Abstract
A series of novel quinazoline derivatives containing piperazine analogs are synthesized via substitution reactions with 6,7-disubstituted 4-chloroquinazoline and benzyl piperazine (amido piperazine). Potent antiproliferative activities are observed against A549, HepG2, K562, and PC-3 with N-(3-chlorophenyl)-2-(4-(7-methoxy-6-(3-morpholino-propoxy)quinazoline-4-yl)piperazine-1-yl)acetamidename

Introduction
It has been reported that the quinazoline unit is of great value as a chemical skeleton with diverse pharmaceutical and physiological utility, including antimicrobial, 1 antihyperlipidemic, 2 anticonvulsant, 3 antihypertensive, 4 and anti-inflammatory 5 activities. Therefore, many quinazolines have contributed to the quest for an ultimate antitumor chemotherapeutic agent. Moreover, several small molecules such as gefitinib (Figure 1), erlotinib, and lapatinib, which containing quinazoline analogs, were designed to inhibit epidermal growth factor receptor (EGFR) kinase activity. Among them, gefitinib—an oral small molecule agent that inhibits EGFR tyrosine phosphorylation, was approved by the US Food and Drug Administration (FDA) for locally advanced or metastatic non-small cell lung cancer therapy—is involved in cellular signal-transduction pathways that regulate essential functions such as proliferation, differentiation, and apoptosis.6–14 However, its efficient use is seriously hampered by EGFR mutation. Even though tumors were sensitive to gefitinib, tumor regrowth occurs after several months and all of the mechanisms of the resistance have not been clarified. Thus, it is necessary to exploit novel antitumor drugs with high efficiency and low toxicity due to the various side effects of gefitinib. 15

The structure of gefitinib.
Over past decades, a large number of piperazine heterocyclic derivatives have attracted significant attention as chemotherapeutic drugs. Numerous studies have illustrated that the introduction of piperazine moieties can influence the physicochemical properties and enhance the bioactivity of compounds. It is known that piperazine is versatile and privileged in drug discovery,16–20 for example, in ciprofloxacin for the treatment of bacterial infections, trifluoperazine for psychosis, cetirizine for the treatment of allergies, and tanitinib for the treatment of acute myeloid leukemia. A series of 4-amino-2H-benzo(h)chromen-2-one analogs containing the piperazine moiety were reported by Chen in 2019, and bioassays showed that these compounds exhibited potent antagonistic potency against androgen receptors (AR; inhibition >50%), and exhibited potent AR binding affinities as well as displaying higher activities than finasteride toward LNCaP cells (AR-rich) versus PC-3 cells (AR-deficient). 21
Based on the active substructure combination theory, the piperazine ring was utilized in this research. Thus, two series of novel 4-piperazine quinazoline derivatives were designed and synthesized. The preliminary antitumor activity of these new compounds was also evaluated in vitro. In addition, the primary apoptotic effect mechanism induced by a representative compound
Results and discussion
Synthesis
The synthetic route for the preparation of the novel quinazoline derivatives containing a piperazine moiety is summarized in Scheme 1. The key intermediate
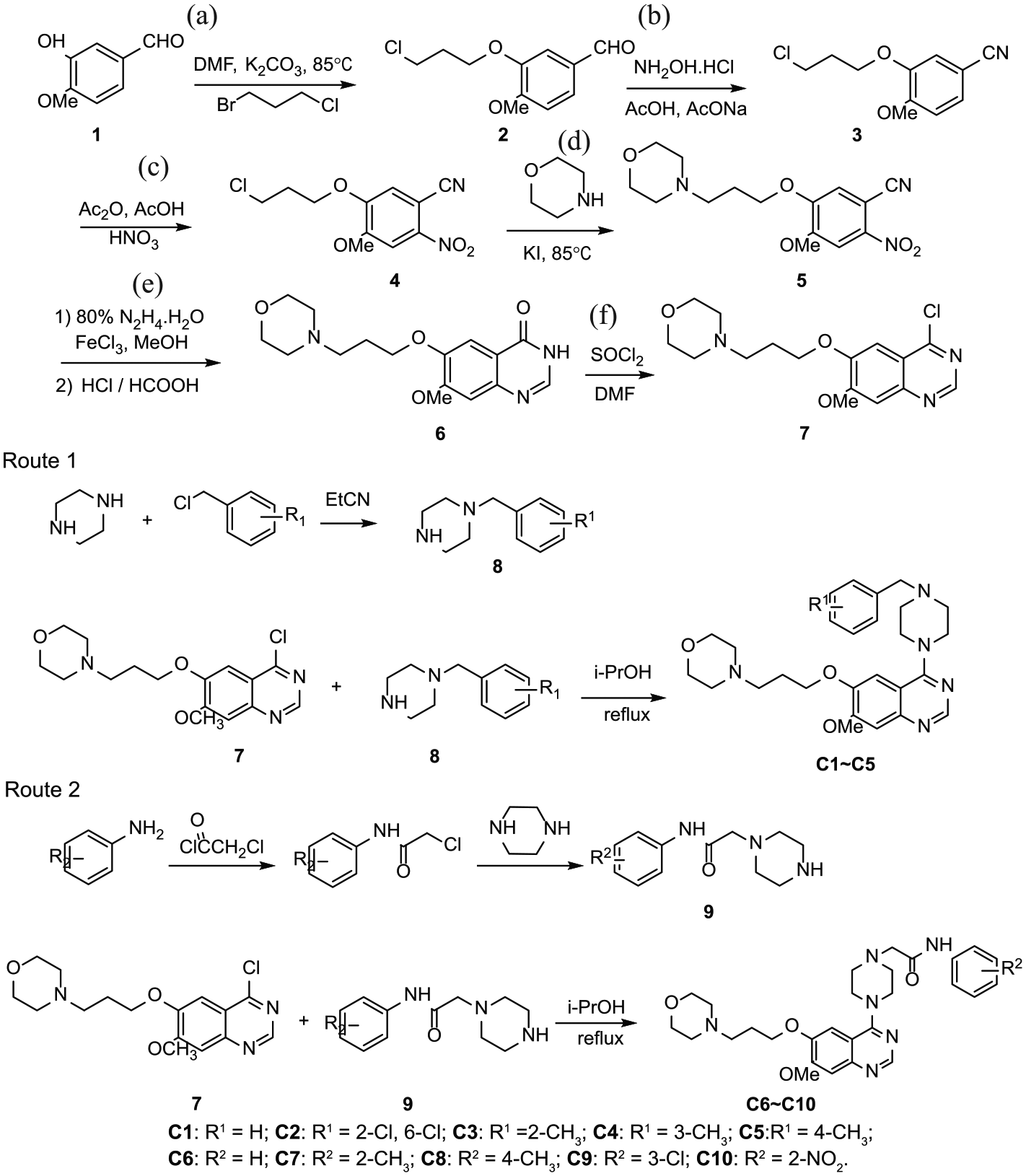
Synthetic routes to the target compounds
Biological activity
The antitumor activities of the synthesized compounds against four cell lines were evaluated using gefitinib as the control. The IC50 values indicated that the compounds exerted antitumor effects, with varying degrees of antitumor activity against the four cell lines. As shown in Table 1, the IC50 values ranged from 35.56 ± 9.39 to 8.24 ± 1.40 µM against A549, from 14.57 ± 5.52 to 5.62 ± 3.23 µM against PC-3, from 56.42 ± 0.14 to 8.63 ± 0.86 µM against HepG2, and from 159.3 ± 14.28 to 15.02 ± 0.88 µM against K562, being typically better than gefitinib (IC50 values were 5.475 ± 1.06 µM against A549, 5.99 ± 1.47 µM against PC-3, 31.48 ± 10.23 µM against HepG2, and 13.67 ± 1.44 µM against K562). The cytotoxicities of the target compounds against the normal cell line NRK-52E in vitro are shown in the Supporting Information Figure S1). The result showed that the survival rate of the cells at low concentration was close to 100%, and the cytotoxicity of compound
The IC50 values of compounds
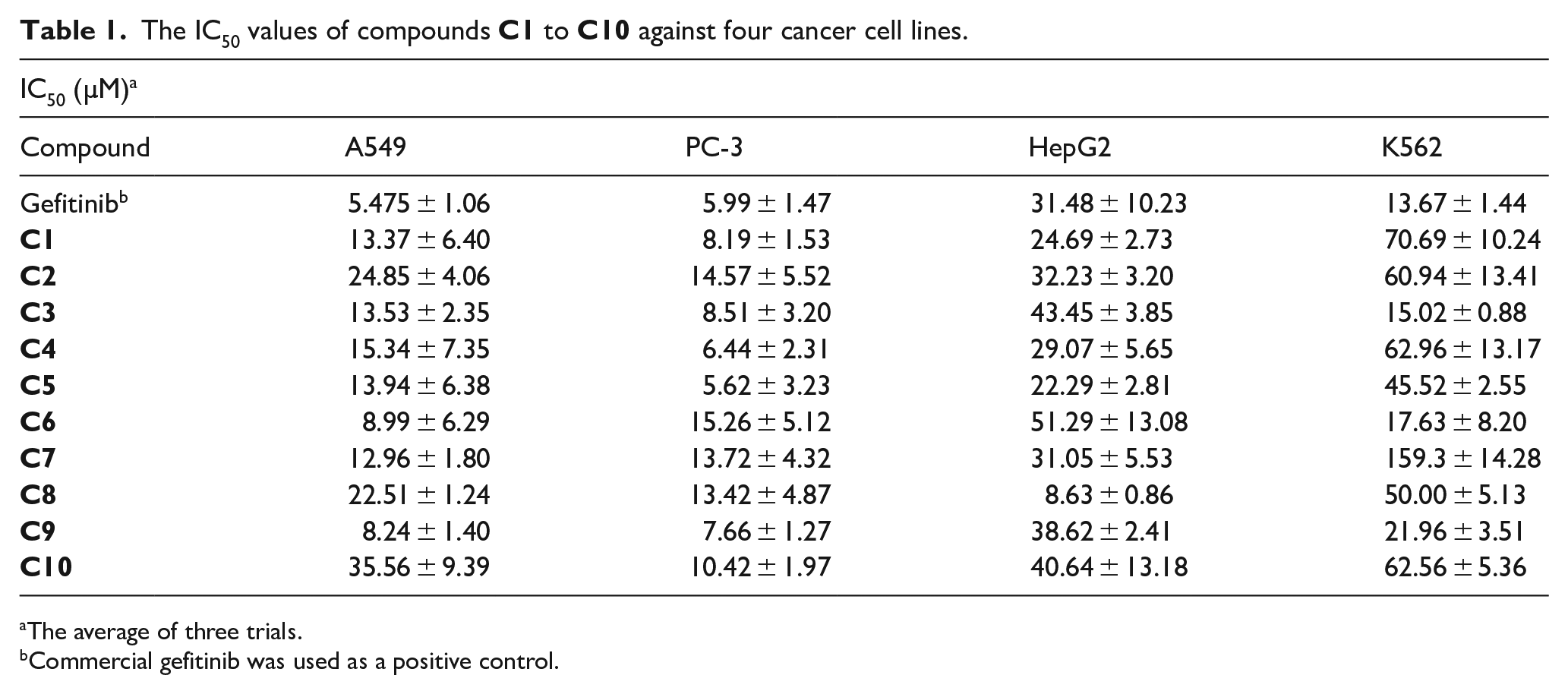
The average of three trials.
Commercial gefitinib was used as a positive control.
To further investigate the cell migration ability of compound
To evaluate the cell proliferation effects of
In order to investigate the induction of apoptosis in A549 and PC-3 cells, the Hoechst staining assay was conducted in nuclear morphology by fluorescent microscopy. As shown in Figure S4, it was easy to observe the distinct pattern of morphological changes, including cell shrinkage, fragmentation of the nucleus, and chromatin condensation after culturing with gefitinib and
Flow cytometry
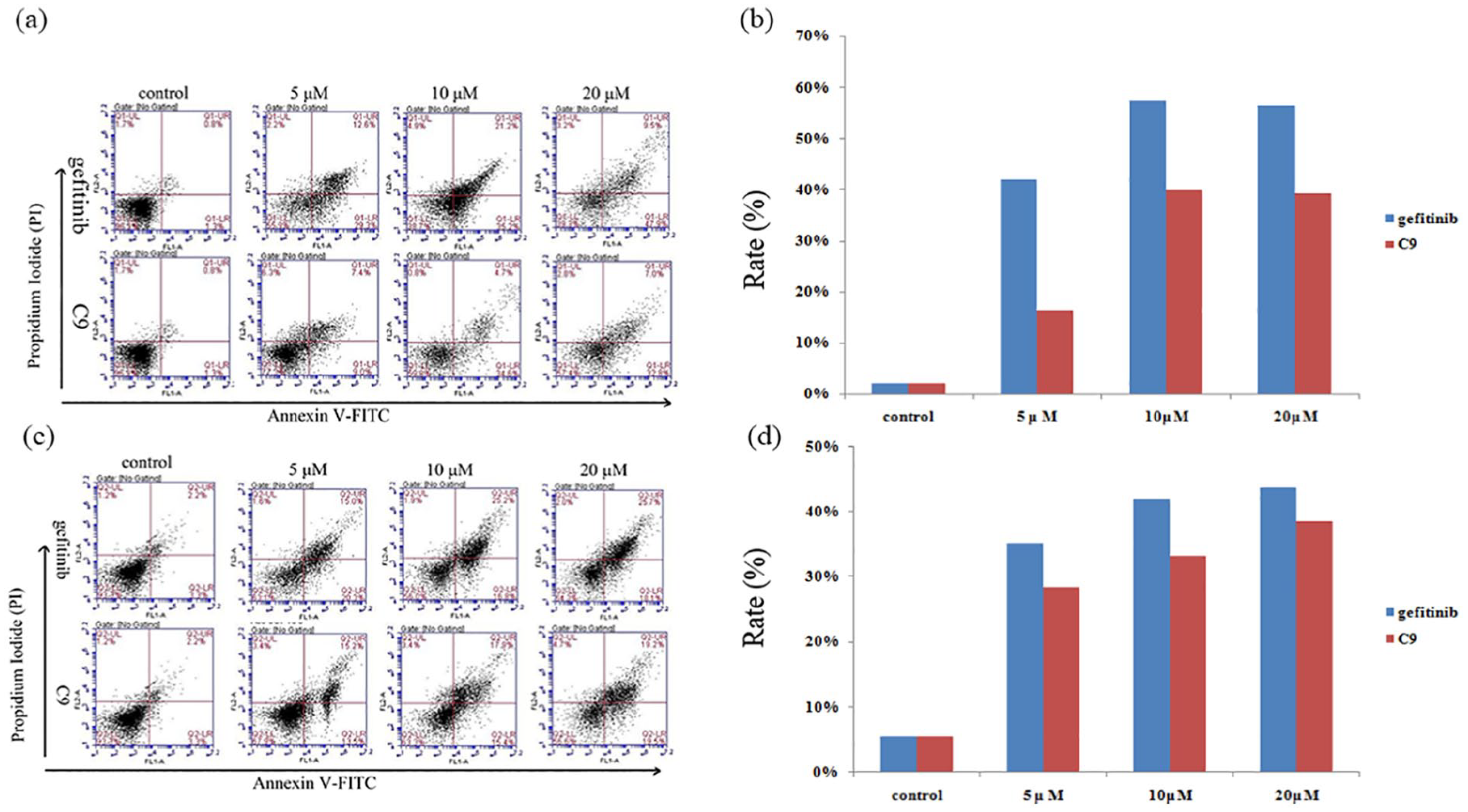
Apoptosis assays can provide preliminary information on the mechanism of growth inhibition of tumor cells. Therefore, the apoptotic effects of
Binding mode analysis
In order to further evaluate these promising compounds and to guide structure–activity relationship (SAR) studies, the interaction effect between compound

(a) Compound
Conclusion
In conclusion, a series of quinazoline derivatives containing a piperazine moiety have been designed and synthesized. Furthermore, the biological activities of these compounds were evaluated according to the Thiazolyl Blue Tetrazolium Bromide (MTT) method, wound healing assay, clonogenic survival assay, Hoechst staining, and flow cytometry. Compound
Experimental
Materials and methods
All reagents were purchased from commercial sources as analytical grade and were used without further purification. All reactions were monitored by thin-layer chromatography (TLC). 1H NMR and 13C NMR spectra were performed on a Bruker Ascend 400 NMR (Bruker, Germany) and JEOL-ECX 500 NMR spectrometers (JEOL, Japan) using methanol (CD3OD) as the solvent and tetramethylsilane as the internal standard. Chemical shifts are expressed in ppm (parts per million) and coupling constants are given in Hz (Hertz). Mass spectra (MS) were recorded on an Agilent 1100 MSD-Trap-VL series with an electron spray ionization (ESI) source. High-resolution mass spectra (HRMS) were recorded on Thermo Scientific Q Exactive series with an ESI source.
The synthesis of intermediate 7
To a solution of
General procedure for the preparation of C1 –C5
Taking compound
General procedure for the preparation of C6 –C10
Taking compound
4-(4-Benzylpiperazin-1-yl)-7-methoxy-6-[3-(4-morpholinyl)propoxy]quinazoline (C1 )
Yield 11%, 0.08 g; yellow oily substance; 1H NMR (CD3OD, 500 MHz): δ 8.44 (s, 1H), 7.23–7.35 (m, 5H), 7.12 (s, 2H), 4.12 (t, J = 6.0 Hz, 2H), 3.94 (s, 3H), 3.68–3.70 (m, 8H), 3.58 (s, 2H), 2.49–2.63 (br, m, 10H), 2.03–2.06 (br, m, 2H); 13C NMR (CD3OD, 125 MHz): δ 163.6, 155.5, 152.0, 148.2, 137.1, 129.3, 128.1, 127.2, 110.6, 105.8, 104.5, 66.9, 66.3, 62.6, 55.4, 55.3, 53.5, 52.6, 49.2, 25.6; ESI-MS: m/z 478.2 [M + H]+. HRMS (ESI) m/z calcd for C27H35N5O3 [M + H]+ 478.2813, found: 478.2808.
4-(4-(2,6-Dichlorobenzyl)piperazin-1-yl)-7-methoxy-6-[3-(4-morpholinyl)propoxy]quinazoline (C2 )
Yield 7%, 0.05 g; yellow oily substance; 1H NMR (CD3OD, 500 MHz): δ 8.43 (s, 1H), 7.35 (d, J = 8.0 Hz, 2H), 7.22 (t, J = 8.0 Hz, 1H), 7.11 (s, 1H), 7.10 (s, 1H), 4.12 (t, J = 6.0 Hz, 2H), 3.94 (s, 3H), 3.82 (s, 2H), 3.63–3.69 (m, 8H), 2.74 (t, J = 4.6 Hz, 4H), 2.49–2.57 (m, 6H), 2.03–2.06 (br, m, 2H); 13C NMR (CD3OD, 125 MHz): δ 163.6, 155.5, 152.0, 148.2, 148.1, 136.8, 133.7, 129.4, 128.3, 110.9, 105.8, 104.6, 66.9, 66.3, 56.0, 55.4, 55.3, 53.5, 52.7, 49.4, 25.7; ESI-MS: m/z 546.4 [M + H]+. HRMS (ESI) m/z calcd for C27H33Cl2N5O3 [M + H]+ 546.2033, found: 546.2026.
4-(4-(2-Methylbenzyl)piperazin-1-yl)-7-methoxy-6-[3-(4-morpholinyl)propoxy]quinazoline (C3 )
Yield 17%, 0.12 g; yellow oily substance; 1H NMR (CD3OD, 500 MHz): δ 8.38 (s, 1H), 6.91–7.13 (br, m, 6H), 3.99 (t, J = 6.0 Hz, 2H), 3.86 (s, 3H), 3.61 (t, J = 4.6 Hz, 4H), 3.41–3.51 (br, m, 4H), 3.39 (s, 2H), 2.46–2.49 (m, 6H), 2.27–2.39 (br, m, 7H), 1.94–1.97 (br, m, 2H); 13C NMR (CD3OD, 125 MHz): δ 163.5, 163.4, 155.2, 152.1, 147.9, 137.4, 135.6, 130.2, 129.9, 127.2, 125.4, 110.7, 106.0, 104.3, 67.0, 66.4, 60.5, 55.4, 55.3, 53.5, 52.8, 49.4, 25.7, 18.4; ESI-MS: m/z 492.5[M + H]+. HRMS (ESI) m/z calcd for C28H37N5O3 [M + H]+ 492.2969, found: 492.2963.
4-(4-(3-Methylbenzyl)piperazin-1-yl)-7-methoxy-6-[3-(4-morpholinyl)propoxy]quinazoline (C4 )
Yield 15 %, 0.11 g; yellow oily substance; 1H NMR (CD3OD, 500 MHz): δ 8.39 (s, 1H), 7.12 (t, J = 7.5 Hz, 1H), 7.04–7.08 (m, 2H), 6.98–7.00 (m, 2H), 6.94 (s, 1H), 4.02 (t, J = 6.0 Hz, 2H), 3.88 (s, 3H), 3.63 (t, J = 2.1 Hz, 4H), 3.58 (s, 4H), 3.42 (s, 2H), 2.40–2.53 (m, 6H), 2.25–2.40 (br, m, 7H), 1.96–1.99 (br, m, 2H); 13C NMR (CD3OD, 125 MHz): δ 163.4, 155.2, 152.1, 148.1, 148.0, 137.7, 137.1, 129.9, 128.0, 127.9, 126.4, 110.8, 105.9, 104.3, 66.9, 66.4, 62.7, 55.4, 55.3, 53.5, 52.7, 49.2, 25.8, 20.4; ESI-MS: m/z 492.5 [M + H]+. HRMS (ESI) m/z calcd for C28H37N5O3 [M + H]+ 492.2969, found: 492.2966.
4-(4-(4-Methylbenzyl)piperazin-1-yl)-7-methoxy-6-[3-(4-morpholinyl)propoxy]quinazoline (C5 )
Yield 14%, 0.10 g; yellow oily substance; 1H NMR (CD3OD, 500 MHz): δ 8.59 (s, 1H), 8.42 (s, 1H), 7.85 (t, J = 9.9 Hz, 2H), 7.45 (t, J = 8.2 Hz, 2H), 7.06 (s, 2H), 4.10 (t, J = 5.7 Hz, 2H), 3.93 (s, 3H), 3.74 (br, s, 6H), 3.65 (t, J = 4.6 Hz, 4H), 3.29 (s, 3H), 2.80 (t, J = 4.6 Hz, 4H), 2.54 (t, J = 7.3 Hz, 2H), 2.46 (s, 3H), 2.01–2.04 (br, m, 2H); 13C NMR (CD3OD, 125 MHz): δ 163.6, 155.4, 152.0, 148.2, 148.0, 136.9, 133.9, 129.3, 128.7, 110.9, 105.9, 104.4, 66.9, 66.4, 62.4, 55.4, 55.3, 53.5, 52.6, 49.2, 25.7, 19.9; ESI-MS: m/z 492.5 [M + H]+. HRMS (ESI) m/z calcd for C28H37N5O3 [M + H]+ 492.2969, found: 492.2962.
2-(4-(7-Methoxy-6-(3-morpholinopropoxy)quinazolin-4-yl)piperazin-1-yl)-N-phenylacetamide (C6 )
Yield 13%, 0.10 g; yellow oily substance; 1H NMR (CD3OD, 500 MHz): δ 8.43 (s, 1H), 7.57 (d, J = 8.0 Hz, 2H), 7.26 (d, J = 9.2 Hz, 2H), 7.06 (s, 1H), 7.04 (s, 2H), 4.09 (t, J = 6.0 Hz, 2H), 3.92 (s, 3H), 3.72 (s, 4H), 3.64 (t, J = 4.9 Hz, 4H), 3.23 (s, 2H), 2.76 (t, J = 4.5 Hz, 4H), 2.54 (t, J = 7.5 Hz, 2H), 2.45 (s, 4H), 2.45–2.52 (m, 4H), 2.02–2.03 (br, m, 2H); 13C NMR (CD3OD, 125 MHz): δ 169.3, 163.5, 155.4, 152.0, 148.2, 148.0, 137.9, 128.6, 124.8, 120.0, 110.9, 105.9, 104.3, 67.0, 66.3, 61.6, 55.4, 55.3, 53.5, 52.9, 49.3, 25.7; ESI-MS: m/z 521.4 [M + H]+. HRMS (ESI) m/z calcd for C28H36N6O4 [M + H]+ 521.2871, found: 521.2863.
2-(4-(7-Methoxy-6-(3-morpholinopropoxy)quinazolin-4-yl)piperazin-1-yl)-N-(o-methylphenyl)acetamide (C7 )
Yield 13%, 0.10 g; yellow oily substance; 1H NMR (CD3OD, 500 MHz): δ 8.46 (s, 1H), 7.69 (d, J = 8.3 Hz, 1H), 7.03–7.18 (m, 5H), 4.12 (t, J = 6.2 Hz, 2H), 3.94 (s, 3H), 3.67–3.74 (m, 8H), 3.26 (s, 2H), 2.82 (t, J = 4.6 Hz, 4H), 2.56 (t, J = 7.6 Hz, 2H), 2.47 (s, 4H), 2.27 (s, 3H), 2.03–2.06 (br, m, 2H); 13C NMR (CD3OD, 125 MHz): δ 169.5, 163.6, 155.5, 152.0, 148.3, 148.1, 135.4, 130.2, 130.1, 126.3, 125.3, 123.0, 111.0, 105.9, 104.3, 67.3, 66.3, 61.4, 55.4, 55.3, 53.5, 53.0, 49.6, 25.7, 16.9; ESI-MS: m/z 535.5 [M + H]+. HRMS (ESI) m/z calcd for C29H38N6O4 [M + H]+ 535.3027, found: 535.3019.
2-(4-(7-Methoxy-6-(3-morpholinopropoxy)quinazolin-4-yl)piperazin-1-yl)-N-(p-methylphenyl)acetamide (C8 )
Yield 16%, 0.13 g; yellow oily substance; 1H NMR (CD3OD, 500 MHz): δ 8.74 (s, 1H), 7.44 (d, J = 8.6 Hz, 2H), 7.16 (d, J = 11.5 Hz, 2H), 7.11 (d, J = 8.6 Hz, 2H), 4.16 (t, J = 6.0 Hz, 2H), 3.96 (s, 3H), 3.80 (s, 4H), 3.69 (t, J = 4.6 Hz, 4H), 3.25 (s, 2H), 2.80 (t, J = 4.6 Hz, 4H), 2.61 (t, J = 7.5 Hz, 2H), 2.53 (s, 4H), 2.28 (s, 3H), 2.05–2.08 (br, m, 2H); 13C NMR (CD3OD, 125 MHz): δ 169.3, 163.7, 155.6, 152.0, 148.3, 148.1, 135.2, 134.0, 128.9, 120.2, 110.9, 105.8, 104.5, 66.9, 66.2, 61.5, 55.4, 55.3, 53.4, 52.8, 49.3, 25.6, 19.6; ESI-MS: m/z 535.5 [M + H]+. HRMS (ESI) m/z calcd for C29H38N6O4 [M + H]+ 535.3027, found: 530.3018.
N-(3-Chlorophenyl)-2-(4-(7-methoxy-6-(3-morpholinopropoxy)quinazolin-4-yl)piperazin-1-yl)acetamide (C9 )
Yield 15%, 0.12 g; yellow oily substance; 1H NMR (CD3OD, 500 MHz): δ 8.41 (s, 1H), 7.74 (t, J = 2.5 Hz, 1H), 7.40–7.42 (m, 2H), 7.17–7.21 (m, 1H), 6.97–7.03 (m, 2H), 4.06 (t, J = 6.0 Hz, 2H), 3.91 (s, 3H), 3.63–3.69 (m, 8H), 3.21 (s, 2H), 2.74 (t, J = 7.4 Hz, 4H), 2.43–2.51 (m, 6H), 1.99–2.02 (br, m, 2H); 13C NMR (CD3OD, 125 MHz): δ 169.5, 163.5, 155.3, 152.0, 148.1, 148.0, 139.4, 134.0, 129.8, 123.8, 119.7, 117.9, 110.9, 105.9, 104.3, 67.0, 66.3, 61.6, 55.4, 55.3, 53.4, 52.8, 49.3, 25.7; ESI-MS: m/z 555.4 [M + H]+. HRMS (ESI) m/z calcd for C28H35ClN6O4 [M + H]+ 555.2481, found: 555.2474.
2-(4-(7-Methoxy-6-(3-morpholinopropoxy)quinazolin-4-yl)piperazin-1-yl)-N-(2-nitrophenyl)acetamide (C10 )
Yield 8%, 0.07 g; yellow oily substance; 1H NMR (CD3OD, 500 MHz): δ 8.66 (d, J = 7.3 Hz, 1H), 8.44 (s, 1H), 8.14 (d, J = 6.6 Hz, 1H), 7.66 (t, J = 7.1 Hz, 1H), 7.22 (t, J = 7.8 Hz, 1H), 7.11 (d, J = 9.6 Hz, 2H), 4.13 (t, J = 6.2 Hz, 2H), 3.94 (s, 3H), 3.84 (m, 4H), 3.67 (t, J = 4.6 Hz, 4H), 3.29 (s, 2H), 2.82 (t, J = 4.6 Hz, 4H), 2.57 (t, J = 7.6 Hz, 2H), 2.48 (s, 4H), 2.03–2.06 (br, m, 2H); 13C NMR (CD3OD, 125 MHz): δ 170.7, 163.6, 155.5, 152.0, 148.3, 148.1, 137.3, 135.3, 125.5, 123.7, 121.9, 110.9, 106.0, 104.6, 67.2, 66.3, 61.7, 61.5, 55.4, 53.4, 52.9, 49.5, 25.7; ESI-MS: m/z 566.4 [M + H]+. HRMS (ESI) m/z calcd for C28H35N7O6 [M + H]+ 566.2722, found: 566.2716.
Biological activity
The antitumor activities of the products against A549, K562, HepG2, and PC-3 were tested at five concentrations (0.625, 1.25, 2.5, 5, and 10 µM). Each test was done five times and assessed according to the MTT assay. The cell cultures were obtained from the Kunming Cell Bank. PC-3 was cultured at 37 °C and 5% CO2 in Dulbecco’s modified eagle medium supplemented with 10% fetal bovine serum and 1% penicillin streptomycin. A549, K562, and HepG2 cells were cultured in Roswell Park Memorial Institute (RPMI) 1640 supplemented with the same mixture. The cells were inoculated in a 96-well plate in 180 µL aliquots at 3 × 104 cells/mL and cultivated for 1 day. The plates were further incubated for 48 h, and the assay was terminated by the addition of 20 µL of 5 mg/mL MTT. Incubation with the dye was continued for 4 h, after which time the medium was removed and the dye was extracted from the cells with 150 µL of DMSO. The absorbance was measured with a Microplate Reader (TECAN infinite M200 pro, TECAN, Switzerland) at 490 nm wavelength. The growth percentage was calculated on a plate by plate basis for test wells relative to control wells. The sensitivity of the cancer cells to each test compound was expressed in terms of IC50, indicated as mean values ± SD for three independent experiments. Statistical analysis was performed using GraphPad Prism (version 7) for the Student’s test. Values of p < .05 were considered significant.
The wound healing assay was as follows. Cells were seeded in a six-well plate and grown for 24 h to 80% to 90% confluence. Cells were washed twice with phosphate-buffered saline (PBS), and the monolayers were scraped with a micropipette tip to create a uniform scratch. Next, cells were washed with PBS again to remove the detached cells. The compounds were added in the RPMI medium at 5 µM. DMSO was used as a control. Digital images of the wounded monolayers were obtained using a photomicroscope (Ti-S, Nikon, Japan) at 0, 6, 12, and 24 h. The unfilled scratched zones were quantified by Java’s ImageJ software.23,24
Antiproliferative activity was measured by the clonogenic survival assay after treating with the test compound. Briefly, cells were seeded in a six-well plate at 3 × 105 cells/well and were stimulated by a range of compound doses (1.25, 2.5, 5, 10, and 20 µM). The plates were further incubated for 24 h and the cells were plated in six wells at 1000 cells per well cultured for 12 days, and the number of surviving colonies (defined as a colony with >50 cells) was counted. The survival fraction was calculated as the number of colonies divided by the number of cells seeded times plating efficiency. Three independent experiments were performed.25,26
Hoechst staining of the cultured cells was used to observe apoptotic nuclei by evaluation of nuclear morphology using a fluorescence inverted microscope. Cells were fixed in 4% paraformaldehyde solution for 10 min at RT. Then the cells were incubated with Hoechst 33258 dye for 20 min. Next, the cells were rinsed with precooling PBS and were observed under a fluorescence inverted microscope with 20 µL of antifluorescent quencher after air-drying. Typical nuclei showed non-condensed chromatin distributed over the whole nucleus. In contrast, apoptotic nuclei were identified by nuclear fragmentation of condensed chromatin. 27
Flow cytometry
To evaluate cell apoptosis, a double staining trial with propidium iodide (PI) and fluorescein isothiocyanate isomer (FITC)-conjugated Annexin V (Solabio, Beijing, China) was performed. Cells were seeded in a six-well plate at 1 × 106 cells/well and were stimulated by a range of compound doses (5, 10, and 20 µM). The original culture medium was collected and the cells were cultured with trypsin, which were subsequently washed twice in binding buffer. Next, 5 µL of Annexin V-FITC and 5 µL of PI were adopted for dying cells for 15 min at RT. The apoptotic A549 and PC-3 cells were observed via flow cytometry (BD Accuri™ C6, BD Biosciences, USA) equipment.28,29
Supplemental Material
JCR_Supplementary_Information-Revised – Supplemental material for Design, synthesis, and biological evaluation of quinazoline derivatives containing piperazine moieties as antitumor agents
Supplemental material, JCR_Supplementary_Information-Revised for Design, synthesis, and biological evaluation of quinazoline derivatives containing piperazine moieties as antitumor agents by Wen Li, Shu-Yi Chen, Wei-Nan Hu, Mei Zhu, Jia-Min Liu, Yi-Hong Fu, Zhen-Chao Wang and Gui-Ping OuYang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (No. 21867004); the Project of the State Key Laboratory of Functions and Applications of Medicinal Plants, Guizhou Medical University (No. FAMP2018); and the Science Technology Program of Guizhou Province (No. 20185781).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
