modifications. 37 Before the procedure, animals were deprived of food and water for 14 h. Each animal was first habituated to drink from two bottles each filled with tap water for 24 h. Then, they were each given free access within another 24 h to two bottles, one containing a sucrose (1%) solution and the other tap water. The position of the bottles (right or left) was reversed 12 h later to avoid any lateralization bias on the part of the animal. Mice were not deprived of food during this period. Finally, the bottles were weighed and recorded before and after the sucrose preference test. The sucrose preference was calculated as the ratio of the consumed sucrose solution relative to the total amount of liquid consumed.
Forced swimming test
According to the previous description, animals were individually placed in glass cylinders (25 cm height, 10 cm diameter) containing 10 cm depth of water at 24 ± 1 °C for 6 min, allowing for free swimming. There was a 15-min pretest followed 24 h later by a 5-min test. A mouse was determined to be immobile when there were only small movements necessary to keep its head above water. The immobility time was recorded during the 5-min testing period.
Locomotor activity test
The locomotor activity (LA) test was assessed using an open-field chamber according to a previous procedure. 38 The floor of the chamber was divided into 16 identical squares. Mice were individually placed in the center of the chamber and allowed to acclimatize for 15 min. Their paws were contacted or disconnected from the active bars which can produce random configurations. The locomotor counts of mice were measured by counting the number of line crossings over 10 min.
cAMP concentration assay
The mice brain tissues were dissected and stored at −80 °C until analysis after the mice had been sacrificed. All the tissues were homogenized in ice-cold RIPA buffer containing 0.1% phenylmethylsulfonyl fluoride and centrifuged at 14,000 rpm for 20 min at 4 °C. The cAMP levels were measured using the mouse cAMP ELISA kit (ab133051, Biocompare, South San Francisco, CA) following the manufacturer’s instructions.
Docking simulations
Molecular docking of compound
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was sponsored by the National Natural Science Foundation of China (NSF 81970475), the Basic Public Research Program of Zhejiang Province (No. LBY21H030002) and the “Pioneer” and “Leading Goose” R&D Program of Zhejiang (2022C03145).
