Abstract
Polyether is a synthetic polymer that has low toxicity and, therefore, it is used in many applications. It can be used as a detergent, emulsifier, drug excipient, pesticide emulsifier, antiseptic, and papermaking additive agent. This study outlines the synthesis, characterization, and fluorescence evaluation of polyethers derived from curcumin analogs. Condensation polymerization of curcumin analogs and dibromoalkanes produces the corresponding polyethers. The Fourier-transform infrared and proton nuclear magnetic resonance spectroscopy confirmed the structures of the newly synthesized polyethers. The thermal stability and solubility of the synthesized polyethers of various solvents were investigated. They showed high stability at high temperatures as thermoplastics. The synthesized polyethers have fluorescent properties in solutions, and the absorption and intensity were dependent on the type of functional groups present in the polymers. The fluorination intensity of polyethers was found to be dependent on the presence of electron-donating groups, the pH of the solution, and the type of solvent used. One of the synthesized polyethers exhibited strong emissive properties at one or two specific wavelengths.

Introduction
Turmeric root contains a primary curcuminoid called curcumin, which has a chemical structure of diferuloylmethane. The curcuma plant (Curcuma longa L.) belongs to the ginger family (Zingiberaceae) and is a perennial herb. The turmeric plant produces a rhizome that is used as a spice, colorant, and medicinal herb, often as a substitute for ginger. Turmeric became a key component of Ayurvedic medicine in 500 BCE. Curcumin, demethoxycurcumin, and bisdemethoxycurcumin are the most significant curcuminoids. The benefits of curcumin are diverse and include antioxidant, antiamyloid, antimicrobial, antitumor, immune-modulating, and neuroprotective properties. 1 Moreover, curcumin has been shown to have antidepressant properties by regulating the level of serotonin and dopamine release in the brain.2,3
Polyethers have high stability and flexibility since they contain repeated C–O–C bonds that are stable and flexible. 4 Two types of polyethers are known as natural and synthetic polymers. Natural polyethers include linear and polycyclic polymers. On the contrary, synthetic polyethers received particular attention due to their structural diversity and antibiotic and antifungal properties.5,6 Polyethers have good thermal and chemical stability and can be used in multiple applications. High molecular weight polyethers are synthesized through condensation polymerization using the Williamson ether method, which involves the nucleophilic substitution of halogenated alkanes. Although such a method is highly efficient, side products are generated (e.g. hydrochloric acid), which are toxic and reduce the sustainability of the process. 7 Polyethers are technically classified as thermoplastic and thermosetting polymers. Thermoplastic polyethers are a popular choice across different industries due to their sustainability, ease of recyclability, and high resistance to environmental factors. In addition, they can be welded using other processes, depending on the application, instead of using adhesive bonding or mechanical fixation. Non-malleable polyethers are involved in many industrial applications due to their high strength, hardness, and excellent thermal and chemical properties, which are attributed to the cross-linked nature of the polymeric chains.8,9
Fluorescent polymers emit light when excited by electric current or ultraviolet (UV) light. 10 They have several potential applications for new display technology. The industrial applications of conjugated polymers, known as photoluminescence (PL) and electroluminescence (EL) materials, are limited because of their poor solubility in most organic solvents. 11 Introducing flexible spacers into the main polymeric chains enhances the solubility of conjugated polymers. Highly or fully conjugated fluorescent polymers have been widely studied. 12 Therefore, the current work reports the synthesis and structural characterization of new polyethers derived from chalcone monomers and dihaloalkanes. In addition, their thermal behavior and ability to exhibit fluorescence properties have been investigated.
Results and discussion
Synthesis of A1–G1 and A2–G2
Curcumin analogs A–G were synthesized in good yields (Table 1) based on previously reported synthetic procedures (Scheme 1).13,14
Structure, yield, and melting point of curcumin analogs A–G.

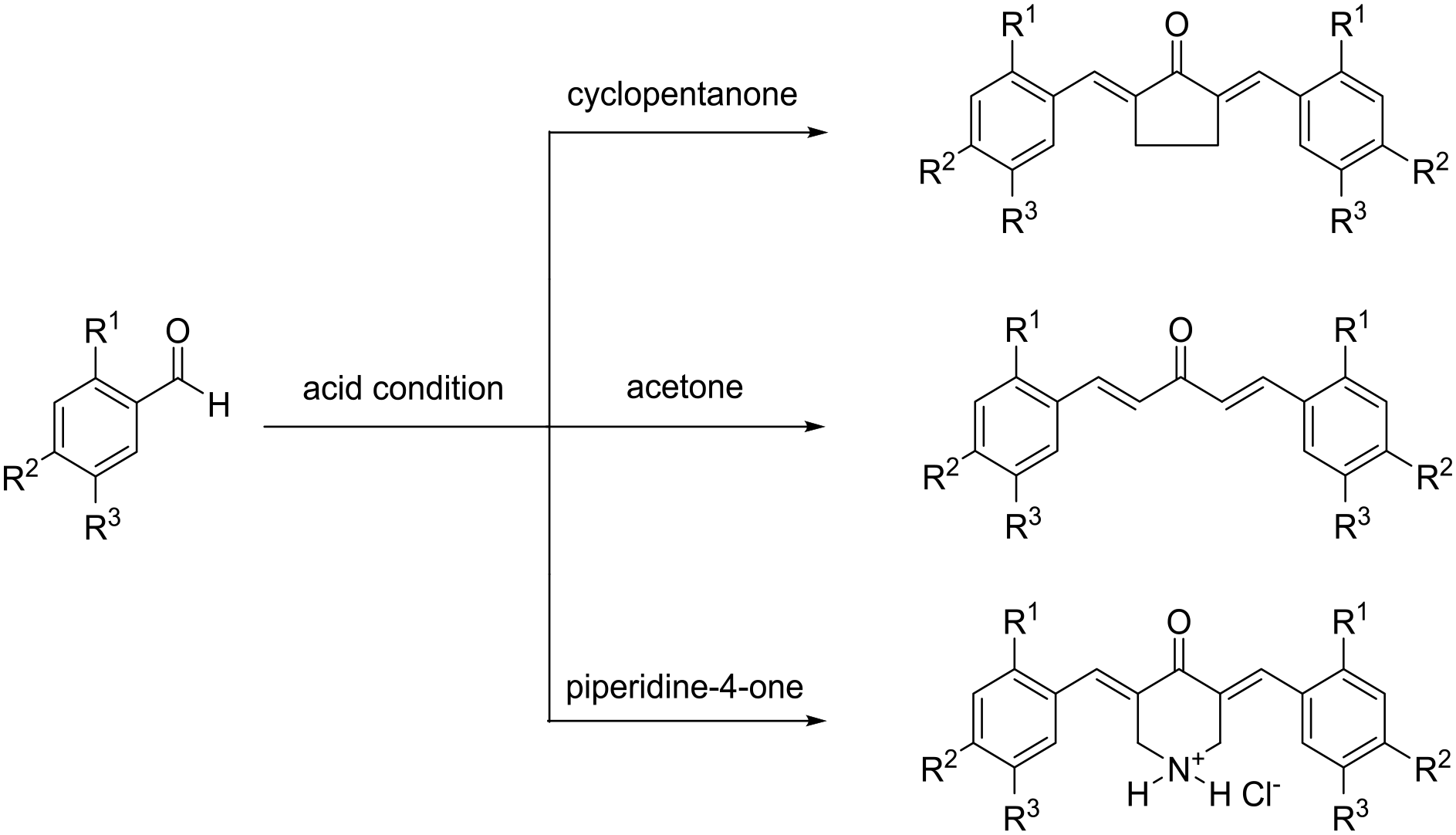
Synthetic routes for curcumin analogs A–G.
Polyethers A1–G1 (Scheme 2) were synthesized based on a modified literature procedure. 15 Reactions of chalcone (A, C, D, F, or G) and 1,4-dibromobutane in N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO) under basic conditions led to the production of the corresponding polyethers in good yields (Table 2).
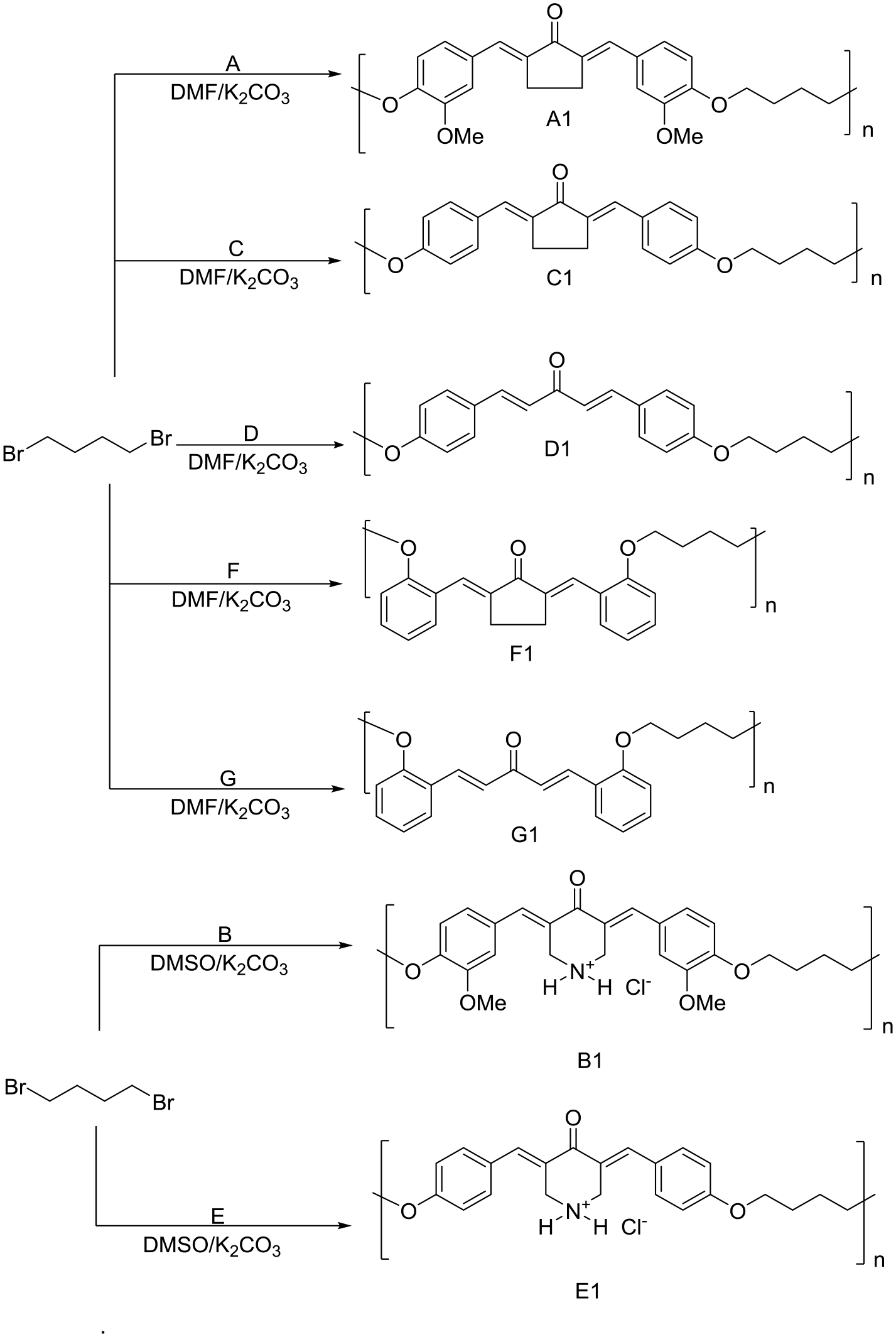
Synthetic routes for polyethers A1–G1.
Color and yield of polyethers A1–G1 and A2–G2.
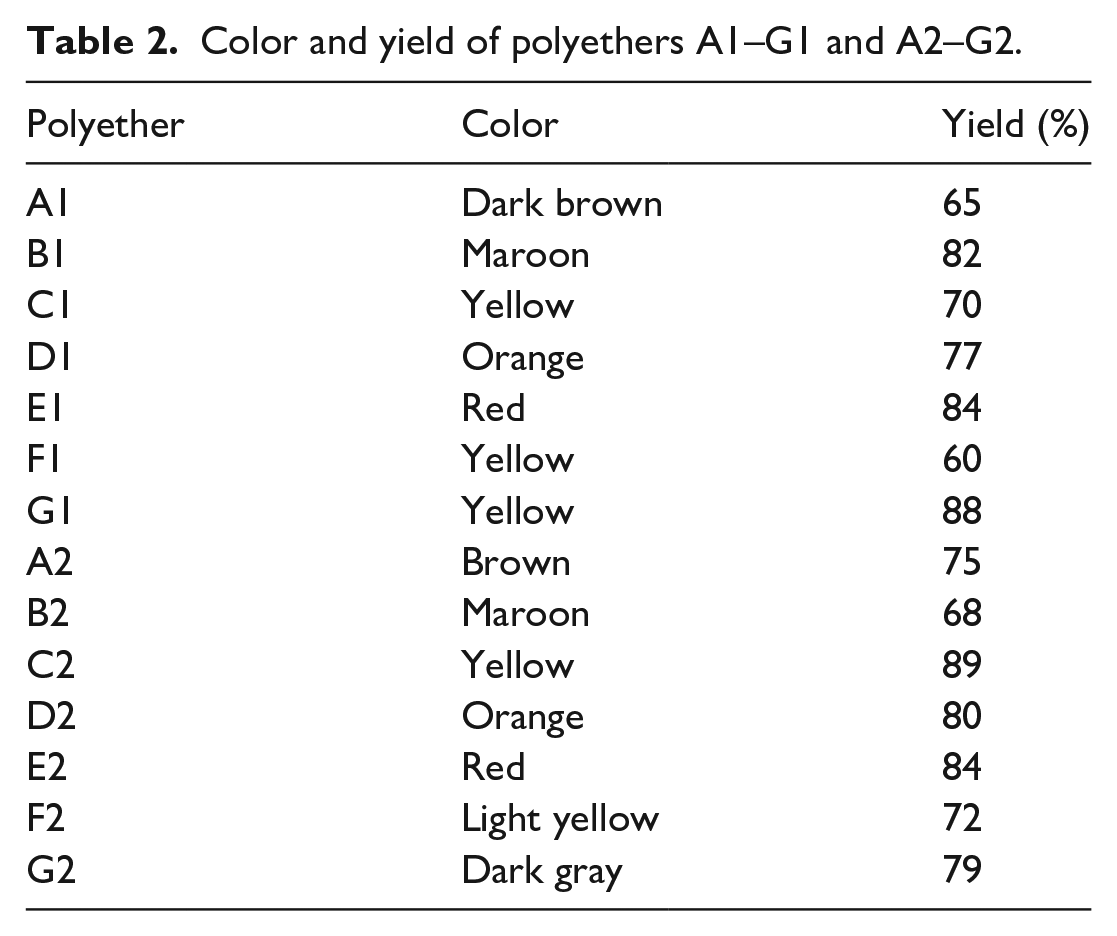
Polyethers A2–G2 (Scheme 3) were synthesized using a similar procedure to that used to produce A1–G1 (Scheme 2). Table 2 summarizes the color and yield of A2–G2.

Synthetic routes for polyethers A2–G2.
Fourier-transform infrared spectra of polyethers A1–G1 and A2–G2
The Fourier-transform infrared (FTIR) spectra of A1–G1 and A2–G2 (Supplementary Materials) showed a band at 3010–3111 cm−1 corresponding to the stretching vibration of the aromatic C–H bond (Table 3). The stretching vibration for the aliphatic C–H bond appeared at the 2911–2945 cm–1 region. A strong band has been seen in the 1620–1688 cm–1 region due to the C=O group stretching vibration. The stretching vibrations for the C=C of the aromatic rings and the C–O–C appeared at 1513–1601 cm−1 and 1220–1282 cm−1, respectively. The appearance of the latter band is evidence of the formation of ether linkages in the polymeric chains. 16
Some FTIR spectral data (ν, cm−1) of polyethers A1–G1 and A2–G2.

Nuclear magnetic resonance spectra of polyethers A1–G1 and A2–G2
The 1H NMR spectra (Supplementary Materials) showed the protons corresponding to the repeating units containing chalcone, methylene, and butylene moieties. They show the absence of an exchangeable singlet due to the OH protons, which appeared at a low field (around 9.86 ppm) for dihydroxy chalcones (monomeric units; A–G). In addition, they show the presence of O–CH2–O protons (6.09–5.81 ppm) for polyethers A2–G2 (Scheme 3) due to the substitution of the OH group in monomers. Such observation provides clear evidence for the formation of polyethers A1–G1 and A2–G2. The aromatic protons appeared within the expected chemical shifts (7.96–6.83 ppm). Assigning the OCH2CH2CH2CH2O protons in polyethers A1–G1 proved challenging since their 1H NMR spectra are complex.
Thermal stability of polyethers A1–G1 and A2–G2
Thermal stability is one of the main features of polymers since their properties are changed as a function of temperature. 14 Thermal decomposition of polymers has occurred due to partial fragmentation of polymeric chains due to exposure to high temperatures. Pyrolysis depends on the nature and composition of polymers. The high thermal stability of polymers enables them to resist changes in the polymeric chains when exposed to heat.17,18 The thermal stability of polyethers A1–G1 and A2–G2 was assessed to measure many essential functions to understand the behavior of these polymers. The initial degree of dissociation (Ti), the final degree of dissociation (Tf), the weight loss (Wt %), and the remaining polymer following the Char residue process were measured (Table 4). 19
TGA data of polyethers A1–G1 and A2–G2.

The thermogravimetric analysis (TGA) of polyethers A1, B1, F1, A2, and D2 showed one stage of decomposition at 166–405 °C. In contrast, the other polyethers had two stages of decomposition, the first one at 155–318 °C and the second at 318–465 °C. For B2, three stages of decomposition took place at 102, 223, and 415 °C. The synthesized polyethers have different decomposition rates and high stability. The differences in the decomposition stages are due to chemical structure variations in chalcone units within the polyethers. The high stability of polymers can be attributed to their high aromatic contents. The high residual polymers indicate that the synthesized polyethers have a high thermal stability. The first phase of weight loss can be attributed to the removal of small molecules (e.g. water), and the second one can be due to the removal of larger molecules (e.g. aromatic rings and polyether fragments from the polymer backbone). The other remaining materials above the final temperature include non-volatile ash.20,21 The activation energy (Ea) was determined using the Broido 1965 model, as shown in equations (1) and (2) 22


where Wi is the polyether initial weight, Wt is the weight at any temperature of the polymer, W ∞ is the final weight of the polymer, R is a general constant of gases, and T is the temperature range measured at Wt; plotting graphs between ln[ln(1/y)] versus 1/T gives a straight line. The slope is related to the activation energy.
Solubility of polyethers A1–G1 and A2–G2
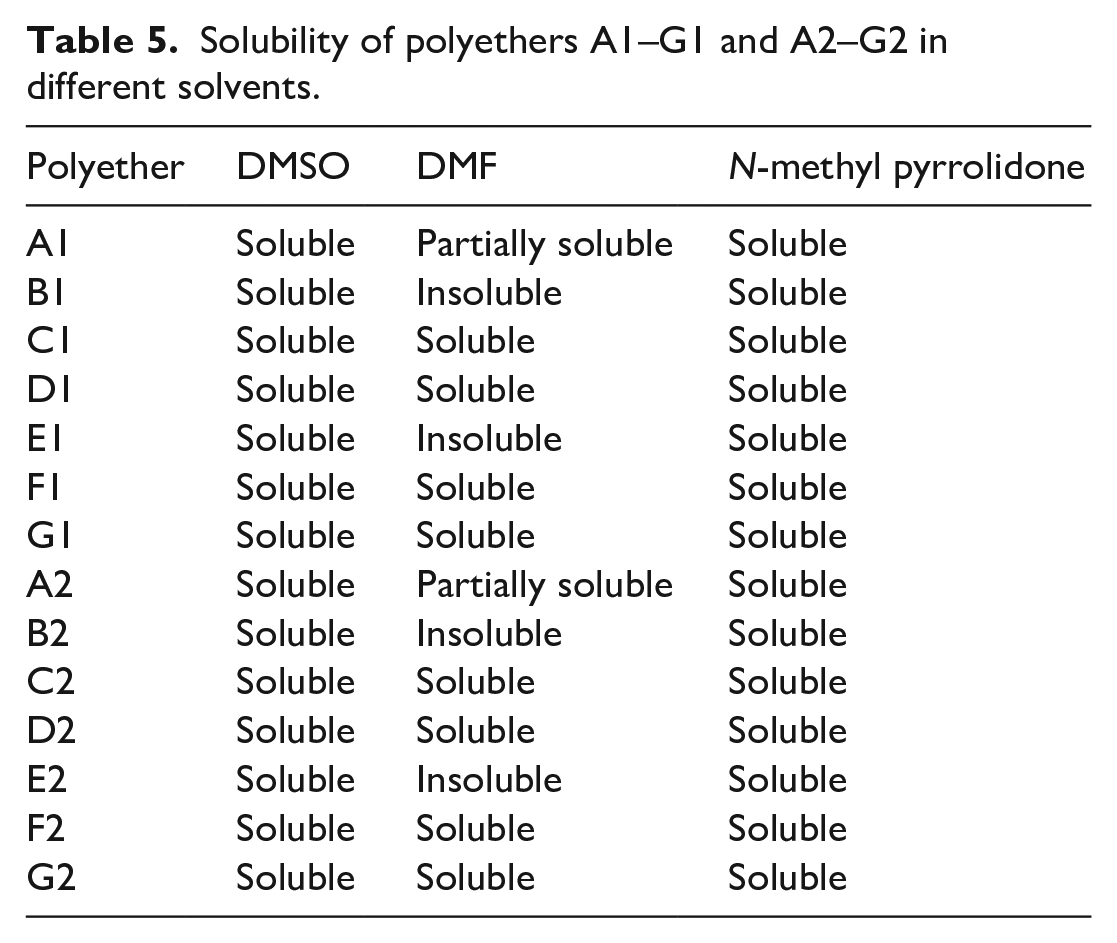
The solubility of polyethers A1–G1 and A2–G2 was tested in the most common solvents. The polyether (50 mg) was left in an appropriate solvent (3 mL) for 24 h and stirred occasionally, and the results are summarized in Table 5. The polyethers were soluble in the two aprotic solvents (DMSO and N-methyl pyrrolidone). However, their solubility in DMF (aprotic solvent) varied between complete solubility and partial solubility in some polyethers. Polyethers A1–G1 and A2–G2 were insoluble in acetone (aprotic solvent), ethanol, and methanol (protic solvents). Similar observations have been seen in other solvents, such as toluene, chloroform, tetrahydrofuran, and dichloromethane.
Solubility of polyethers A1–G1 and A2–G2 in different solvents.
Photophysical properties of polyethers A1–G1 and A2–G2
The UV-Vis absorption and fluorescence spectra of polyethers A1–G1 and A2–G2 were recorded in diluted DMSO (1 × 10−4 M) (Figures 1–7). Table 6 summarizes the absorbance and fluorescence spectral data and energy gap obtained from the curves. The absorption spectra of polyethers A1–G1 and A2–G2 exhibit a low-energy (high wavelength) absorption peak due to the π–π* electronic transition of the C=C bonds. The higher number of successive double bonds in the polymer leads to an increased wavelength. An increase in the chromophore length is observed in polymers containing OH and OMe groups. The absorption peaks observed at high energy were due to the n–π* electronic transition in the two-electron lone pairs on the oxygen atom, which interacts electronically with successive double bonds.
Absorption and fluorescence data of polyethers of polyethers A1–G1 and A2–G2.


Fluorescence of polyethers A1 and A2.
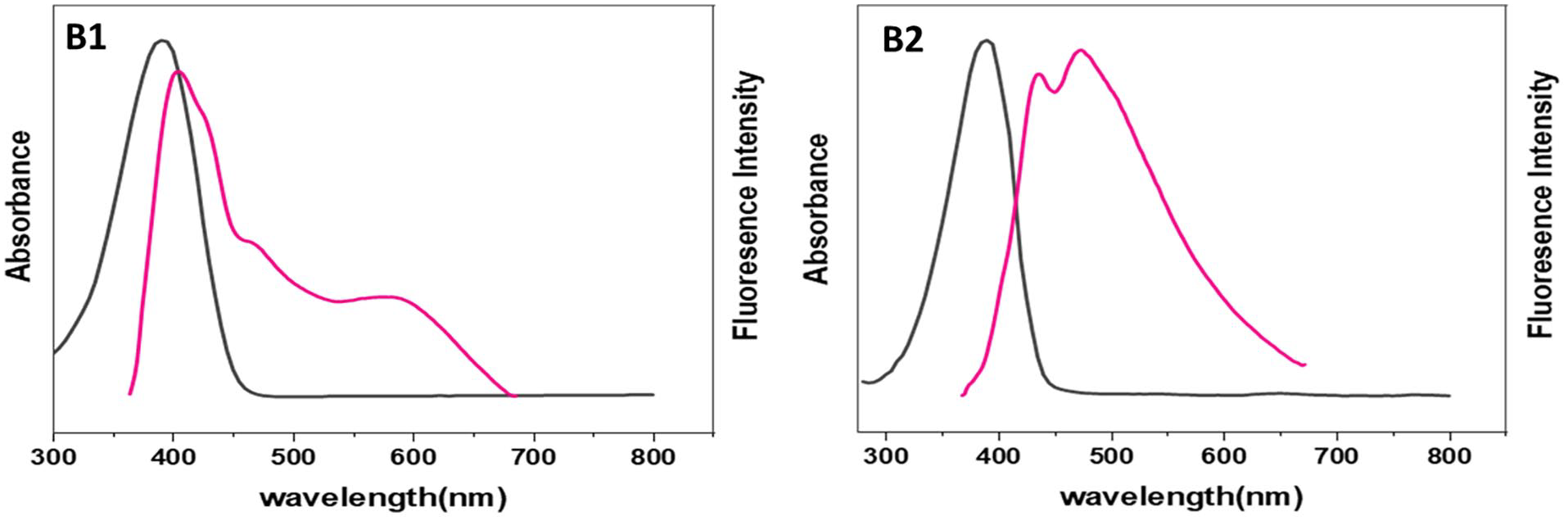
Fluorescence of polyethers B1 and B2.
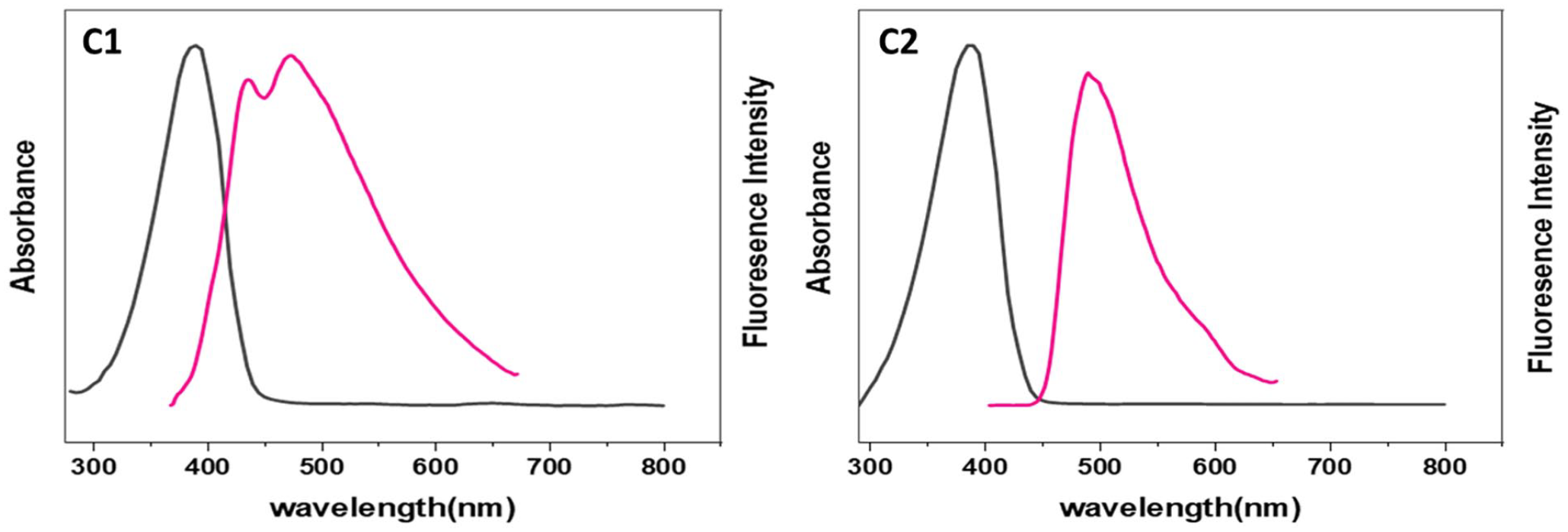
Fluorescence of polyethers C1 and C2.

Fluorescence of polyethers D1 and D2.

Fluorescence of polyethers E1 and E2.

Fluorescence of polyethers F1 and F2.

Fluorescence of polyethers G1 and G2.
The intensity of fluorescence of polyethers varies due to several factors. For instance, the presence of electron-propelling aggregates like OMe and OH causes a shift toward higher wavelengths, resulting in green emission and increased fluorination intensity. In addition, the fluorescence intensity increases with the pH level of the solution. The type of solvent used also affects the fluorescence intensity, with highly polar solvents resulting in a shift toward higher wavelengths and expanded emission due to an increase in the number of hydrogen bonds. The intensity of fluorescence can be reduced due to various factors, such as ultraviolet radiation used to excite the sample, which can cause breakdown or disintegration of the fluorescence compound. In addition, intramolecular or intermolecular reactions, including excited state reactions and energy dissipation, can also lead to a decrease in fluorescence intensity.
The diverse structures of the synthesized polyethers are due to condensation reactions between various monomers. This structural variation can affect the sensitivity of the polyether to different irradiation intensities, which can be reflected in the resulting spectra. The differences in fluorescence and absorption spectra among the polyethers may arise from several factors. For example, increasing the molecular weight of polyethers leads to higher viscosity. High molecular weight polymers have an increased chain length that restricts their movement. The rate of collisions between large polymer molecules decreases, reducing energy dissipation and increasing fluorescence intensity. Polymer chains’ elasticity is another factor caused by differences in composition. Ether bonds increase the chains’ flexibility compared to other bonds, such as ester and imide bonds. The mobility of the polymer chains is also affected by the length of the ether bond. Polyethers A1–E1 contain butylene ether bonds, while polymers A2–E2 have methylene ones. As the number of carbon atoms increases, the flexibility of polymer chains also increases.
The presence of aromatic rings can increase the intensity of the fluorescence spectrum due to their improved resonance structures. Therefore, aromatic moieties in polyethers are more suitable for fluorination compared to polyethers containing methylene aggregates (known as aromatic polyethers-aliphatic). Polymers with a high degree of sequence are less soluble in most organic solvents, but the presence of aliphatic spacers can improve their solubility. The presence of terminal end groups affects the fluorescence spectrum. When these monomers undergo polymerization, the resulting polymers contain these groups at the ends of the chains. Therefore, it may cause a shift to a higher or lower wavelength, depending on their ratio and the nature of their effect (e.g. the formation of hydrogen bonds or their effect on absorption intensity).23,24 The synthesized polyethers showed fluorescence in the DMSO solution, but the intensity and adsorption varied depending on the nature of the polymers.
Conclusion
Several polyethers derived from curcumin analogs were synthesized through condensation polymerization of curcumin analogs and dibromoalkanes using simple procedures. Their chemical structures were confirmed using spectroscopy. The stability of the synthesized polyethers was assessed, and the thermal gravimetric analysis indicated that polyethers displayed stability, holding up well between 250 and 300 °C. Synthesized polyethers are useful thermoplastics and have the potential to be used in a variety of applications. Moreover, the synthesized polyethers exhibited fluorescence properties in solutions. The intensity and absorption of polyethers varied depending on the properties of each polymer.
Materials and methods
General
Solvents, chemicals, and reagents were purchased from Merck (Darmstadt, Germany), Bakr (NJ, USA), and HiMedia (PA, USA). They have been used as received without further purification. The FTIR spectra (400–4000 cm−1) were recorded on a Bruker Alpha II spectrometer (Tokyo, Japan). The 1H NMR spectra (400 MHz; δ in ppm and J in Hz) were recorded in dimethyl sulfoxide (DMSO-d6) using a Bruker Avance spectrometer (Tokyo, Japan). The absorption spectra (200–800 nm) were carried out on a Shimadzu UV-1800 spectrophotometer (Tokyo, Japan). The fluorescence spectra were performed using a FluoTime 300 spectrophotometer (PicoQuant, Berlin, Germany). Thermogravimetric data were recorded on a Shimadzu TGA-50 thermogravimetric analyzer (Tokyo, Japan) using a nitrogen flow of 30 mL/min and a heating (30–600 °C) rate of 20 °C/min.
Synthesis of curcumin analogs
Curcumin analogs A–G were synthesized based on previously reported synthetic procedures (Scheme 1).13,14 Glacial acetic acid and anhydrous hydrogen chloride (10:1; 11 mL) were added to a stirred mixture of appropriate aldehyde (10 mmol), ketone (cyclohexanone, acetone, or piperidine-4-one; 5 mmol) in dry ethanol (EtOH; 5 mL). The mixture was stirred for 2 h and then left for 48 h (11 days for D and G) at room temperature. Cold distilled water was added, and the solid was collected by filtration, washed, dried, and recrystallized methanol (MeOH) to give curcumins A–G (Scheme 1) in good yields (Table 1). The FTIR and 1H NMR of the synthesized curcumin analogs A–G were identical to those reported.13,14
Synthesis of polyethers A1–G1
A mixture of appropriate chalcone (A, C, D, F, or G), 1,4-dibromobutane (0.86 g, 4 mmol), and potassium carbonate (K2CO3; 16 mmol) in DMF (30 mL) for A1, C1, D1, F1, and G1 or DMSO (30 mL) for B1 and E1 was stirred for 24 h at 60 °C in which a dark red color was observed. 15 The mixture was left to cool to room temperature, and the polymer was precipitated through the addition of MeOH (50 mL). The polymer was filtered, washed with MeOH (3 × 20 mL) and water (3 × 20 mL), and dried in air. Crystallization using aqueous MeOH gave polyethers A1–G1 (Scheme 2) in good yields (Table 2).
Synthesis of polyethers A2–G2
The synthetic procedure for polyethers A2–G2 (Scheme 3) was similar to that used for the production of A1–G1 (Scheme 2). DMF was used for the synthesis of A2, C2, D2, F2, and G2, and DMSO was used to produce B2 and E2. Table 2 summarizes the color and yield of A2–G2.
Supplemental Material
sj-docx-1-chl-10.1177_17475198231221458 – Supplemental material for Synthesis, characterization, and fluorescence properties of new polyethers derived from curcumin analogs
Supplemental material, sj-docx-1-chl-10.1177_17475198231221458 for Synthesis, characterization, and fluorescence properties of new polyethers derived from curcumin analogs by Walaa H Ali, Mohammed A Bahili, Basil A Saleh and Gamal A El-Hiti in Journal of Chemical Research
Footnotes
Author contributions
W.H.A., M.A.B., and B.A.S. contributed to conceptualization. W.H.A., M.A.B., and B.A.S. contributed to methodology. W.H.A., M.A.B., B.A.S., and G.A.E.-H. contributed to formal analysis. W.H.A., M.A.B., and B.A.S. contributed to investigation. W.H.A., M.A.B., B.A.S., and G.A.E.-H. contributed to data curation. W.H.A., M.A.B., B.A.S., and G.A.E.-H. contributed to writing. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank the University of Basrah for the technical support. G.A.E.-H. acknowledges the support from the Researchers Supporting Project (no. RSP2023R404), King Saud University, Riyadh, Saudi Arabia.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
