Abstract
Novel 3-(3-benzyl-2,3-dihydrobenzo[d]thiazol-2-yl)benzo[d]oxazole-2(3H)-thione derivatives are efficiently synthesized in excellent yields by reactions of benzothiazoles, benzyl bromides, and 2-mercaptobenzoxazole in acetone under catalyst-free conditions in the presence of triethylamine. The structures of the products are confirmed by nuclear magnetic resonance spectroscopy, infrared spectroscopy, electron ionization mass spectrometry, and elemental analyses.
Introduction
Multi-component reactions (MCRs) are economically and environmentally beneficial to industry and have attracted much attention. These reactions are perfectly applicable to organic synthesis and agrochemistry.1,2 Benzoxazoles are an important class of heterocyclic compound consisting of a benzene-fused oxazole ring, and are known to exhibit remarkable biological activities.3,4 Benzothiazole is a heterocyclic compound containing sulfur and nitrogen atoms and possesses high chemical and photophysical stability compared to other heteroaromatic molecules, and it is sometimes used as an electron acceptor due to its strong electron-withdrawing capability.
5
Benzothiazoles and their analogues are important building blocks and synthetic intermediates in the synthesis of new drugs, pesticides, and dyes, and one utilized medicinal in chemistry.6,7 Derivatives of benzothiazole have been reported to show a broad range of biological and agrochemical applications like antitubercular,
7
antifolate,
8
antihelmintic,
9
anti-inflammatory,
10
antioxidant,
11
and antileishmanial.
12
However, benzothiazoles are also widely used in a variety of consumer and industrial products such as in nonlinear optics (NLOs), organic light-emitting diodes (OLEDs), fluorescent probes, luminescence, chemosensors,13–15 and liquid crystals.
16
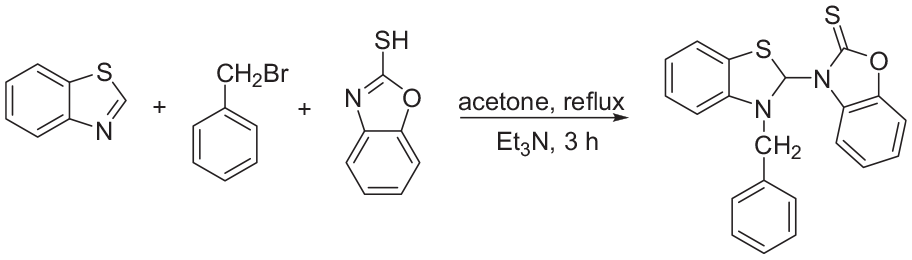
In this paper, the results of studies involving simple, novel, and catalyst-free reactions of benzothiazoles (
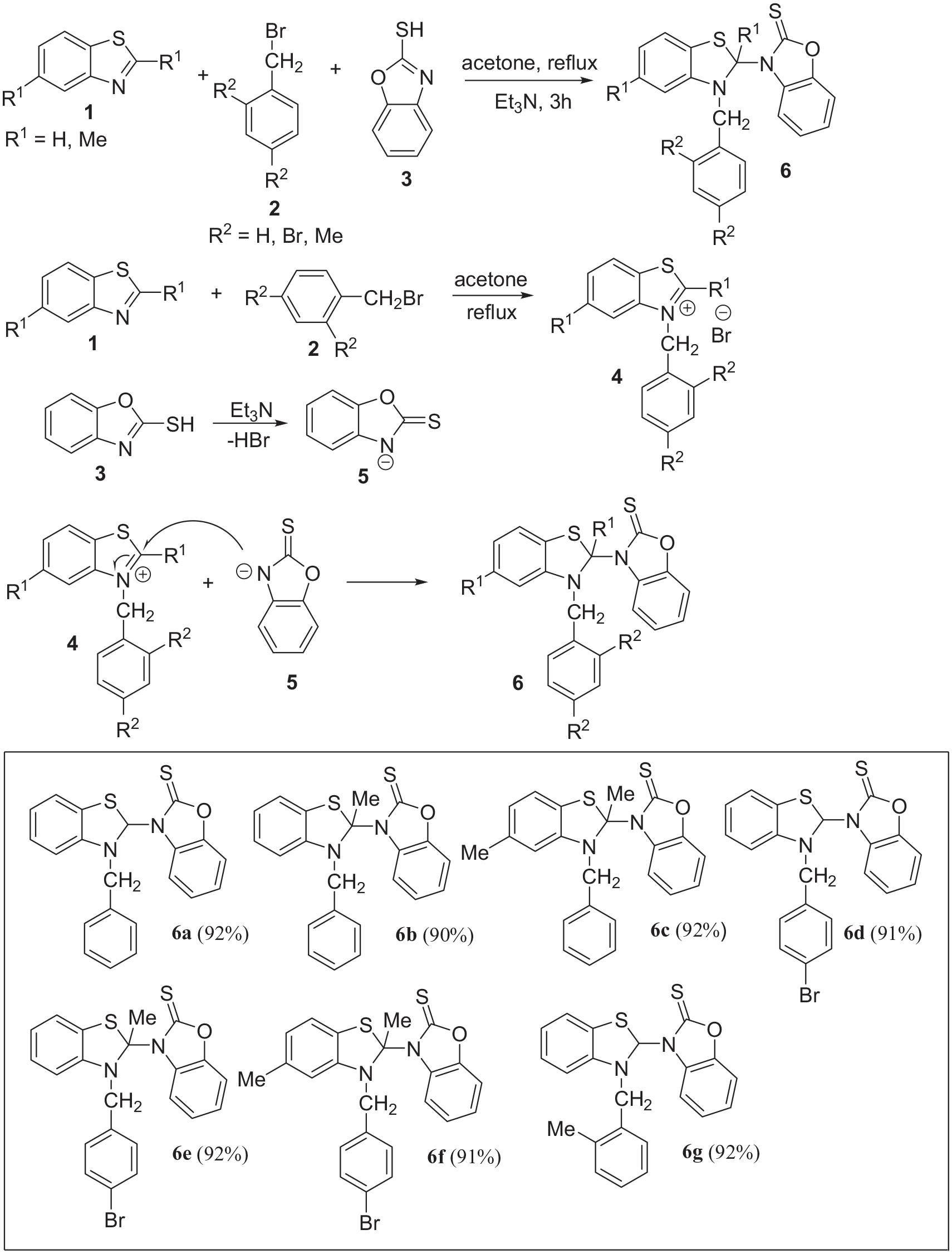
General procedure and the structures of compounds
Results and discussion
In continuation of our studies on the application of both benzothiazole and 2-mercaptobenzoxazole in MCRs,17–19 a new, easy, and efficient route for the synthesis of 3-(3-benzyl-2,3-dihydrobenzo[d]thiazol-2-yl)benzo[d]oxazole-2(3H)-thione derivatives
The 1H and 13C nuclear magnetic resonance (NMR) spectra of the crude products clearly indicated the formation of compounds
Conclusion
In this study, the target compounds containing both benzothiazole and benzoxazole moieties have been synthesized with excellent yields via a convenient and efficient method under catalyst-free conditions at reflux for 3 h. The method utilizes easily available starting materials in acetone as the solvent, and the work-up procedure is simple.
Experimental
Compounds
Preparation of compound (6a ); typical procedure
A mixture of benzothiazole (
3-(3-Benzyl-2,3-dihydrobenzo[d]thiazol-2-yl)benzo[d]oxazole-2(3H)-thione (
3-(3-Benzyl-2,3-dihydro-2-methylbenzo[d]thiazol-2-yl)benzo[d]oxazole-2(3H)-thione (
3-(3-Benzyl-2,3-dihydro-2,5-dimethylbenzo[d]thiazol-2-yl)benzo[d]oxazole-2(3H)-thione (
3-(3-(4-Bromobenzyl)-2,3-dihydrobenzo[d]thiazol-2-yl)benzo[d]oxazole-2(3H)-thione (
3-(3-(4-Bromobenzyl)-2,3-dihydro-2-methylbenzo[d]thiazol-2-yl)benzo[d]oxazole-2(3H)-thione (
3-(3-(4-Bromobenzyl)-2,3-dihydro-2,5-dimethylbenzo[d]thiazol-2-yl)benzo[d]oxazole-2(3H)-thione (
3-(3-(2-Methylbenzyl)-2,3-dihydrobenzo[d]thiazol-2-yl)benzo[d]oxazole-2(3H)-thione (
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: I gratefully acknowledge the financial support received from the Research Council of Chabahar Maritime University.
