Abstract
Activated carbon from cottonwood was synthesized by microwave-assisted method with activator NaOH. Activated carbon from cottonwood has a rough surface like coral, an amorphous structure with surface functional groups such as O–H, C–H, C=C, and C–O. The surface area and pore size were recorded at 395.48 m2 g−1 and 2.35 nm. The adsorption capacity of activated carbon from cottonwood was evaluated on two groups of anion dyes (congo red) and cation dye (methylene blue) to evaluate the influencing factors and predict the adsorption process. The best adsorption conditions were recorded for methylene blue (time: 30 min, temperature: 30 °C, pH: 10, activated carbon from cottonwood of 1 g L−1, methylene blue of 80 mg L−1, and adsorption capacity of 30.875 mg g−1) and congo red (time: 150 min, temperature: 60 °C, pH: 2, activated carbon from cottonwood of 2 g L−1, congo red of 100 mg L−1, and adsorption capacity of 45.97 mg g−1), respectively. The adsorption process was consistent with the Bangham kinetic and Dubinin–Radushkevich (D–R) isotherm model. The process of evaluating reusability shows that activated carbon from cottonwood can be reused 4 times.

Introduction
Today, with the strong development of modern society, people’s quality of life is increasingly improving. This has also led to an increase in environmental pollution, especially from industrial and residential activities. One of the pollutants that affects humans and aquatic animals is organic dyes. Organic colorants are widely present in industrial wastewater from the paper, textile industries, and so on. 1 The existence of colorants in wastewater can create many environmental problems with harmful effects of cytotoxicity, carcinogenicity, and so on. 2 Therefore, a series of studies have been widely applied and rigorously evaluated over the years to remove organic pigments from wastewater. There are many methods for treating colorants in wastewater including physical methods (adsorption, ion exchange, and ultrafiltration), chemical methods (flocculation and chemical oxidation), and biological methods (aerobic and anaerobic).3–5 In particular, the adsorption method on activated carbon has been attracting much attention to researchers. Although their potential has been demonstrated through adsorption processes, commercial activated carbons are produced from expensive and non-renewable fossil fuel sources, making activated carbon unsuitable for the environment, economic and environmentally unsustainable. Therefore, with high ligocellulose composition and low ash content, agricultural by-products are a suitable and cheap source of raw materials for research into activated carbon production.
In the textile industry, by-products from cotton (Ceipa pentandra) or Levant cotton (Gossypium herbaceum) are very large. According to The Food and Agriculture Organization (FAO), the world’s cotton growing area is 32.2 million hectares in 2020. The products after the processing are cotton fiber (24 million tons) and cottonseed oil (4.2 million tons). Cotton trees bring many applications and high economic value. The cotton fibers are used to stuff pillows or mattresses as well as an insulating layer for cold clothing. 6 Wood is used to make boats, canoes, wood carvings, musical instruments, fence posts, and furniture. 7 The seeds of the cotton plant are used to produce biodiesel. 8 The bark of the tree is used to make medicine for diabetes, anti-inflammation, or pain relief, 9 and the leaves are used as an antipyretic. 10 After the harvesting process, the bark, branches, and dry leaves of the tree will be cut off to allow the tree to grow. According to Renny Purnawati et al., 11 the main chemical components of the cottonwood plant are cellulose, hemicelluloses, and lignin. These components are present in the cotton plant stem: cellulose is the most abundant polysaccharide. In addition, by-products of cotton during the production process are used to produce activated carbon to treat pollutants in wastewater.12,13 In 2019, cotton by-products were used to synthesize activated carbon to treat Cr(VI) with activated carbon using FeCl3, by Zhihua. The recorded adsorption capacity was 267.12 mg g−1 according to the pseudo-second-order (PSO) model and the Langmuir isotherm model. 14 In 2020, Zhang et al.’s 15 group researched the synthesis of activated carbon from cottonwood (ACCW) to adsorb methylene blue (MB) dye. The process of synthesizing activated carbon is by chemical activation method with phosphoric acid as the activator. ACCW shows an ability to remove colorants of only about 9.2%. This shows that the processing ability is still limited while the activator helps the coal surface area reach 1020 m2 g−1. In 2021, Jie Cheng et al. 16 researched the synthesis of activated carbon from cotton stalk with a one-step and step-step synthesis process. The surface area was recorded as 1342.93 m2 g−1 with calcination at 600 °C. Activated carbon after synthesis is used as supercapacitors. Similar research by Panchanan, cotton stems are also used to synthesize activated carbon. Activated carbon is synthesized by activating with KOH and denatured with Al2(SO4)3·12H2O. The surface area was recorded as 2695 m2 g−1 and was used for CO2 adsorption. In addition to evaluating the ability to adsorb CO2, activated carbon from cotton is applied to an economic problem and the possibility of commercialization in practice. 17 In 2019, Kulumkan detailed research on ACCW trees. The activation process was carried out by heating the material at a temperature range of 500–800 °C, in which the raw material was pretreated with HCl before heating for 30 min. The research was carried out with different parts of the cotton plant. 18 It can be seen that cotton has been studied in recent years. Studies show that activated carbon was synthesized with chemical and physical processes. Economic problems and commercial possibilities are also studied. However, the synthesis saves costs to process, and the adsorption capacity improvement of coal is still quite limited. This is a gap for the research team to synthesize activated carbon using microwave-assisted chemical activation and apply organic dye treatment.
Experimental
Pretreatment and chemicals
Sodium hydroxide (99% purity) and hydrochloric acid (38% purity) were purchased from Xilong Chemical Co, Ltd. The synthetic dyes, MB, crystal violet (CV), brilliant green (BG), congo red (CR), methyl orange (MO), methyl red (MR), and Acid Yellow 17 (AY) were provided by Sigma-Aldrich Pte. Ltd. Cottonwood was washed with distilled water to remove adhering substances and dust and cut into small pieces. Then, the sample was dried at room temperature and ground to fine particles. These grounded grain flours were sieved to get uniform particles.
Preparation of activated carbon
In total, 50 g of cottonwood is impregnated in a 1:1 ratio with NaOH for 2 h. The mixture was dried at 100 °C for 24 h to remove any water. The mixture was then carbonized in a microwave oven at 600 W for 2 min. After activation, AC products were washed several times with HCl (1 M) and distilled water to remove excess NaOH until the pH remained constant. Finally, Activated carbon from cottonwood (ACCW) was dried at 110 °C for 24 h and stored in a desiccator for later use in the experiment.
Characterization
Surface morphology of ACCW was examined by scanning electron microscope (SEM) Hitachi S-4800 (Japan). Surface functional groups were measured by Fourier transform infrared (FTIR) FTIR6600 type A with wave number 400–4000 cm−1. X-ray diffraction (XRD) was used to analyze the structure of the material using a D8 Advance powder diffraction machine. Brunauer, Emmett, and Teller known as the BET equation are used to calculate the surface area and pore size of materials using the Gemini VII 2390 Series machine.
Adsorption experiment
The adsorption experiment was performed based on Quyen et al.’s 19 research with some changes to evaluate different influencing factors such as time, pH in solution, adsorbent dosage, dye concentration, and temperature. The factors are evaluated through the value of adsorption efficiency (H, %) and adsorption capacity (qe, mg g−1). The formula for calculating the value is as follows:
where C0 and Cf are the initial and equilibrium dye concentrations (mg L−1); V (L) is the volume of dye, and m (g) is the adsorbent dosage.
The acid/base surface and zeta potential measurement (pHpzc)
The pH point zero charge (pHpzc) was measured by the method reported in Thuan and Bich’s study with little variation.20,21 In total, 100 mL of 0.1 mol L−1 KCl solution was prepared in an Erlen flask and set at pH values from 2 to 10 (initial pH, pHi) with NaOH and HCl solutions. Add 0.1 g ACCW to the flask and shake for 3 h and leave to stabilize for 24 h at room temperature. Then, the activated carbon sample was separated from the mixture and the pH value (pH final, pHf) recorded. The pH value was determined using a Hanna Instruments HI2210-02 benchtop pH meter. pHpzc is determined at the point pHi = pHf in the graph of final pH value versus initial pH value.
Adsorption kinetic and isotherm model
The adsorption mechanism provides a way to predict the adsorption that takes place between the ACCW and the pigment. The kinetic and isothermal models are used as nonlinear models. The coefficients of the model are calculated along with the adjusted R2 coefficient. The adsorption kinetic models evaluated include pseudo-first-order (PFO) and PSO.21,22 The adsorption isotherm models evaluated include Langmuir, Freundlich, Dubinin–Radushkevich (D–R), Temkin. 21 The equations and formulas used are based on Quyen et al.’s research. 19
Reusability of AC
Reusability experiments were performed under optimal conditions. The washing process was evaluated by washing with solvents: 96% ethanol, acetone, H2O, a mixture of ethanol–H2O, and a mixture of acetone–H2O. We can choose the appropriate solvent to continue the washing process based on the adsorption capacity after washing the solvents. If the selected solvent is a mixture of ethanol–H2O or a mixture of acetone–H2O, then we wash according to the soaking ratios 20/80, 40/60, 50/50, 60/40, and 80/20. The evaluation process metrics are selected after the first reuse.
Results and discussion
Preparation and characterization of AC
SEM was used to analyze the surface morphology of activated carbon materials. Figure 1(a) scanned at 50 μm has a rough surface structure with pore formation as demonstrated in the study of Saad et al. 22 In Figure 1(b), with a scanned size of 5 μm, there are interconnected spheres of relatively uniform size and a smooth outer surface. This is thought to be due to the incomplete activation process during material synthesis as explained in the study of Sabar and colleagues. 23

Scanning electron microscope image of activated carbon from cottonwood at 50 μm (a) and 5 μm (b).
Determining the material structure by XRD method is shown in Figure 2(a), we see the appearance of sharp peaks at 29.44° indicating the maximum structure of amorphous carbon similar to that in Wang et al.’s 24 research. According to the research by Isahak et al. 25 the presence of NaOH along with materials with high cellulose content can remove water molecules from the sample to form highly porous activated carbon materials. This proves that ACCW was successfully synthesized via the microwave-assisted method with NaOH as the activator. Infrared spectroscopy (FTIR) analysis method was used to identify functional groups on the surface of activated carbon of cottonwood stem bark. The results of FTIR spectroscopy are shown in Figure 2(b), in which the absorption peak. The broad sub-wavelength at wavelength 3426 cm−1 represents the stretching vibration of the O–H group corresponding to the presence of free hydroxyl groups on the surface of the material. 12 The formation of O–H bonds is caused by the activation of the material. 26 The adsorption peak appears at wavelengths of 2879 and 877 cm−1 demonstrating the stretching vibration of the C–H groups of alkanes and alkenes.26,27 The functional group at the 1164 cm−1 adsorption peak is assigned to the formation of the C–O–C group of the ester. This was predicted in GeethaK and Shabudeen’s study. The adsorption peak at wavelength 1419 cm−1 belongs to the C=C group of aromatic or the O–H group of carboxylic acid. This may be due to the reaction formed between some carbons with strong activators such as NaOH and KOH as demonstrated in the study of Achmad Chafidz et al. 26 The adsorption peak at wavelength 1558 cm−1 is believed to be the formation of the C=C group of aromatic. The characteristics of this group were predicted in the study of Chafidz et al. 26 The adsorption peak at wavelength 648 cm−1 is assigned to the formation of C–Cl of the halogen group, which can be explained that they appear due to the interaction between the coal surface and residual Cl during the synthesis of the material. 28 OH, C–H, and C=C groups are functional groups related to the composition of cellulose in materials that play an important role in the adsorption process. 29

Fourier transform infrared (a) and X-ray diffraction (b) of activated carbon from cottonwood.
Multilayer adsorption method (BET) was used to determine the pore structure on the surface of activated carbon. The results of BET analysis showed that ACCW has a specific surface area of 395.48 m2 g−1, with a pore size of 2.35 nm. The BET multilayer adsorption diagram was shown in Figure 3. Looking through the diagram, we can see the adsorption isotherms of the AC material. The amount of adsorption increases markedly with the pressure difference from 0 to 0.2 and 0.8 to 1, which indicates the high porosity of the material. The isotherm of the material at the pressure difference from 0.4 to 0.9 indicates that the material has a pore distribution consisting of pores. The characteristics of the adsorption isotherm show that the material tends to increase along the second-order isotherm with the mechanism of gas molecules dispersing into the pores on the AC surface, creating an adsorption force with chemical bonding, geometry of the AC surface. The adsorption process is described by adsorption constants and adsorption energy parameters such as maximum adsorption energy and adsorption activation energy, according to isoline classification criteria, IUPAC adsorption heat. This result has been similarly proven in the research of Yuan et al. 30 and Chowdhury et al. 31 This shows that the activated carbon from the bark of the cottonwood tree has a high porosity consisting of pores with a relatively large surface area. This result shows that ACCW is a potential material to study the adsorption process to remove organic pigments.

BET diagram.
Evaluation of selective adsorption
Selective adsorption capacity was evaluated on seven types of cationic and anionic colorants, including MB, CV, BG, CR, MO, MR, and AY. The selectivity evaluation results shown in Figure 4 show that the material has good adsorption capacity for both cationic and anionic color groups. In the cationic color group, ACCW adsorbed best with MB colorant with an adsorption capacity of 11.6 mg g−1. In the anionic color group, ACCW adsorbed best with CR colorant with an adsorption capacity of 16.39 mg g−1. Therefore, we will choose MB and CR to evaluate the influencing factors and reusability.
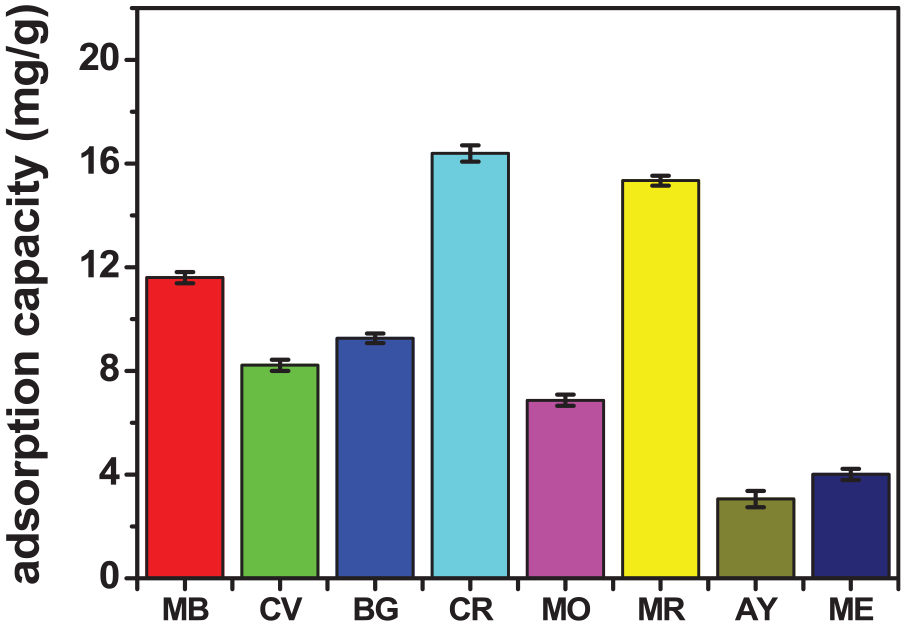
Assessment of selective adsorption capacity.
Adsorption studies
The effect of time on the adsorption capacity of ACCW for MB and CR was conducted with time intervals from 0 to 180 min and from 0 to 210 min, respectively. According to the results in Figure 5(a), the adsorption capacity of ACCW with MB dye increased from 0 to 10.547 mg g−1 in the first time period from 0 to 10 min. The time from 10 to 20 min showed that the adsorption capacity increased from 10.547 to 11.32 mg g−1; the time from 20 to 30 min showed that the adsorption capacity continued to increase from 11.32 to 11.59 mg g−1; and after this period of time from 30 to 180 min, the adsorption capacity did not change. It can be said that the adsorption process has reached an equilibrium state. As for CR pigment, the adsorption capacity increased rapidly from 0 to 13.41 mg g−1 in the period from 0 to 90 min. From 90 to 120 min, the adsorption capacity decreased from 13.41 to 13.24 mg g−1; from 120 to 150 min, the adsorption capacity increased to 14.78 mg g−1; and then during the period from 180 to 210 min, the difference in adsorption capacity was negligible. It can be seen that the adsorption process has reached an equilibrium state. Therefore, the adsorption time chosen as the optimal value for the next experiments with MB dye is 30 min and CR is 150 min. The effect of temperature on the adsorption capacity of ACCW was conducted at different temperatures (30, 40, 50, and 60 °C). According to Figure 5(b), with MB colorant at temperatures from 30 to 60 °C, the adsorption capacity does not change significantly. This proves that the adsorption capacity of ACCW with MB dye is not much affected by temperature. This also shows that ACCW can be used to process MB pigment at high temperatures. However, we choose a temperature of 30 °C because this is the closest temperature range to room temperature, besides helping us save energy for conducting surveys of the next factors. With CR colorants, when the temperature increases from 30 to 60 °C, the adsorption capacity increases due to the increased disorder between the colorant molecules, making it easier for the material to adsorb inside. This has also been proven in Ganesan et al.’s 32 study. Therefore, the adsorption temperature selected to remove MB and CR dyes is 30 and 60 °C, respectively.
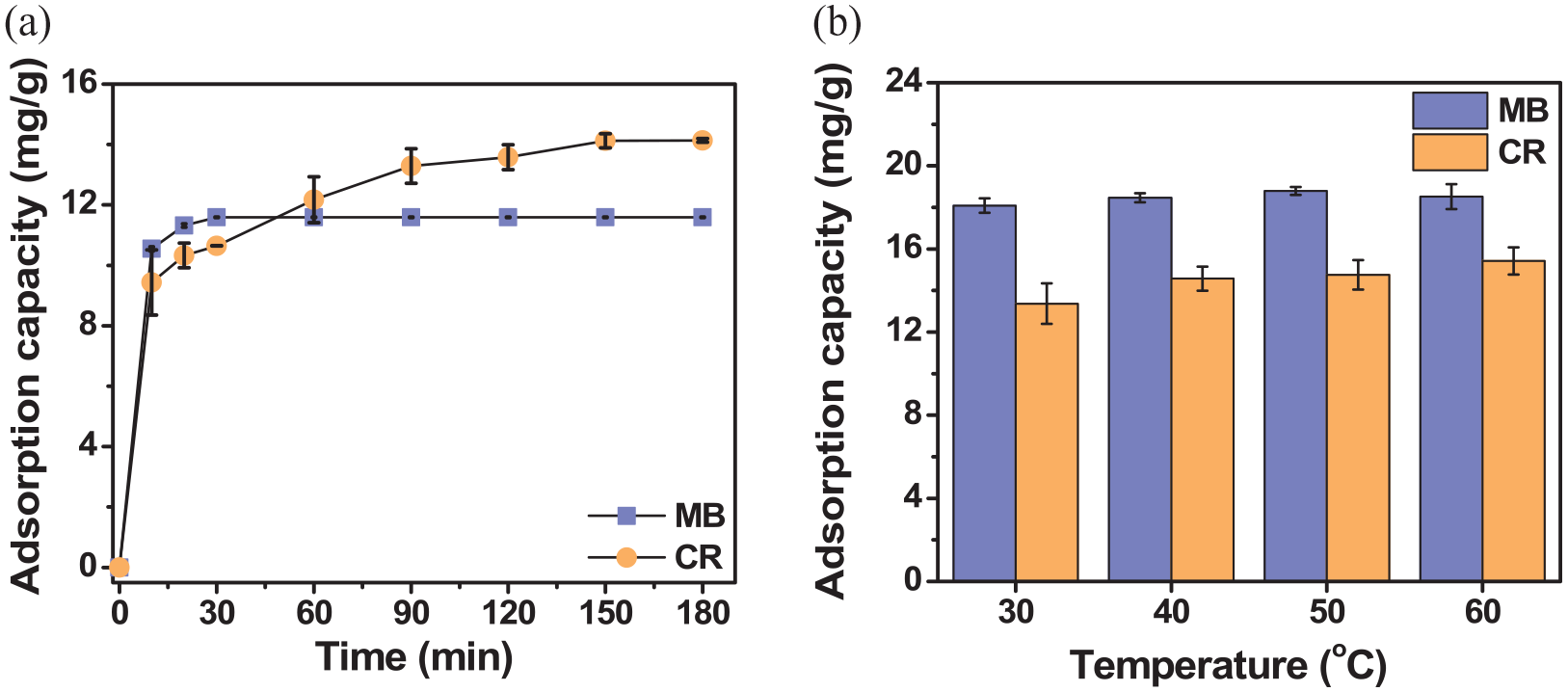
The effects of time (a) and temperature (b) of activated carbon from cottonwood.
In A. Azari’s research, it was shown that solution pH is an important parameter in the adsorption process. It is related to the surface charge of the adsorbent and the degree of ionization of the dye molecules. pHpzc represents the pH value at the point of zero charge and the cation and anion exchange capacity on the surface of the adsorbent. When the solution pH is lower than pHpzc, the AC surface is positively charged. In contrast, when the solution pH is higher than pHpzc, the activated carbon surface is negatively charged.33,34 To evaluate the influence of pH on adsorption capacity, the pHpzc experiment was conducted. The pHpzc result in Figure 6(a) shows that the ACCW material gives a result of 4.165. As shown in Figure 6(b), the solution pH of MB gives the highest adsorption capacity at pH 10 (19.9 mg g−1) and the solution pH of CR gives the highest adsorption capacity at pH 2 (19.771 mg g−1). Figure 6(b) shows that the MB’s adsorption improved as the pH of the solution increased. At pH 4 and above, the adsorption capacity is almost stable. MB adsorption in neutral and basic environments is higher than in acidic environments. An excess of OH− ions in the medium and the cationic dye’s structure can be suggested as causes. 35 For CR dye, the adsorption capacity decreased with increasing pH value from acidic to neural environments. The results showed the best CR removal ability at pH 6. The CR dye’s molecules have a negative charge in solution from the sulfonic groups (SO)3− possibly due to electrostatic interactions or diffusion into the pores of ACCW. To explain the adsorption mechanism, ACCW will be evaluated by adsorption kinetic and isothermal models. Therefore, pH 10 and pH 2 are the best results chosen to evaluate the next experiments on the adsorption capacity of ACCW with MB and CR dyes.

The effects of pHpzc (a) and pH (b) of activated carbon from cottonwood.
The results of investigating the effect of content on the adsorption capacity of ACCW with MB and CR pigments are presented in Figure 7. It can be seen that as the AC content increases, the adsorption capacity decreases. This is explained through the formula for calculating the adsorption capacity, so to choose the ACCW content we must rely on the adsorption efficiency to obtain the optimal conditions. According to Figure 7(a), it shows that the content ratio of ACCW is inversely proportional to the adsorption efficiency of 0.1 g L−1 (64.83 mg g−1; 25.99%), 0.5 g L−1 (41.03 mg g−1; 82.23%), 1 g L−1 (23.17 mg g−1; 92.88%), 2 g L−1 (12.47 mg g−1; 100%), and 3 g L−1 (8.32 mg g−1; 100%). Therefore, the adsorbent content of ACCW to remove MB pigment is 1 g L−1. Figure 7(b) shows that the content ratio of AC for CR colorant is similar to MB colorant 0.1 g L−1 (50.17 mg g−1; 25.56%), 0.2 g L−1 (18.48 mg g−1; 47.07%), 1 g L−1 (14.29 mg g−1; 72.8%), 2 g L−1 (9.53 mg g−1; 97.08%), and 3 g L−1 (6.42 mg g−1; 98.17%). Thus, the content of 2 g L−1 is the content with good adsorption capacity of the material to remove the CR colorant. From the above results, the content of ACCW to remove MB and CR pigments is 1 and 2 g L−1, respectively, and will be chosen for the next experiments.

The effects of activated carbon from cottonwood dosage (a) and dyes concentration (b).
The results of investigating the influence of solution concentrations of MB and CR dyes on the adsorption capacity of ACCW are presented in Figure 7(b). With the solution concentration of MB colorant, the concentration increases from 10 to 80 mg L−1 (4.435–31.858 mg g−1), while with CR colorant the concentration increases from 10 to 100 mg mL−1 (4.148–46.602 mg g−1). As the concentration of the two pigments increases, the disturbance of the molecules in the solution increases. This makes the interaction between the colorant molecules and the active sites on the surface of the material easier. With the solution concentration of MB colorant, the adsorption capacity decreased from 31.858 to 31.608 (80–100 mg L−1), and with the solution concentration of CR colorant, the adsorption capacity decreased from 46.602 to 17.867 mg g−1 (100–200 mg L−1). This shows that when the color molecules increase to a certain level, the adsorption process is hindered, so the adsorption capacity does not change. In summary, the best adsorption capacity of ACCW was recorded with MB dye (time: 30 min, temperature 30 °C, pH: 10, content: 1 g L−1, and concentration: 80 mg L−1), and with CR colorant (time: 150 min, temperature: 60 °C, pH: 2, content: 2 g L−1, and concentration: 100 mg L−1). This is the best adsorption condition selected to evaluate the material reuse process.
Adsorption kinetic and isotherm model
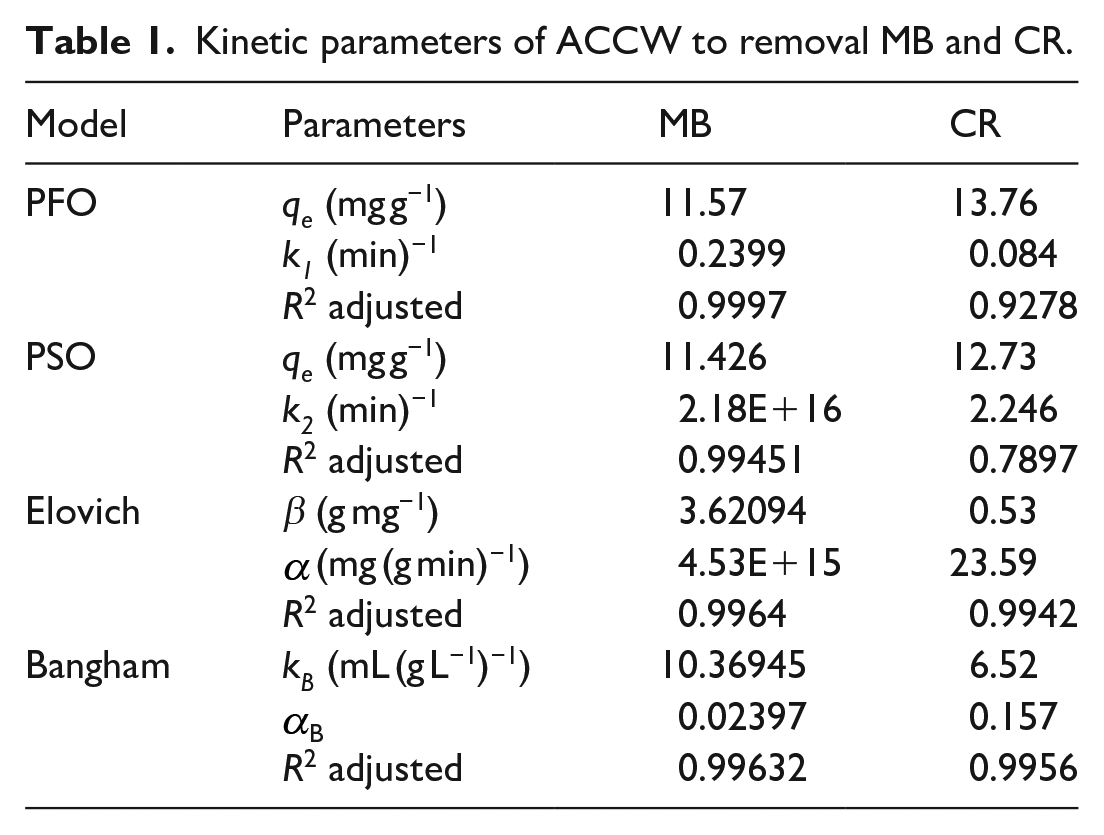
The adsorption kinetic model was used to predict the adsorption process of activated carbon. Applying this model can help optimize parameters in the adsorption process such as adsorption rate and adsorption capacity, and optimize the size and structure of the adsorbent to increase adsorption capacity. The parameters obtained from the adsorption kinetic model of ACCW with MB dye have been summarized in Table 1. The diagrams for the first-order pseudo-adsorption kinetics, second-order pseudo-adsorption kinetics, Elovich kinetics, and Bangham kinetic models are presented in Figure 8(a). The first-order pseudo-adsorption kinetic models, Elovich kinetics, and Bangham kinetics are suitable for the adsorption process of the material with a high correlation coefficient of 0.9997, respectively, 0.9964 and 0.9963. The results show that the adsorption of MB on ACCW is mainly controlled by the physical adsorption mechanism. Besides, the process of intragranular diffusion and diffusion on the surface of the material is also one of the processes that need attention due to the high level of model suitability.
Kinetic parameters of ACCW to removal MB and CR.

Adsorption kinetics of activated carbon from cottonwood with methylene blue (a) and congo red (b).
For the CR colorants summarized in Table 1, the diagrams for the first-order pseudo-adsorption kinetics, second-order pseudo-adsorption kinetics, Elovich kinetics, and Bangham kinetics are presented in Figure 8(b). The Bangham kinetic model and the Elovich kinetic model fit well with the adsorption data on the ACCW surface as demonstrated by the correlation coefficients of 0.9956 and 0.9942, respectively. It is demonstrated that the adsorption of CR on ACCW is controlled by the mechanism of intraparticle diffusion and diffusion on the surface of the material. However, the intraparticle diffusion mechanism largely accounts for controlling the adsorption process of CR on ACCW with a higher correlation coefficient of 0.9956. It can be seen that between the two pigments MB and CR, intragranular diffusion and diffusion on the surface of the material take place.
The adsorption isotherm model is used to determine the adsorption capacity and to explain the mechanism of the incorporation of color molecules into the adsorbent and the relative affinity of the color molecules toward the adsorbent. The Langmuir, Freundlich, Temkin, and D–R adsorption isotherm models are presented in Figure 9(a). The parameters obtained from the adsorption isotherm model of ACCW with MB dye are summarized in Table 2. It can be seen that Langmuir isotherm model and Temkin isotherm model appropriately describe MB adsorption on ACCW with correlation coefficients of 0.9842 and 0.9859, respectively. The results show that the ACCW adsorption process is a monolayer adsorption, in which the Temkin isotherm model has the highest correlation coefficient of 0.9859, which shows that electrostatic interactions occur and the uniformity of pores on ACCW surface plays an important role in the process, MB adsorption. The results are similar to the study of Osman Üner and Geçgel Bayrak. 36

Adsorption isotherm of activated carbon from cottonwood with methylene blue (a) and congo red (b).
Isothermal parameters of ACCW to removal MB and CR.

The Langmuir, Freundlich, Temkin, and D–R adsorption isotherm models are presented in Figure 9(b). The parameters obtained from the adsorption isotherm model of ACCW with CR dye are summarized in Table 2. It can be seen that the isotherm model that best describes the adsorption process of ACCW materials is the D–R isotherm model, which is demonstrated by the highest correlation coefficient (R2 = 0.9559). This shows that the adsorption process of the material is controlled by the adsorption mechanism with the Gaussian energy distribution on the heterogeneous surfaces. Furthermore, it is reported by Ibrahim and Sani (2014) that when the E value is below 8 kJ mol−1, the adsorption process can be considered as physical. 37
Reusability of AC
The washing and reuse process is done to increase efficiency and save costs in the process of using activated carbon. In this study, the washing process was evaluated using solvents: ethanol, acetone, water, acetone mixed with water, and ethanol mixed with water. According to the results in Figure 10(a) and (b), the ability to wash MB and CR on ACCW with alcohol mixed with water with the highest adsorption capacity is 31.65, 52, and 45 mg g−1, respectively. To ensure effective washing and save solvent during the process, choosing the appropriate solvent-soaking ratio is important. So, alcohol-based solvents are made in the proportions of 20:80, 40:60, 50:50, 60:40, and 80:20. The appropriate soaking ratio for washing MB and CR on ACCW with adsorption capacity is 32.77 and 45.68 mg g−1, respectively, corresponding to a ratio of 80: 20 and 20:80. Therefore, the washing process is performed using alcohol mixed with water with the solvent soaking ratio for MB washing process being 80:20 and CR being 20:80.

Washing process of methylene blue and congo red (a) with ethanol, water, acetone, ethanol 50%, and acetone 50%; percent ethanol solvent mixed with water of MB and CR (b).
Based on the soaking process, we can choose the appropriate solvent and soaking rate to continue the reuse process. The reuse experiments were repeated many times, and the results are shown in Figure 11. For MB colorant, after six reuses, the adsorption capacity decreased from 27 to 17.28 mg g−1. This also shows that the material lost about 36% of its adsorption capacity. For CR pigment, after five reuses, the adsorption capacity decreased from 45.115 to 20.7 mg g−1. This also shows that the material has lost about 54.12% of its adsorption capacity. Because the material’s CR adsorption capacity decreased by 50%, the reuse evaluation process stopped. It can be seen that, for CR colorants, the material can only be reused up to 5 times, but for MB pigments, the ability to reuse is more.

Number of reuses of activated carbon from cottonwood through the washing process of methylene blue and congo red.
Conclusion
Cotton stalk bark by microwave-assisted method with activator NaOH was synthesized into activated carbon. The structural features were clearly shown through analytical methods such as SEM, FTIR, BRT, and XRD. ACCW has an amorphous structure with a rough surface like coral along with surface functional groups such as O–H, C–H, C=C, and C–O. The surface area and pore size were recorded at 395.48 m2 g−1 and 2.35 nm, indicating potential in organic pigment adsorption. The adsorption capacity of ACCW was evaluated on two groups of negatively charged and positively charged dyes. In particular, MB and CR were selected to evaluate the influencing factors and predict the adsorption process. The results showed that the best MB organic dye removal ability occurred in the following conditions: time: 30 min, temperature: 30 °C, pH: 10, ACCW content: 1 g L−1, and color concentration: 80 mg L−1 with a recorded adsorption capacity of 62.94 mg g−1 The adsorption process is consistent with the PFO kinetic and the Temkin isotherm model. The best removal of CR organic dye occurred at time: 150 min, temperature: 60 °C, pH: 2, ACCW content of 2 g L−1, and color concentration of 100 mg L−1 with an adsorption capacity of 62.69 mg g−1. The adsorption process is consistent with Bangham kinetic and D–R isotherm model. To enhance the value of AC, the reusability of the material has also been evaluated. For CR pigments, the ability to reuse materials is 5 times, and for MB pigments, the ability to reuse materials is more than 5 times. It has good adsorption capacity and high reusability, helping to reduce production costs and can treat a variety of pollutants. To be able to evaluate the possibility of practical application, ACCW needs to evaluate its adsorption capacity for real wastewater and water treatment systems. In summary, ACCW is a material that has the potential to replace commercial activated carbon.
Footnotes
Author contributions
Original draft preparation: H.N.B. and H.N.T.H; data curation: H.L.Q. and C.S.P.; methodology: T.C.Q.N. and H.L.Q.; review and editing: T.T.T. and H.N.B. All authors have assented to the published version of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by Nguyen Tat Thanh University, Ho Chi Minh city, Vietnam, with code (2023.01.159/HĐ-KHCN).


