Abstract
The tannic acid–based iron nanoparticle (TAN-Fe-NP) was prepared by a redox reaction between tannic acid and FeCl3 in water and characterized by the Fourier transform infrared spectroscopy, ultraviolet–visible spectroscopy, energy dispersive X-ray analysis, X-ray diffraction, scanning electron microscopy, and transmission electron microscopy techniques. Then, a TAN-Fe-NP nanocatalyst was used as an efficient catalyst for the synthesis of polyhydroquinolines from the reaction of ethyl acetoacetate, benzaldehydes, dimedone, and ammonium acetate in ethanol at 60 °C in high yields and low reaction times. Since this catalyst is made of natural materials, it has a great advantage compared with the other catalysts and is cheap, available, natural, safe, nontoxic, and eco-friendly as well.

Introduction
There are several physical (pyrolysis, destruction), chemical (electrochemical, photochemical, reduction, oxidation), and biological (plant extracts, fungi, algae, microbes, yeasts and) methods for the preparation of different nanoparticles. For example, Eucalyptus, 1 Oolong tea, 2 watermelon, 3 Amaranthus dubius, 4 Lawsonia inermis, 5 Tridax procumbens, 6 and Punica granatum 7 were used as plant extracts for the synthesis of iron nanoparticles. On the other hand, it should be noted that the conventional nanoparticle synthetic methods like attrition and pyrolysis have drawbacks, such as defective surface formation, low production rate, high cost of manufacturing, and large energy requirement. 8 Chemical synthesis methods (e.g. chemical reduction, sol–gel technique) involve the usage of toxic chemicals, formation of hazardous byproducts, and contamination from precursor chemicals. 9 Hence, there is a growing need to develop clean, nontoxic, and environment-friendly procedures for nanoparticle synthesis. It has been well documented that the optical, electronic, and catalytic properties of metal nanoparticles are functions of nanoparticle size shape and crystal structure. For instance, differently shaped nanostructures of silver and gold embody unique optical scattering properties.10–13
Many recent studies have indicated the potential of Fe-NPs for environmental remediation. Nanoscale materials such as nanoadsorbents, nanocatalysts, nanofiltration, and nanobiocides, such as metal and metal oxide nanoparticles, are currently being employed for the remediation of water and wastewater pollutants. Among these metallic nanoparticles, Fe-NPs have promising advantages that can combat environmental pollution. The interest in nanoscale zero-valent iron in environmental remediation is increasing due to the reactivity of nanoscale iron having a large surface area to volume ratio. 14 The production of iron nanomaterials, such as metallic iron and oxide of iron via a more convenient greener route, is a great step forward in the development of nanomaterials.
The genus Pistacia (family of Anacardiaceae) includes over 600 species. Pistacia vera, Pistacia atlantica, Pistacia terebinthus, Pistacia khinjuk, and Pistacia lentiscus are the most famous species of Pistacia that are widely distributed in the Mediterranean and middle eastern areas.15,16 P. atlantica is a tree with a length of 2–5 m. The branches of the tree are grayish-white and have leaves composed of 9–11 leaflets. 17
Important phytochemicals of P. atlantica are mainly terpenoids, 18 phenolic compounds,19,20 fatty acids, and sterols. 21 The P. atlantica Desf. has diverse pharmacological properties, such as antimicrobial, 22 antihyperlipidemic, 23 antioxidant hypoglycemic,24,25 anticholine sterase, 26 wound-healing effects and gastrointestinal benefits, 27 and anticancer 28 activities, in which most of the mentioned properties are probably due to the presence of phenolic structure. 29
The mechanism behind plant extract mediated metallic nanoparticle formation has not been clearly defined up until now. Not a single biomolecule of plant extract was involved in the fabrication of nanoparticles. Various plant components are rich in secondary metabolites and responsible for synthesis of metallic nanoparticles. Secondary metabolites include the poly-phenols, flavonoids, tannic acid, terpenoids, ascorbic acids, carboxylic acids, aldehydes, and amides. Many reducing sugars are commonly found in plants, and their presence is confirmed by the IR spectroscopic technique in different studies. 30
The plant extracts possess ideal redox properties that allow efficient reduction of metal precursors for conversion into their corresponding metallic nanoparticles. In another study, 31 the researchers utilized the tannin of alfalfa.
Tannic acid and tannins are polyphenolic compounds able to build very complex chemical structures. The tannic acid, which has an estimated chemical formula of C76H52O46 is a commercial form of tannins. Researchers utilized tannin powder a green reagent for the synthesis of Fe-NPs. Tannins consist of nontoxic polyphenolic compounds that act as reducing and stabilizing agents to produce Fe-NPs. 32 These facts show the properties of tannic acid, which is a reducing and stabilizing compound in alkaline conditions at room temperature that is related to the numerous phenolic groups in its structure. These phenols take part in redox reactions by forming quinones donating electrons. Due to their tanning capacity, these compounds were very important in tannin industries, though the synthetic tannins have acquired more importance than tannins extracted from natural sources. In the last two decades, the isolation and structural determination of many types of tannins that also include hydrolysable and condensable tannins have been possible using techniques such as UV-Vis, 1H NMR, 13C NMR, and mass.33,34
Polyhydroquinolines (PHQs) are compounds with the 1,4-dihydropyridine core which are an important structure in the synthesis of pharmaceutical materials. Nowadays, because of the wide range of biological activity of 1,4-dihydropyridines, much attention is paid to the synthesis of these compounds that have various biological properties such as antioxidant, anticonvulsant, antidepressant, antitumor, calcium channel opener, liver protector, and pain killer. 35
They are also used for treatment of Alzheimer’s disease and cancer. 36 Today, several quinoline and quinolone antibacterial compounds are widely used for the treatment of various infections. Also, quinoline and quinolones with specific functional groups have been applied for the synthesis of pharmaceuticals and agrochemicals reagents in the preparation of new alkaloids.37,38
Due to diverse biological and medicinal properties of PHQs, several methods have been reported for their synthesis, such as application of ionic liquids, 39 p-toluene-sulfonic acid, 40 Fe3O4 NPs, 41 cerium ammonium nitrate (CAN), 42 FeF3, 43 tetrabutylammonium hexatungstate, 44 poly(4-vinylpyridinium) hydrogen sulfate, 45 alginic acid, 46 nicotinic acid, 47 copper nanoparticles, 48 Ni complex, 49 1,4-diazabicyclo[2.2. 2]octane (DABCO), 50 MoO3 promoted CeO2–ZrO2, 51 and cobalt complex 52 in solvent-free conditions or ultrasonication. However, many of these methods have disadvantages, such as long reaction time, low yields, use of toxic substances, high temperature, expensive catalysts, environmental pollutants, difficult work-up, and separation. For this reason, we wish to report a natural, nontoxic, eco-friendly extracted tannic acid-based iron as a nanocatalyst (TAN-Fe-NP) for the synthesis of PHQs in short reaction times and excellent yields. The P. atlantica Desf. tree bark extract was chosen because of its benign nature as well as the presence of diverse functional groups that allow the Fe-NPs to be stable even after 30 days (Scheme 1).

Synthesis of PHQs by the TAN-Fe-NP catalyst.
As far as we know, there is no report on the synthesis of Fe-NPs using tannic acid.
Results and discussions
Tannic acid was characterized by the following methods
Characterization by Fourier transform infrared spectroscopy
Figure 1 shows the Fourier transform infrared (FTIR) spectrum of the tannic acid extracted from the P. atlantica Desf. tree bark. A peak at about 2923 cm−1 shows the C–H bond and a peak at about 3426 cm−1 shows the symmetric OH stretching vibrations. Also, there is no signals of free water molecules since there is no evidence of bands at 2330 and 1625 cm−1 (H–O–H bending vibrations and at about 1070–625 cm−1). In addition, at about 1730–1625 and 1070–1300 cm−1, there are two characteristic bonds that are related to the C=O and the C–O stretching vibration modes. 53

The FTIR spectrum of tannic acid extracted from the Pistacia atlantica Desf. tree bark.
Characterization by NMR
Figure 2 shows the 1H NMR spectrum of the tannic acid extracted from the P. atlantica Desf. tree bark. The corresponding explanation is as follows: 1H NMR (CDCl3, 300 MHz): δ = 0.9 (m, 9H), 1.2–1.4 (m, 60H), (1.5–1.7 (m, 6H), 2.3 (m, 6H), 4.2 (dd, 2H), 4.3 (dd, 2H), and 5.3 (m, 1H) ppm; 13C NMR (CDCl3, 75 MHz): δ = 14.08 (C-14, C-28), 22.66 (C-13, C-27), 24.85, 24.89 (C-3, C-17), 29.06–31.90 (C-4–C-12, C-18–C-26), 34.04, 34.20 (C-2, C-16), 62.08 (C-30), 68.85 (C-29), 172.85 (C-15), and 173.26 (C-1) ppm.

The 1H NMR spectra of tannic acid extracted from the Pistacia atlantica Desf. tree bark.
Characterization of the TAN-Fe-NP catalyst
The TAN-Fe-NP catalyst was characterized by the following procedures.
Characterization by FTIR
Figure 3 shows the FTIR spectrum of the TAN-Fe-NP catalyst. The peaks at 3472, 1732, 1636, and 1071 cm−1 are related to the O–H, C=O, C=C, and C–O bonds, respectively.

The FTIR spectrum of the TAN-Fe-NP catalyst.
Characterization by UV-Vis
Figure 4 shows the UV-Vis spectrum of the TAN-Fe-NP catalyst. A long and sharp absorption at 229 nm indicates the stability of Fe-NPs and conversion of Fe3+ to Fe(0) leading to higher Fe-NPs concentrations. 54

The UV-Vis absorption spectrum of the TAN-Fe-NP catalyst.
Characterization by the scanning electron microscopy images
Figure 5 shows the scanning electron microscopy (SEM) images of the Fe-NPs, which are well separated in the range of nanometers (29–45 nm) and their structures are almost spherical and inhomogeneous. The results obtained from SEM showed how the nanostructures of the synthesized particles functioned. The obtained results are in line with the previous research. 55

The SEM images of the TAN-Fe-NP catalyst.
Characterization by the energy dispersive X-ray analysis
Figure 6 shows the energy dispersive X-ray (EDX) analysis of the TAN-Fe-NP catalyst indicating the presence of the anticipated elements. 56

The EDX analysis of the TAN-Fe-NP catalyst.
It should be noted that carbon comes from the plant extract components existing in the surface of NPs (also shown in FTIR data) and Cl is usually observed as impurity during the Fe3O4 NPs synthesis from ferric chloride as observed and reported by others.53,54 The relative percentage of elements are as below: Fe (12.08%), carbon (36.46%), oxygen (22.20%), B (15.40%), S (3.98%), Cl (2.36%), and N (7.5%).
Characterization by the X-ray diffraction analysis
The X-ray diffraction (XRD) analysis was used with an angle ranging from 20 to 80º to determine the particle size of Fe-NPs and its unique nature (Figure 7). The XRD showed the crystalline morphology of Fe-NPs and the average crystal size. The three peaks in the diffraction pattern at angles of 30.24, 35.32, 43.03, 56.95, and 62.98 are related to the Joint Committee on Powder Diffraction Standards (JCPDS) standard Fe numbered 89-2838. 55 The four notable peaks appeared, which correspond to (200), (311), (440), and (511) reflections of the face-centered cubic (fcc) phase of Fe-NPs.

The XRD pattern of the TAN-Fe-NP catalyst.
Characterization by the transmission electron microscopy images
The transmission electron microscopy (TEM) characterization tool relies on the interaction of a narrow iron oxide NPs specimen and current density electron beam.57,58 So, the TEM images were recorded to determine the shapes and sizes of the TAN-Fe-NP catalyst (Figure 8), which was found to be in the range of 30–51 nm.

The TEM images of the TAN-Fe-NP catalyst.
Proposed mechanism of the tannic acid–based reduction of metal salt
The phenolic forms in tannic acid were converted (oxidized) to quinone forms by the electron loss, 59 then, the electrons in the reaction medium can reduce the Fe3+ ions to Fe(0) (metallic iron) (Figure 9).
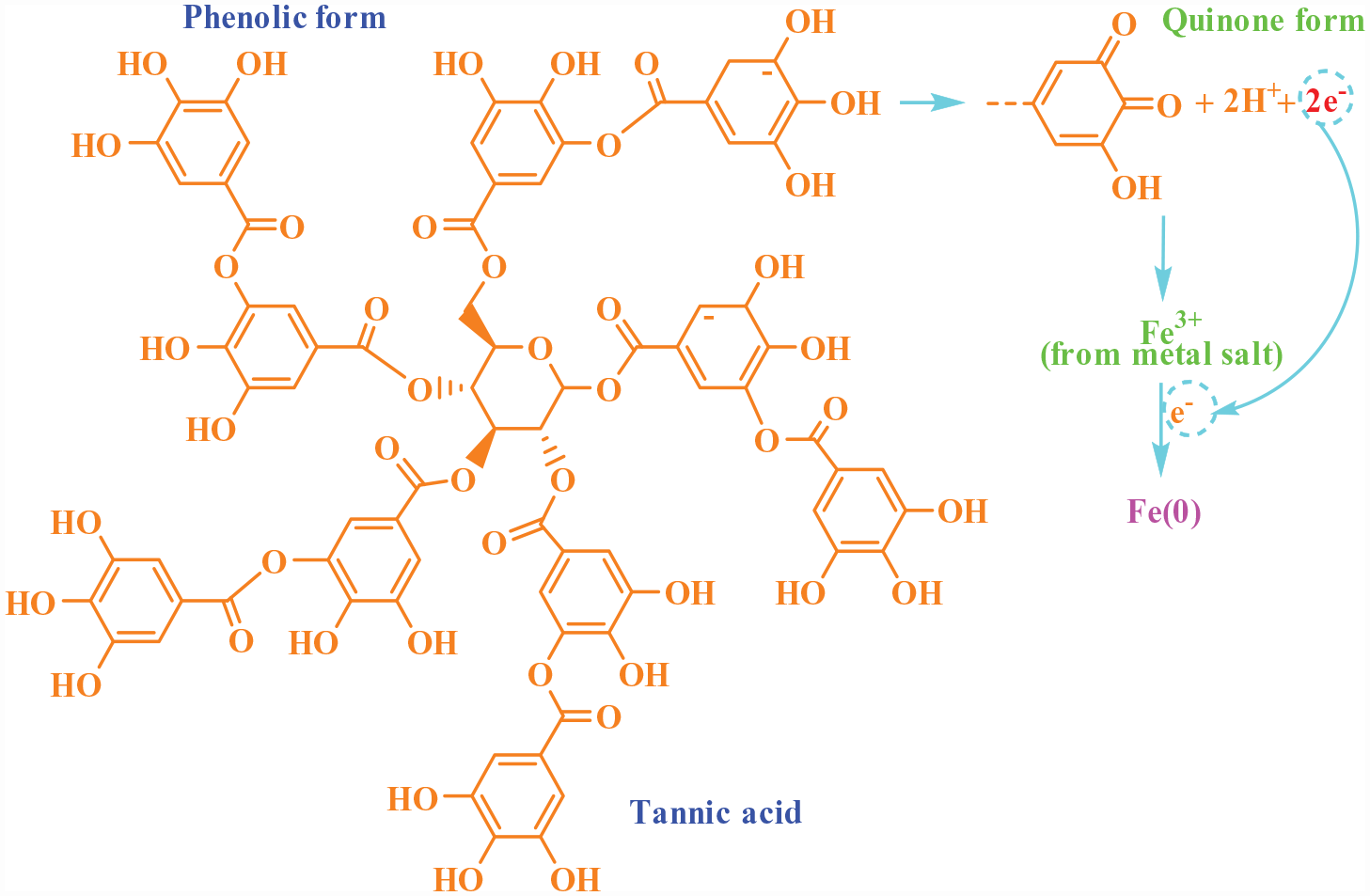
Mechanism of the metal salt reduction by tannic acid in water.
Optimization of the reaction conditions
To optimize the reaction conditions, a reaction of benzaldehyde, dimedone, ethyl acetoacetate, ammonium acetate, and the TAN-Fe-NP catalyst was carried out for the synthesis of PHQs in different temperatures, amounts of the catalyst, and solvents.
Optimization of solvent
The reaction of benzaldehyde, dimedone, ethyl acetoacetate, ammonium acetate, and the TAN-Fe-NP catalyst was carried out for the synthesis of PHQs in different solvents with 20 mg of the catalyst at 60 °C. After completion of the reaction, EtOH was selected as a best solvent (Table 1).
Effect of solvent on the synthesis of PHQs.

Optimization of temperature
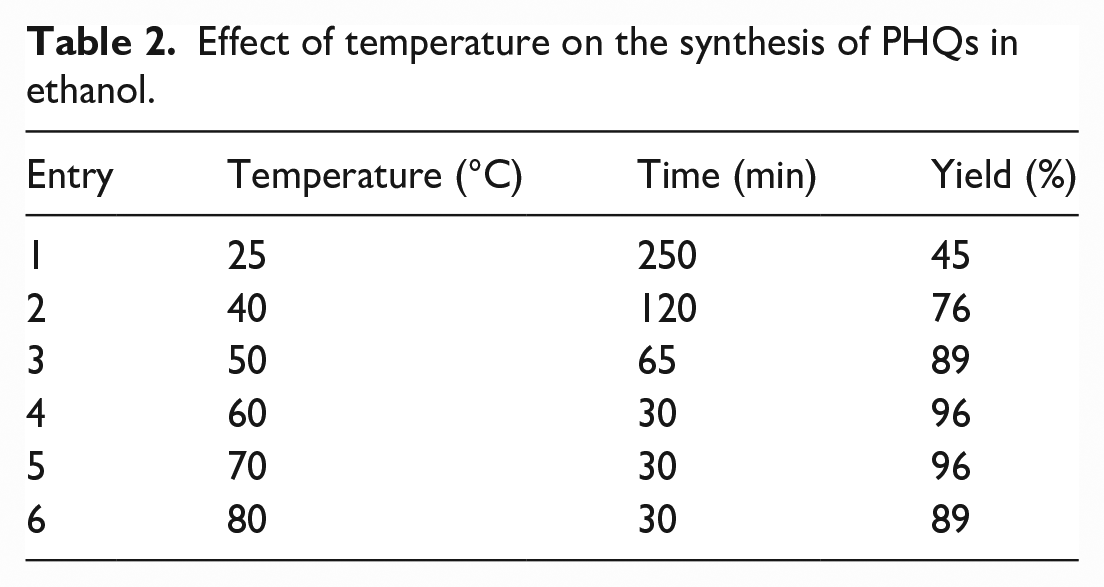
The reaction of benzaldehyde, dimedone, ethyl acetoacetate, ammonium acetate, and the TAN-Fe-NP catalyst was carried out for the synthesis of PHQs in different temperatures with the same amount of the catalyst (20 mg) in EtOH. After completion of the reaction, a temperature of 60 °C was selected as an optimum temperature (Table 2).
Effect of temperature on the synthesis of PHQs in ethanol.
Optimization of the catalyst amount
The reaction of benzaldehyde, dimedone, ethyl acetoacetate, ammonium acetate, and the TAN-Fe-NP catalyst was carried out for the synthesis of PHQs in different amounts of the catalyst at 60 °C in EtOH. After completion of the reaction, a 20 mg of the catalyst was selected as an optimum amount (Table 3).
Effect of the catalyst amount on the synthesis of PHQs in EtOH at 60 °C.

So, the best condition was found to be 1:1:1:1 mmol of benzaldehyde, dimedone, ethyl acetoacetate, and ammonium acetate with 20 mg of the TAN-Fe-NP catalyst in EtOH at 60 °C (model reaction).
Synthesis of PHQs
According to the results obtained from the model reaction, synthesis of diverse PHQs was carried out from the reaction of benzaldehyde (1.0 mmol), dimedone (1.0 mmol), ethyl acetoacetate (1.0 mmol), ammonium acetate (1.0 mmol), and the TAN-Fe-NP catalyst (20 mg) in EtOH at 60 °C in appropriate times (Table 4 and Figure 10).
Synthesis of PHQs 2(a-k).


2(2-k).
Recyclability of the TAN-Fe-NP catalyst
To investigate the recyclability of the TAN-Fe-NP catalyst, it was filtered off at the end of the reaction (model reaction), washed with water, dried overnight, and used again in three consecutive runs that showed no significant loss of activity (96%, 95%, 93%, and 91%, respectively) (Figure 11).

Recyclability of the catalyst in four consecutive runs.
Comparison of the catalyst activity
Table 5 shows comparison of our catalyst (the TAN-Fe-NP catalyst) with other catalysts applied for the synthesis of PHQs reported in literatures. It can be seen that the results of our catalyst are better in terms of efficiency and reaction time compared with the other methods. Also, it is important to say that the PHQs were synthesized in green condition, which is the most important advantage of our method.
Comparison of the PHQs synthesis.

Conclusion
A green synthetic protocol was applied to prepare the stable Fe-NPs without the use of any toxic materials by application of P. atlantica as a reducing and stabilizing agent. Then, the TAN-Fe-NP was characterized by different methods and used as a recyclable catalyst for the synthesis of PHQs in high yield, low reaction time and easy separation.
Experimental
The bark of P. atlantica tree was gathered from the Zagros Mountain, the western part of Iran and FeCl3.6H2O purchased from Merck. UV-Vis spectra were recorded on the Phystec-Miniature UVS-2500 spectrophotometer and employed with one nm resolution in the range of 190–1100 nm. An Avatar Thermo Spectrophotometer instrument was used to record FTIR spectra. Analyses of the particle size and surface morphologic features were performed by the SEM images recorded in SEM FEI Quanta 200 SEM Tescan Mira3. The TEM images were recorded using the Philips EM208S 100 kV to determine the shapes and sizes of the TAN-Fe-NP catalyst. The XRD spectra were analyzed by a Philips PW 1730 instrument. An advanced diffractometer was used with Fe (Kα) radiation (wavelength: 1.5406 Å) at 40 kV and 40 mA at ambient temperature in a 2θ at a range of 20–80°.
Preparation of Pistacia tree barks
The fresh P. atlantica Desf. tree bark were collected from the Ilam province in west of Iran, washed several times with water, chopped, and spread out in a shady place for about 10 days to be dried.
Extraction of tannic acid from the P. atlantica Desf. tree bark
The dry P. atlantica Desf. tree bark was placed in 70% acetone at room temperature for 1 week. Then, the mixture filtered to separate the barks pieces and the solvent removed to get the yellowish-brown solid. Afterward, diethyl ether was added to the obtained solid to dissolve any non-tannic compound and the mixture filtered to remove the solvent and getting acid tannic as a brown product. Also, the presence of tannic acid can be confirmed by addition of FeCl3 and appearance of the blue color (five drops of the tannic acid extract were mixed with two drops of 10% FeCl3 and the content carefully shook in which the appearance of the blue color confirmed the presence of tannic acid). Moreover, the tannic acid structure confirmed by the FTIR and NMR spectra. Also, different times (1–7 days), solvents (water, methanol, EtOH, and acetone with different concentrations) and temperatures were used to find the best optimum condition.
The following tests were also performed to confirm whether there was another compound with tannic acid.
Flavonoids diagnosis test
The acetonic extract (1.0 g) was added to distilled water (10 mL), then, 100 mg of magnesium powder added to 3 mL of the resulting mixture and about 0/5 mL of concentrated HCl added as well. Since the color of the mixture was not changed into the red color, it was found that the acetonic extract does not contain flavonoids. 84
Anthocyanins diagnosis test
The pistachio extract (0.25 g) was added to distilled water (10 mL) and the resulting solution was acidified with HCl (0.1 M). Since the color did not change, it was concluded that the pistachio extract does not contain anthocyanins.85–88
Green synthesis of the TAN-Fe-NP catalyst
A mixture of FeCl3.6H2O (1.35 g, 0.005 M) and the P. atlantica tannic acid extract (8.5 g, 0.005 M, an estimated chemical formula of tannic acid is: C76H52O46, so its molecular weight (MW) is about 1700) in water was stirred for about 30 min at room temperature. The precipitate (the TAN-Fe-NP catalyst) separated by centrifugation, washed with water/EtOH, and oven dried.
General method for the synthesis of PHQs by the TAN-Fe-NP catalyst
The mixture of dimedone (1.0 mmol), ethyl acetoacetate (1.0 mmol), benzaldehydes (1.0 mmol), ammonium acetate (1.0 mmol) and the TAN-Fe-NP catalyst (20 mg) was stirred in EtOH (5 mL) at 60 °C and progress of the reaction monitored by thin layer chromatography (TLC). After completion of the reaction, the TAN-Fe-NP nanocatalyst was separated by filtration, solvent evaporated, and the precipitate washed with water and dried. Then, the resulting compound (PHQ) was characterized with FTIR and NMR.
The proposed mechanism for the synthesis of PHQs by the TAN-Fe-NP catalyst can be found in Supporting Information.
The spectral data
Ethyl 2,7,7-trimethyl-5-oxo-4-phenyl-1,4,5,6,7,8-hexahydroquinoline-3-carboxylate (
Ethyl 4-(2-methoxyphenyl)-2,7,7-trimethyl-5-oxo-1,4,5,6,7,8-hexahydroquinoline-3-carboxylate (
Ethyl 4-(2-chlorophenyl)-2,7,7-trimethyl-5-oxo-1,4,5,6,7,8-hexahydroquinoline-3-carboxylate (
Ethyl 4-(4-chlorophenyl)-2,7,7-trimethyl-5-oxo-1,4,5,6,7,8-hexahydroquinoline-3-carboxylate (
Ethyl 4-(4-nitrophenyl)-2,7,7-trimethyl-5-oxo-1,4,5,6,7,8-hexahydroquinoline-3-carboxylate (
Ethyl 4-(3-nitrophenyl)-2,7,7-trimethyl-5-oxo-1,4,5,6,7,8-hexahydroquinoline-3-carboxylate (
Ethyl 4-(3,4-dimethoxyphenyl)-2,7,7-trimethyl-5-oxo-1,4,5,6,7,8-hexahydroquino-line-3-carboxylate (2g, Table 4, entry 7). FTIR (KBr): ν (cm−1) = 3281 (N–H), 3083 (C–H), 1695 (C=O), 1604 and 1491 (C=C), 1380, 1216 (C–O), 1138, and 1029; 1H NMR (250 MHz, DMSO-d6) δ = 9.0 (s, 1H), 6.8–6.7 (m, 2H), 6.6 (d, J = 8.5 Hz, 1H), 4.8 (s, 1H), 4.0 (q, J = 7.0 Hz, 2H), 3.6 (s, 3H), 3.6 (s, 3H), 2.3 (dd, J = 19.8 Hz, 2H), 2.2 (s, 3H), 2.1 (dd, J = 34.0 Hz, 2H), 1.1 (t, J = 7.0 Hz, 3H), 1.0 (s, 3H), and 0.9 (s, 3H). 13C NMR (62.5 MHz, DMSO-d6) δ = 194.8, 167.4, 149.8, 148.4, 147.4, 145.0, 140.9, 119.7, 112.2, 111.9, 110.5, 104.3, 59.4, 55.8, 50.7, 35.6, 32.5, 29.6, 26.9, 18.7, and 14.7.
Ethyl 4-(4-bromophenyl)-2,7,7-trimethyl-5-oxo-1,4,5,6,7,8-hexahydroquinoline-3-carboxylate (
Ethyl 4-(4-methylphenyl)-2,7,7-trimethyl-5-oxo-1,4,5,6,7,8-hexahydroquinoline-3-carboxylate (
Ethyl 4-(3-hydroxyphenyl)-2,7,7-trimethyl-5-oxo-1,4,5,6,7,8-hexahydroquinoline-3-carboxylate (
Ethyl 4-(4-methoxyphenyl)-2,7,7-trimethyl-5-oxo-1,4,5,6,7,8-hexahydroquinoline-3-carboxylate (
Supplemental Material
sj-docx-1-chl-10.1177_17475198231183348 – Supplemental material for The tannic acid–based iron nanoparticle: A capable nanocatalyst for the green synthesis of polyhydroquinolines
Supplemental material, sj-docx-1-chl-10.1177_17475198231183348 for The tannic acid–based iron nanoparticle: A capable nanocatalyst for the green synthesis of polyhydroquinolines by Noorkhoda Yousefzadeh, Davood Habibi, Mohammad Hadi Meshkatalsadat and Maryam Mahmoudiani Gilan in Journal of Chemical Research
Footnotes
Acknowledgements
The authors are grateful to the Bu-Ali Sina University, Hamedan Iran, for the support of this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
