Abstract
A series of symmetric, silicon-linked organic fluorescent compounds with two electron-deficient 1,3,4-oxadiazole units were synthesized and characterized. The compounds possessed a V-shaped structure with a silicon atom, which weakened π–π stacking, promoting aggregation-induced emission. The compounds were fluorescent in both solution and solid-state thin films. The efficient fluorescent behavior of the materials was confirmed through optical and electrochemical measurements. The compounds displayed excellent thermal stability, with decomposition temperatures exceeding 400 °C. Amorphous films of the compounds possessed high morphological stability. These results indicate that the compounds may be promising emissive and electron-transporting materials.
Keywords

Introduction
Organic fluorescent materials have been the subject of great interest because of their potential applications in many aspects of modern life, such as lighting, displays, bioprobes, and chemosensors.1–5 Generally, the luminescence properties of organic molecules in the solid state are determined mainly by their chemical structures. Most fluorescent organic molecules with highly conjugated planar structures emit reasonably intense fluorescence in dilute solutions but emit weak or nonexistent emission in the solid state because of aggregation-induced quenching.6–9 However, some groups have recently reported the unique phenomenon of aggregation-induced emission (AIE).10,11 Propeller-shaped molecules such as tetraphenylethene, hexaphenylsilole, and quinoline–malononitrile, along with their derivatives, are known to display AIE.12–17 Molecular design optimization can be used to increase molecular luminous efficiency and stability, leading to organic light-emitting diodes (OLEDs) with improved optoelectronic properties.18–21
Considering all organic luminescent materials, silicon (Si)-based compounds have been reported to perform well in optical applications because of their high brightness, thermal stability, and amorphous film-forming capability.22–24 Si-based tetrahedral organic molecules and polymers have been intensively investigated as electroluminescent materials. For example, Partee et al. 25 and Zhen et al. 26 recently reported a series of tetrahedral luminescent materials with SiAr4 cores. Meanwhile, 1,3,4-oxadiazoles are classic heterocyclic compounds that have also attracted substantial interest for use in OLEDs because of their favorable electronic transmission properties. Many kinds of oxadiazole-based materials have been synthesized and used in different applications, and some are considered to be potential blue emitters for OLEDs.27,28
Results and discussion
Continuing our interest in V-shaped luminescent materials,29,30 in this article, we report the development of a series of V-shaped fluorescent molecules (
Starting compounds
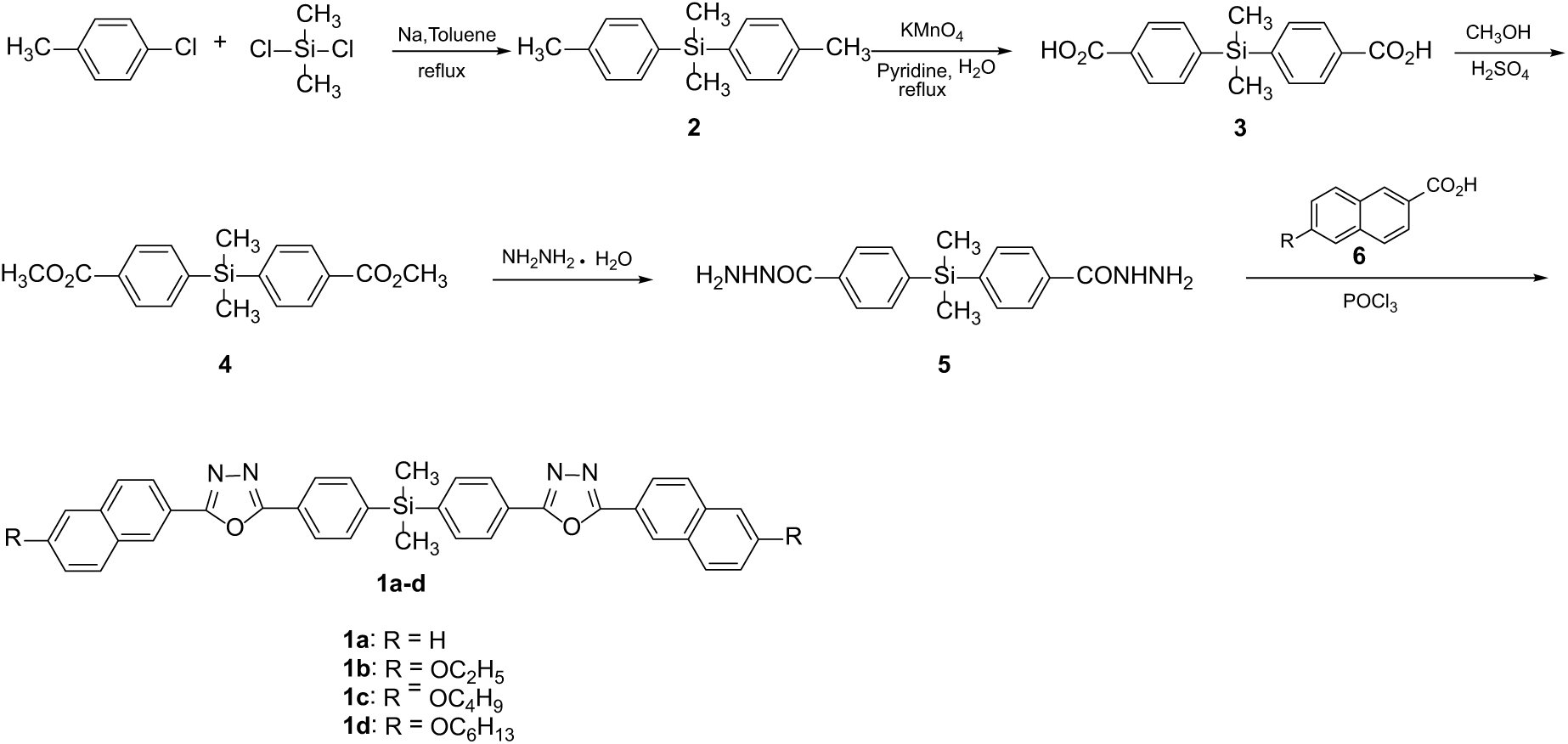
Synthetic route of
The ultraviolet (UV)-Vis absorption properties of compounds
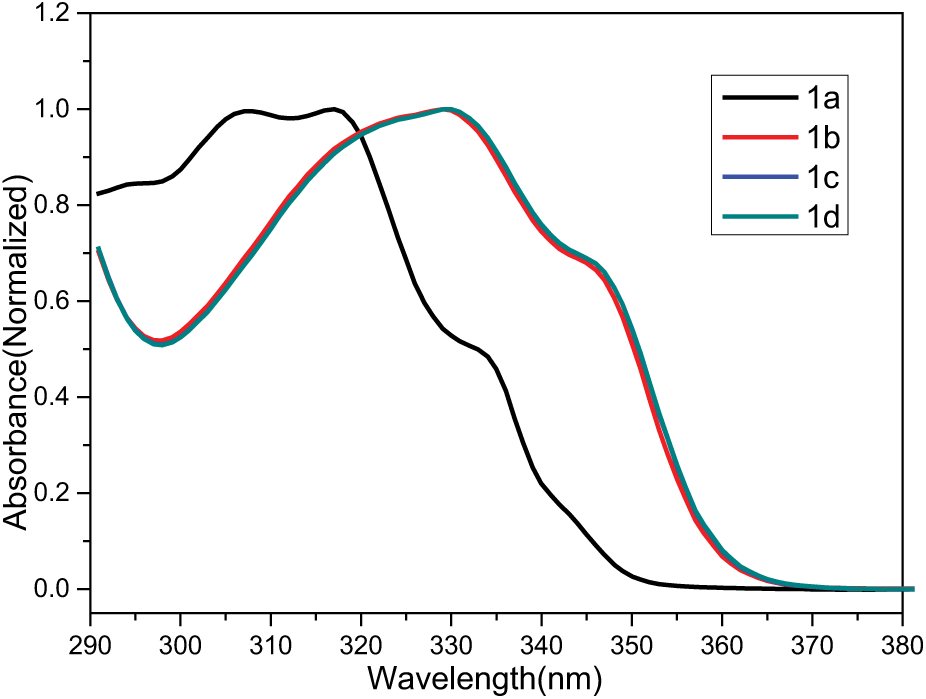
UV-Vis absorption spectra of compounds
The photoluminescence (PL) spectra of compounds

Normalized PL spectra of compounds
The electrochemical properties of

CV of
Photophysical, physical, and electrochemical data.

ΦF is the fluorescence quantum yield, which was measured in CHCl3 using quinine sulfate as a standard (ΦF = 0.55);
34
Measured in CHCl3.
Calculations of

Contour plots of the HOMO and LUMO for complexes
The thermal stability of a material is important for device longevity. Figure 5 and Table 1 show the results of thermogravimetric analysis (TGA) of compounds

Thermal gravimetric analysis and differential scanning calorimetry thermograms of
In summary, we developed a facile and effective method to synthesize V-shaped organic fluorescent compounds. These synthesized compounds do not aggregate in the condensed state as shown by their similar properties either in solution or in film states. These emitters are solution processable and exhibit good film-forming ability as well as high thermal stability, making them attractive for use in OLEDs.
Experimental
NMR spectra were recorded in CDCl3 with a Bruker AV-400 spectrometer. Chemical shifts were referenced relative to tetramethylsilane (δH/δC = 0). Infrared (IR) spectra were recorded on a Perkin Elmer 2400 spectrophotometer (using KBr pressed disks). Mass spectra were collected on an Autoflex Speed™ matrix-assisted laser desorption/ionization time-of-flight (MALDI-TOF) spectrometer. Elemental analyses were performed on an Elementar Vario EL CHN elemental analyzer. UV-Vis spectra were recorded on a Lambda 25 spectrophotometer in CHCl3. Fluorescence spectra were obtained from a Shimadzu RF-5301PC fluorescence spectrophotometer in CHCl3. CV was conducted using a CHI 852C instrument with 10−3 M solutions using CH2Cl2 as the solvent (10−3) and containing 0.1 M Bu4NPF6 as the supporting electrolyte. Counter and working electrodes consisted of a Pt wire and a Pt disk, respectively, and the reference electrode was Ag/AgCl. The thermal stability of the target compounds was characterized using a Diamond TG/DTA thermogravimetric analyzer. Starting compounds
General procedure for the preparation of compounds 1a–d
A mixture of the appropriate 2-naphthoic acid
Dimethylbis{4-[5-(naphthalen-2-yl)-1,3,4-oxadiazol-2-yl]phenyl}silane (
Bis{4-[5-(6-ethoxynaphthalen-2-yl)-1,3,4-oxadiazol-2-yl]phenyl}dimethylsilane (
Bis{4-[5-(6-butoxynaphthalen-2-yl)-1,3,4-oxadiazol-2-yl]phenyl}dimethylsilane (
Bis{4-[5-(6-hexyloxynaphthalen-2-yl)-1,3,4-oxadiazol-2-yl]phenyl}dimethylsilane (
The 1H and 13C NMR spectra, TOF-MS data, cyclic voltammograms, and TGA data for
Supplemental Material
Supplementary_info – Supplemental material for Design, synthesis, and photoelectric properties of V-shaped organic fluorescent compounds with a 1,3,4-oxadiazole moiety
Supplemental material, Supplementary_info for Design, synthesis, and photoelectric properties of V-shaped organic fluorescent compounds with a 1,3,4-oxadiazole moiety by Rui-Bin Hou, Ji-Ying Su, Ling-Ling Zhang, Dong-Feng Li and Yan Xia in Journal of Chemical Research
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the National Science Foundation of China (No. 21442004 and 21502008) and the Education Office of Jilin province (No. 2016320).
Supplemental material
The supplemental material with 1H and 13C NMR spectra, TOF-MS data, cyclic voltammograms, and TGA data for
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
