Abstract
Phorbol esters and their derivatives, such as TPA, have been reported as potential natural antitumor products, with some derivatives entering clinical research for leukemia treatment. In this study, 16 phorbol derivatives were synthesized from phorbol, and their anti-leukemia activity against Jurkat, HL-60, and K562 were tested. The results showed that several derivatives had anti-leukemia activity, with

Introduction
Acute myeloid leukemia (AML) is the most common form of leukemia among adults, 1 characterized by the malignant proliferation and accumulation of immature myeloid cells, resulting in failure of normal hematopoiesis. 2 Intensive chemotherapy (IC) and allogeneic stem cell transplantation (HSCT) have been the primary treatment strategies for AML for many years.3,4 Despite these efforts, the prognosis for individuals with AML remains poor due to high relapse rates and therapy resistance, which can further impair the immune system. Therefore, the development of new compounds with high efficacy against leukemia and low toxicity to normal cells is urgently needed.
The conventional chemotherapy drug cytarabine is derived from a natural product found in a marine sponge. 5 In addition, numerous other natural products have exhibited antitumor properties, including callyaerins, gallic acid, and shikonin.5–8 Recently, TPA (12-O-tetradecanoyl-phorbol-13-acetate, see Figure 1) has garnered attention from researchers as a promising anti-leukemia drug. TPA is a phorbol ester which was first isolated from the seed oil of croton tree, and it has shown tumor-promoting properties. 9 Weber and Hecker 10 subsequently isolated several phorbol esters from the roots of Croton flavens L., and confirmed their skin-irritating effects on mice. In addition, numerous phorbol esters have been isolated from the leaves and seed oil of Jatropha curcas L., which belongs to the same family as croton (Euphorbiaceae).11–13

Phorbol and phorbol derivatives.
J. curcas is an energy crop which is capable of withstanding drought, and it contains high oil content in its seeds. However, the utilization of the seeds and seed oil is hindered by the presence of toxic phorbol esters, which would cause skin irritation and promote tumor growth.14,15 To address this issue, many studies have been conducted in recent years to detoxify J. curcas.16–18
However, phorbol esters exhibit a dual nature, as they have both tumor-promoting and promising antitumor properties.16,19,20 The protein kinase C (PKC) family plays key roles in regulating various cellular responses, such as cell growth, differentiation, secretion, survival, and apoptosis, and is an attractive drug target for treating cancer. The diacylglycerol (DAG) within cells could activate PKC. 21 The search for drugs that could activate PKC for antitumor purposes is a promising direction. The natural product bryostatins is a typical PKC activator with good antitumor activity. 22 Phorbol esters act at the same site as DAG within cells, and both compounds activate PKC. 23 The main difference between them is that phorbol esters are not as easily metabolized as diglycerides. As a result, they can rapidly activate PKC in a short term, but prolonged exposure to phorbol esters can reduce or even inactivate PKC activity, leading to the promotion of tumor growth. Due to their unique effect on PKC, phorbol esters can not only promote the growth of tumor cells, but also exhibit potential in fighting against multiple cancers such as leukemia and viral conditions, such as AIDS.
In the treatment of acute myeloid leukemia, there are many studies about TPA, and it has entered the clinical trial stage.24,25 Various tumor-promoting phorbol esters, including TPA, also induce differentiation in certain leukemia cell lines. Takeda et al. 26 induced differentiation of ML-1 cells from patients with acute myeloid leukemia into cells with monocyte and macrophage characteristics by treatment with TPA combined with vitamin D3 analog KH1060. In addition, Zheng et al. 27 combined TPA with capsaicin on HL-60 and HL525 cells, demonstrating that capsaicin could improve the efficacy of TPA treatment and overcome the resistance of some myeloid leukemia patients to TPA. Ma et al. 28 combined DTC (diethyldithiocarbamate) and TPA in animal experiments to more effectively inhibit the growth of HL-60 tumors.
Although TPA can effectively induce leukemia cell differentiation and apoptosis, it has strong tumor-promoting activity, and some studies have proved that TPA can induce apoptosis of myeloid progenitor cells while inducing apoptosis of leukemia cells. 29 Therefore, we still need new drugs that exert low toxicity to normal cells and increase or maintain the activity against anti-leukemia cells in the meantime. Structural modifications of discovered natural compounds, especially those with promising activity, are an important tool for the discovery of new drugs. In order to optimize the synthesis strategy, researchers devoted to summarizing the relationships between the structures and biological activities of phorbol esters, including antitumor activity, tumor-promoting activity, and anti-HIV activity.
Wender et al.
30
designed and synthesized novel phorbol ester dimers and presented that the affinity of these dimers for PKC binding is related to their tether length. Among the synthesized dimers, the inhibition constant for the binding of

Chemical structures of reported phorbol derivatives. (a) dimers (b) esters (c) aminoacylphorbol derivatives.
In this study, we describe the preparation of selected phorbol 13,20-diesters (
Results and discussion
Although there are a number of studies about bioactivities of phorbol esters,34,35 a lack of a structure-activity study prompted us to design and synthesize phorbol analogs, evaluate their anti-leukemia activity, and clarify the structure-activity relationships (SARs).
With the aim to analyze the effect of an extra free hydroxyl group, phorbol 13,20-diesters and phorbol 12,13,20-triesters were prepared. Furthermore, to evaluate the effect of benzene on the biological activity, phorbols bearing p-chlorobenzoyl, cinnamoyl 4-benzoylbutyl chains were prepared. We also prepared a set of phorbol esters bearing heterocycle ester chains to analyze the effects of heteroatom-bearing substituents on activity.
Chemistry
A set of phorbol esters were prepared by selective esterification with different organic acids at C-12, C-13 and C-20 positions on the phorbol (see Scheme 1).
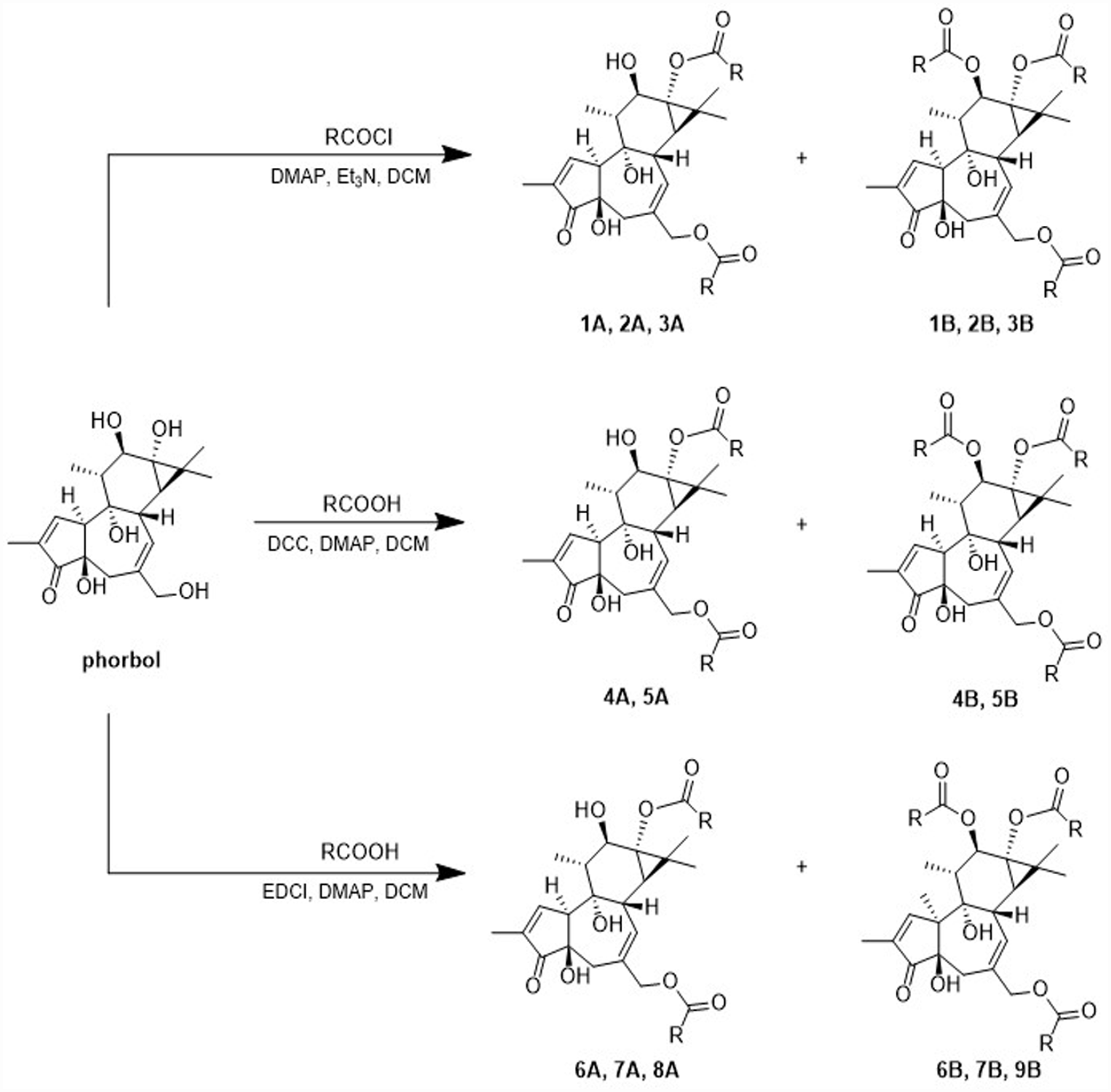
Synthesis of phorbol esters.
Phorbol 13,20-diesters and phorbol 12,13,20-triesters could be prepared from the parent compound phorbol, which is obtainable from croton oil by reported prodecure 36 or from commercial sources.
Only three of the five hydroxyl groups on phorbol are reactive, namely, those at positions C-12, C-13, and C-20. Due to steric effects, the three hydroxyl groups in phorbol are acylated in the following order: C-20 > C-13 > C-12.23,37 Therefore, in the presence of a sufficient amount of carboxylic acid or acyl chloride, the alcohol at the C-12 position of phorbol 13,20-diesters will continue to be acylated to form phorbol 12,13,20-triesters under the same conditions.
The reactivity of hydroxy with acyl chloride is higher than that with carboxylic acids. In this study, subject to the availability of commercial reagents, we gave preference to acyl chlorides for esterification.
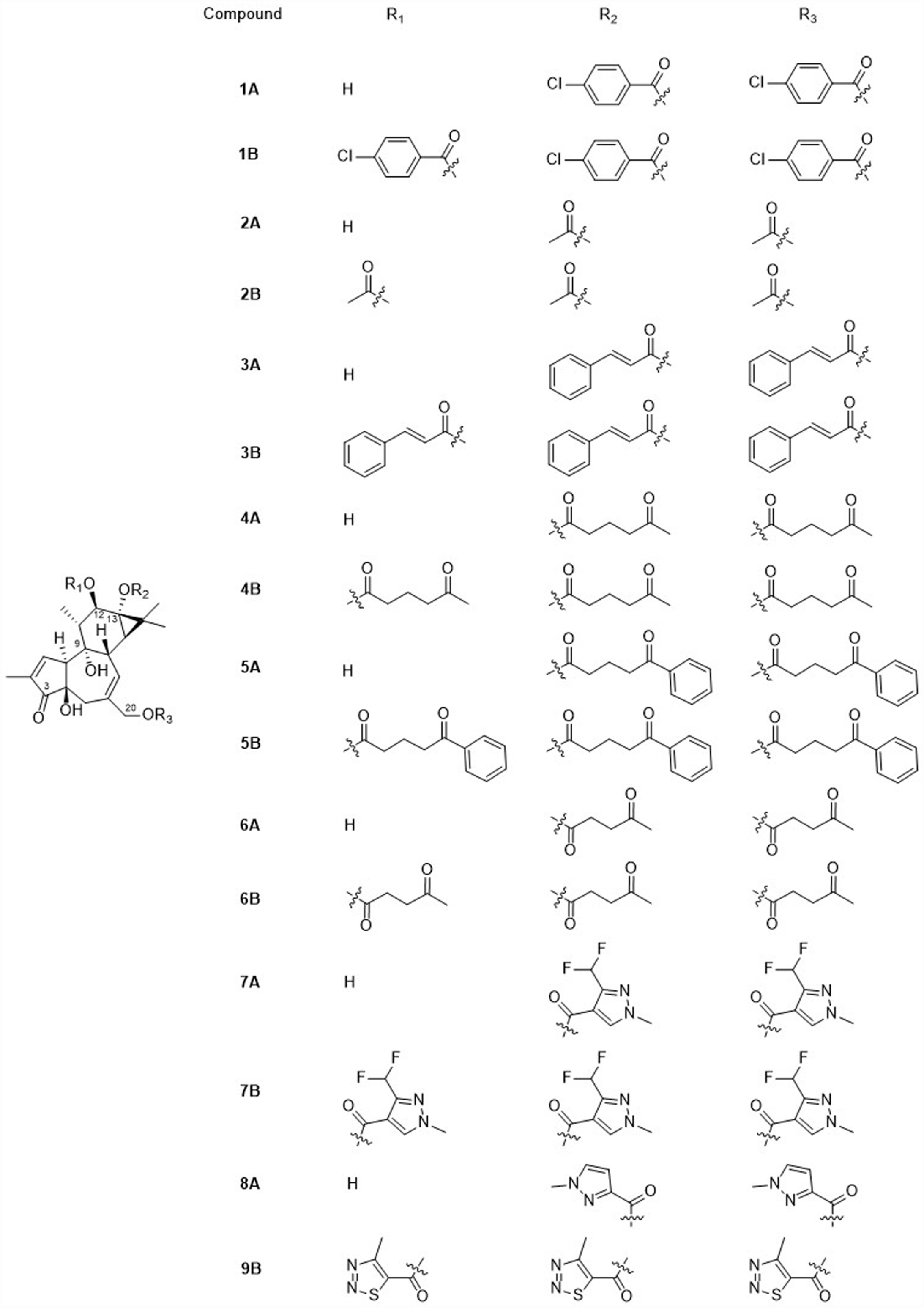
Chemical structures of phorbol derivatives.
Without commercially available acyl chloride, phorbol diesters and triesters were prepared by using the corresponding carboxylic acid instead.
Biology
In vitro cytotoxicity of phorbol derivatives
A previous study showed that TPA induced cytodifferentiation or apoptosis in leukemia cell lines by activating PKC. 38 In order to explore whether the synthesized compounds had a similar ability to induce AML cells apoptosis, we tested their cytotoxicity. We chose three leukemia cell lines (K562, Jurkat, and HL-60) and one normal human cell line (HFL1) as a comparison for any selective cytotoxicity, and the results were considered as indicators of anti-AML activity and toxicity to normal cell.
The cell proliferation or apoptosis was detected via the cell counting kit 8 (CCK8) assay, and the IC50 values were calculated for phorbol (as a negative control), TPA (as a positive control), and the results are shown in Table 1.
Activities of phorbol esters against leukemia XXXXXs (IC50).

Activities of the synthetic phorbol derivatives varied considerably against the three test leukemia cell lines. In general, the inhibitory activities of synthesized compounds were best against K562 cells. The inhibitory activity of most of the esterified phorbol derivatives against leukemia cells increased when compared against phorbol itself as a negative control, except for the derivatives produced by esterification with heterocyclic acids.
Among all 16 phorbol derivatives,
For K562 cell line, compounds
In addition, as shown in Table 1, the IC50 values of phorbol derivatives
The mechanism of phorbol derivatives against leukemia cells
On account of TPA inhibits leukemia cells by inducing apoptosis,
38
we presumed that the general mechanism of the other phorbol derivatives promoting leukemia cells death is similar. The annexin V-FITC/PI double staining assay was used to explore the induction of apoptosis in K562 cells by

Apoptosis rate.

The proportion of apoptotic cells increased as the concentration of compound
Docking analysis
According to previous studies, PKC activated by TPA generates substrate phosphorylation that induces mitogen-activated protein kinase (MAPK) cascades. 41 MAPK cascades are associated with multiple physiological processes in cells, such as growth, differentiation, and apoptosis.
DAG is the natural substrate for PKC. Phorbol esters, a variety of natural products that can replace DAG binding with the PKC C1 domain, show higher affinity than DAG. 42 This might be the reason for their ability to promote PKC activation. Phorbol derivatives were docked with PKCδC1B (PDB: 1PTR) in order to study the binding of different substitutions of phorbol to the protein.
PKCδC1B binds to phorbol 13-acetate through three residues (Gly-253, Thr-242, and Leu-251) by H-bonds. We chose the same binding groove to dock several phorbol derivatives with PKCδC1B (see Supplemental Figure S1). The docking conformation of TPA to C1B was the same with phorbol 13-acetate, the carbonyl group of C3 formed a H-bond with Gly-253, and the hydroxy groups of C20 formed two H-bonds with Gly-253, Thr-242, and Leu-251. The other compounds bind to residues differently due to the differences in position, number, and size of substituent.
Four synthesized compounds with the best activity (
Conclusion
Using phorbol as a starting scaffold, 16 phorbol esters were synthesized by esterification with three types of organic acids, namely, straight chain acids, heterocyclic acids, and aromatic acids. By comparing the anti-leukemia cellular activities of the 16 compounds, some SARs can be deduced to provide a basis and direction for future study. In vitro cytotoxicity assay indicated that substituents containing benzene rings could increase the activities of the compounds, while those containing heterocycle had no cytotoxicity. The aryl substituent-containing derivatives in which all three active hydroxyl groups were esterified have stronger inhibition effect than those in which two active hydroxyl groups were esterified. Among them,
Experimental
Chemistry
The phorbol used in the experiment was purchased from Nanjing Spring & Autumn Biological Engineering Co., Ltd. Chromatographic analysis was performed on an Agilent 1260 UPLC-DAD-6530 ESI-QTOF MS (Agilent, USA) equipped with a ZORBAX SB C18 column (1.8 μm, 100 × 4.6 mm). Mass Hunter B0.05.0 software was used as the data processor. Mobile phase-A was 1% formic acid in water and mobile phase-B was methanol, and the flow rate was maintained at 0.3 mL/min. The gradient program was 0 min-30% A, 15 min-0% A, 30 min-0% A. The column temperature was set to 35 °C. The MS parameters were as following: Ionization mode: ESI, positive ion, Mass range: 100–1000 m/z, Nebulizer pressure: 50 psi, N2 Drying Gas Flow: 10 mL·min−1, N2 Drying Gas Temp: 350 °C, Capillary Voltage: 4.0 kV. IR spectra were recorded on a Nexus 870 FT-IR spectrometer (Nicolet, USA) by the KBr disk method. The 1H and
13
C NMR spectra were measured on a Bruker DRX500 or 300 NMR spectrometer with 1H and
13
C NMR observed at 500 or 300 and 125 or 75 MHz (Bruker, Germany), using CDCl3 or DMSO-d6 as solvent, and chemical shifts are given in δ ppm relative to tetramethylsilane (TMS).
Synthesis of compounds
1A-3B
; general procedure
To a solution of phorbol (100 mg, 0.28 mmol, 1 equiv.) and Et3N (163 mg, 1.62 mmol, 6 equiv.) in anhydrous CH2Cl2 (10 mL) was added p-chlorobenzoyl chloride (100 mg, 0.57 mmol, 2 equiv.) dropwise under nitrogen at 0 °C. After stirring for 10 min, DMAP (20 mg, 0.16 mmol, 0.6 equiv.) was added. After stirring at 25 °C for 4 h, the reaction was quenched with H2O (20 mL). After partition, the aqueous layer was extracted with EtOAc (30 mL × 3), and the combined organic layers were washed with saturated aqueous NaHCO3. The pH was adjusted to neutral with 1M hydrochloric acid, and then the combined organic layers were washed with brine (20 mL × 2). The solution was dried with anhydrous Na2SO4 and concentrated to dryness in vacuo. The residue was purified by flash column chromatography (PE/EtOAc 5:1 → 3:1) to give
Synthesis of compounds
4A-5B
; general procedure
To a solution of phorbol (100 mg, 0.28 mmol, 1 equiv.) and 5-oxohexanoic acid (178 mg, 1.37 mmol, 5 equiv.) in anhydrous CH2Cl2 (10 mL) was added DCC (20 mg, 0.12 mmol, 1.2 equiv.) dropwise under nitrogen at 0 °C. After stirring for 15 min, 4-dimethylaminopyridine (DMAP, 6 mg, 0.05 mmol, 0.5 equiv.) was added at same temperature and stirred for 10 min. After stirring at 25 °C for 4 h, the reaction was quenched with H2O (20 mL). After partition, the aqueous layer was extracted with EtOAc (30 mL × 3), and the combined organic layers were washed with brine (20 mL × 2).
The solution was dried with anhydrous Na2SO4 and concentrated to dryness in vacuo. The residue was purified by flash column chromatography (PE/EtOAc 1:1 → PE/EtOAc 1:2) to give
Synthesis of compounds
6A-9B
; general procedure
A three-necked flask was charged with phorbol (100 mg, 0.28 mmol, 1 equiv.) and levulinic acid (159 mg, 1.37 mmol, 5 equiv.) and then filled with nitrogen. EDCI (86 mg, 1.5 mmol, 5.5 equiv.) was added dropwise at 0 °C. After stirring for 15 min, DMAP (20 mg, 0.16 mmol, 0.6 equiv.) was added at same temperature and stirred for 10 min. After stirring at 25 °C for 4 h, the reaction was quenched with H2O (20 mL). After partition, the aqueous layer was extracted with EtOAc (30 mL × 3), and the combined organic layers were washed with brine (20 mL × 2). The solution was dried with anhydrous Na2SO4 and concentrated to dryness in vacuo. The residue was purified by flash column chromatography (PE/EtOAc 1:2) to give
Via the same procedure,
Biology
The CCK-8 (Cell counting kit-8) kit was used to detect the proliferation of tumor cells during the experiment. This is a WST-8 based kit for the detection of cell proliferation and cytotoxicity, which is highly sensitive and rapid. The MTT colourimetric assay was used to detect the proliferation of normal cells during the assay. Sixteen synthetic phorbol esters were tested for in vitro cytotoxicity against three leukemia cell lines (K562, Jurkat, and HL-60 cell lines).
In vitro anti-leukemia cells activity
Cells recovery: K562 cells, HL-60 cells, Jurkat cells, and HFL1 cells, which were frozen in liquid nitrogen tanks, were thawed in warm water at 37–39 °C. All resurgent cell lines were grown and maintained in RPMI-1640 medium with 10% fetal bovine serum (FBS) and with 1% penicillin/streptomycin at 37 °C in a 95% air −5% CO2 humidified incubator. Observe the cell growth status in time, and replace the culture medium according to the situation.
Preparation of the test compound solutions: the experimental group, the blank control group, and the positive control group were set up with three replicates in each group. The sixteen phorbol esters, TPA, and phorbol were dissolved in DMSO at the concentration of 10 mmol L−1 as stock solutions, and diluted in culture medium at the concentrations of 6.25, 25, 100, 400, and 1600 nM as working-solutions. TPA was used as the positive control (the experiments have been performed in triplicate).
Cell viability assay: three types of leukemia cells and HFL1 cells were seeded at a density of 5 × 104 cells per well in 96-well plates and grown in 5% CO2 at 37 °C. After 4 h of incubation, cells were treated with various concentrations of compounds or solvent control. Incubation was continued for 48 h at the same condition, and then CCK-8 (5 μL) was added to each well. After 4 h, the OD value of each well was measured on a microplate spectrophotometer at the wavelength of 450 nm.
For HFL1 cells, cells viability was detected by MTT assay. MTT dye solution (10 μL, 5 mg L−1, PBS) was added to each well. After incubated in 5% CO2 at 37 °C for 4 h, 100 μL DMSO was added to each well and the plates were shaked for 5 min. The OD value of each well was measured on a microplate spectrophotometer at the wavelength of 570 nm. The calculation formula of the cell proliferation inhibition rate is as follows:
Cell proliferation inhibition rate (%) = (1 − OD value of experimental group/OD value of blank group) × 100%
The inhibitory rate of each drug on each cell was measured under five concentration gradients, namely, 1600, 400, 100, 25, and 6.25 nmol L−1, and the IC50 value was calculated.
Annexin V-FITC/PI double staining assay for apoptosis
In this experiment, annexin V-FITC apoptosis detection kit was used to detect the apoptosis of tumor cells during the experiment.
According to the test results of anti-leukemia cells activity in vitro, K562 cells and compound
The K562 cells were adjusted to a density of 5 × 104 cells mL−1, and 2 mL of the cell suspension was seeded in four disposable petri dishes in each dish. The petri dishes were then incubated for 4 h in 5% CO2 at 37 °C. The 1640 medium was addedto dilute stock solution of compound
Molecular docking
Molecular docking study of PCKδC1B (PDB code: 1PTR) and phorbol derivatives was performed in AutoDock-Vina program. 44 The ligand compound was allowed to dock anywhere in a 22 Å × 22 Å × 18 Å volume centered around active site, with the Lamarckian genetic algorithm (LGA). The maximum numbers of energy evaluation and generation were set to 250,000 and 25,000, respectively. A total of 20 separate docking runs were performed with the initial population of 20 individuals, and the lowest energy structure from each run was retained. Final docked conformations were clustered by using a tolerance of 1.0 Å root mean square deviation (RMSD). The complex with the strongest binding free energy was picked out as the best conformation for further analyzed.
Supplemental Material
sj-docx-1-chl-10.1177_17475198231180835 – Supplemental material for Synthesis and anti-leukemia activity of phorbol 13,20-diesters and phorbol 12,13,20-triesters
Supplemental material, sj-docx-1-chl-10.1177_17475198231180835 for Synthesis and anti-leukemia activity of phorbol 13,20-diesters and phorbol 12,13,20-triesters by Yan Wang, Yu Shan, Rui Feng, Siyu Wang, Linwei Li, Shu Xu, Yu Chen, Xu Feng, Jinyue Luo and Fei Liu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant nos 32000258 and 31970375).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
