Abstract
Substitution reactions of 1,2-difluoro-4,5-dinitrobenzene were explored in dimethylformamide alone and with KOH/H2O, Hünig’s base or Et3N and with Me2NHCl/Et3N in EtOH. The fluorine atoms were always displaced in preference to the nitro groups. Three compounds were prepared from these studies and were characterised by X-ray single crystal structure determinations.
Keywords
Introduction
The reaction of aniline with 2,4-difluoronitrobenzene


Some commercially available nitrated fluorobenzenes.
Discussion
4,5-Difluoro-1,2-dinitrobenzene

Test 1: No yellow colour forms from compound

Test 2: Yellow colouration forms from compound

Base catalysed generation of dimethylamine from DMF. DMF: dimethylformamide.

Hünig’s base catalysed reaction of compound
In contrast with Hünig’s base, which is hindered to shield the basic nitrogen atom, Et3N reacts with compound
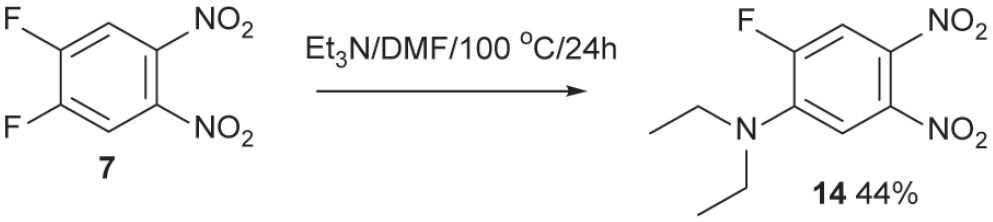
Reaction of triethylamine with compound
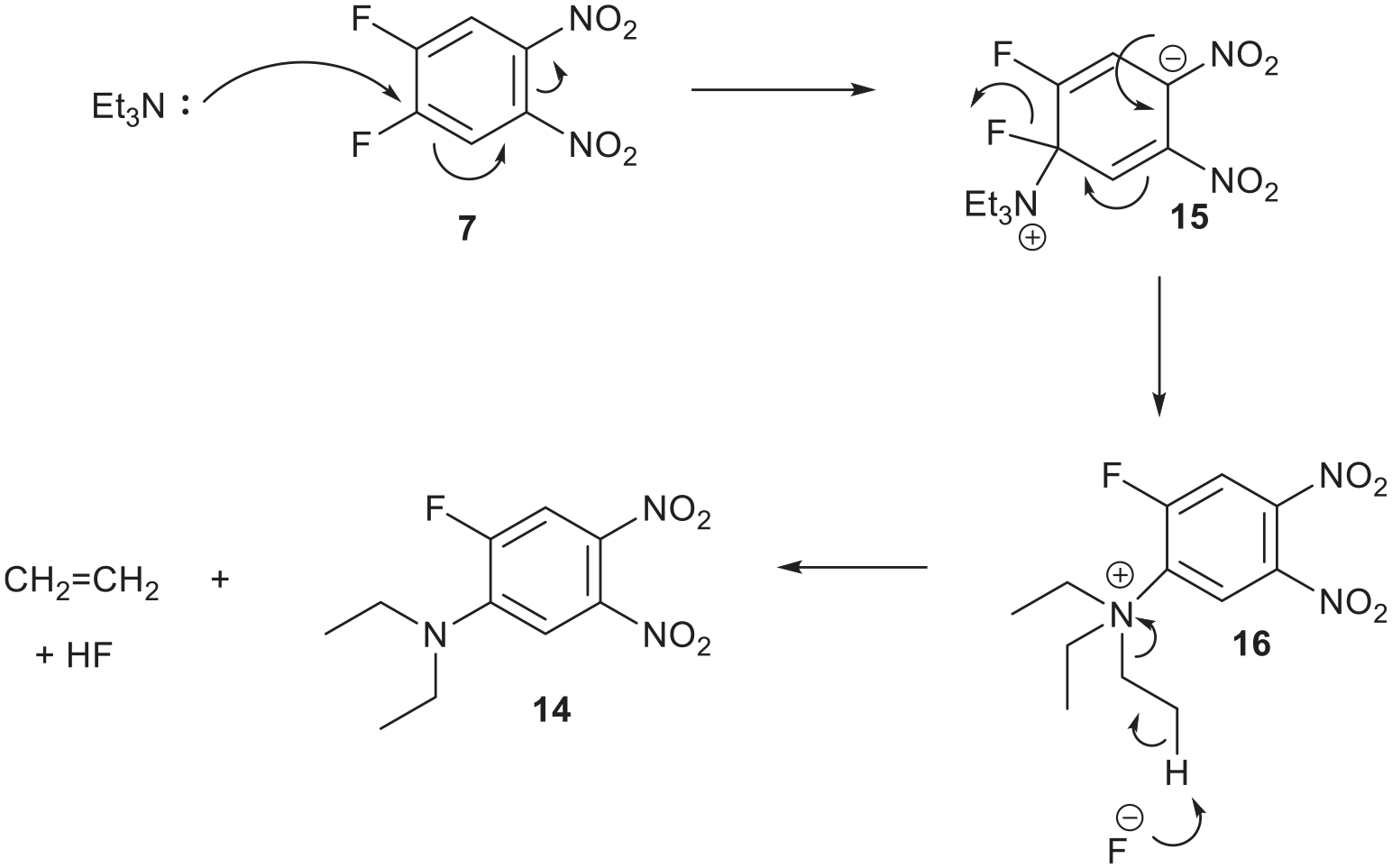
A proposed mechanism for reacting compound
The asymmetric unit of compound

The molecular structure of the C1 molecule in compound
In the extended structure of compound

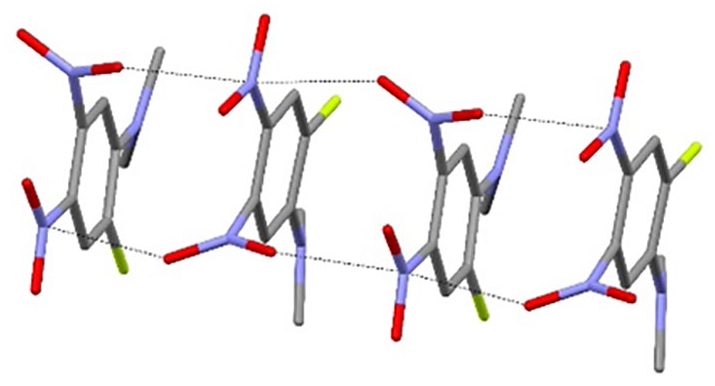
The unit-cell packing for compound
Finally dimethylamine was liberated in situ from dimethylamine hydrochloride in EtOH with Et3N and reacted with compound
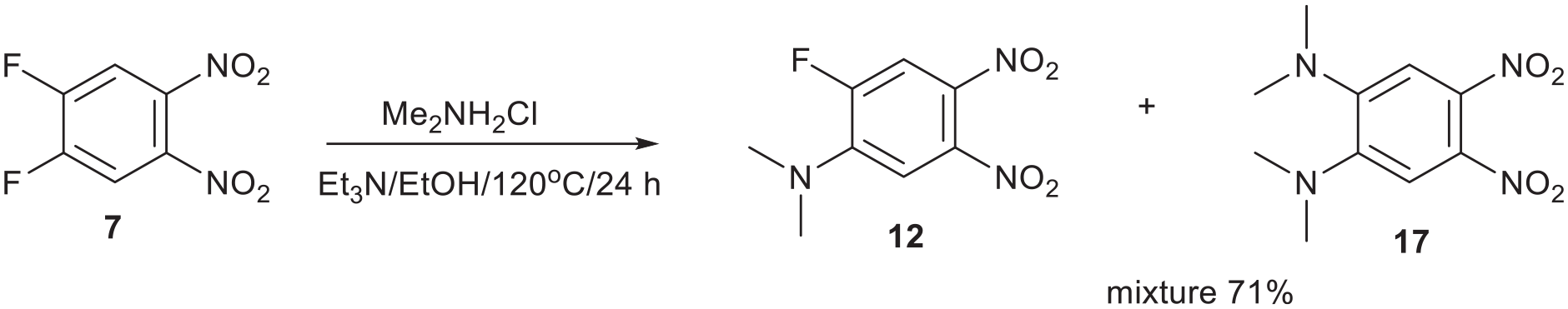
Reaction of dimethylamine with compound
Upon crystallisation, the crystallographer found and separated crystals for both compounds, which were characterised by X-ray single crystal structure determinations.
Compound

The molecular structure of compound
In the extended structure of compound
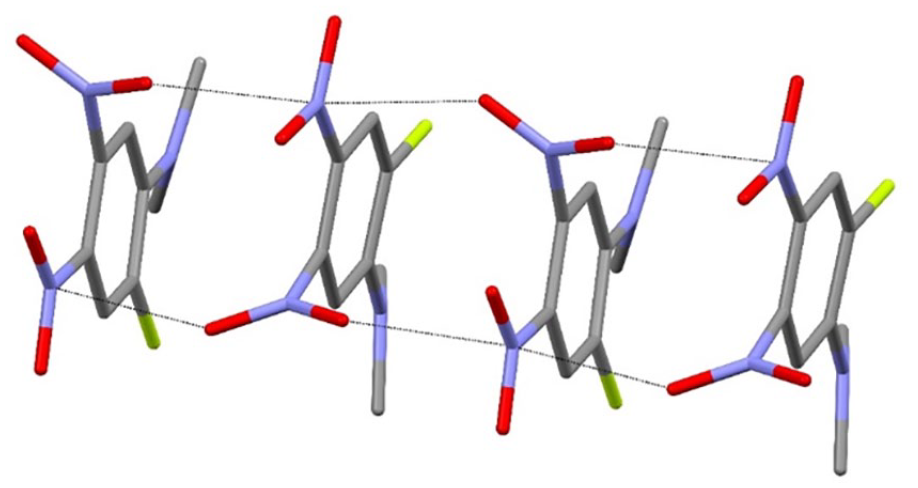
Short N–O···N contacts (black dashed lines) generating [001] chains in the extended structure of compound
Compound

The molecular structure of compound
Experimental
Compounds 12–14 and 17 are new compounds
Infrared (IR) spectra were recorded on a diamond attenuated total reflection (ATR) Fourier transform infrared (FTIR) spectrometer. Ultraviolet (UV) spectra were recorded using a PerkinElmer Lambda 25 UV-VIS spectrometer with EtOH as the solvent. The term sh means shoulder. 1H and 13C NMR spectra were recorded at 400 and 100.5 MHz, respectively, using a Varian 400 spectrometer. Chemical shifts, δ, are given in ppm and measured by comparison with the residual solvent. Coupling constants, J, are given in Hz. High-resolution mass spectra were obtained at the University of Wales, Swansea, using an Atmospheric Solids Analysis Probe (ASAP; Positive mode) Instrument: Xevo G2-S ASAP. Melting points were determined on a Kofler hot-stage microscope.
For Tests 1 and 2 and Methods 1–4, the starting material was not recovered.
Test 1
DMF (5 mL) was treated with one drop of concentrated aqueous HCl and then treated with 1,2-difluoro-4,5-dinitrobenzene (20 mg, 0.098 mmol). The solution remained clear after 15 min. This is a control, which shows that the compound is not coloured in DMF and that it is a base-catalysed reaction, which turns the solution yellow.
Test 2
1,2-Difluoro-4,5-dinitrobenzene (20 mg, mmol) was dissolved in DMF (5 mL) and left standing for 10 min. The solution turned pale yellow. The solution was diluted with water (250 mL), then extracted with dichloromethane (DCM; 50 mL). The pale yellow DCM layer was dried with MgSO4 and decanted. After concentration a TLC plate eluted with 50:50 (DCM: light petrol) showed the single yellow product to have an identical Rf value to 2-(dimethylamino)-4,5-dinitrofluorobenzene
Method 1. 2-(Dimethylamino)-4,5-dinitrofluorobenzene 12 and 2-hydroxy-4,5-dinitrofluorobenzene 13
1,2-Difluoro-4,5-dinitrobenzene
Method 2. 2-(Dimethylamino)-4,5-dinitrofluorobenzene 12
1,2-Difluoro-4,5-dinitrobenzene
Method 3. 2-(Diethylamino)-4,5-dinitrofluorobenzene 14
1,2-Difluoro-4,5-dinitrobenzene
Method 4. Mixture of 2-(dimethylamino)-4,5-dinitrofluorobenzene 12 and 1,2-bis(dimethylamino)-4,5-dinitrobenzene 17
1,2-Difluoro-4,5-dinitrobenzene
Single-crystal diffraction
Yellow and red crystals were separated by the crystallographer. The crystal structure of compound
Crystal data for compound
Crystal data for compound
Crystal data for compound
Conclusion
4,5-Difluoro-1,2-dinitrobenzene
Summary of the reaction data.

Entry 4 was sealed in a Parr vessel and the products were not separated, but the mixture
These results show that DMF can be used in place of EtOH for nucleophilic substitution reactions of activated fluoride but some minor side reactions are introduced which may affect product purity. These studies are helpful for our ongoing work on an iterative approach to NOS-heteroacenes using 4,5-difluoro-1,2-dinitrobenzene
Supplemental Material
sj-pdf-1-chl-10.1177_17475198231154812 – Supplemental material for Reactions of 4,5-difluoro-1,2-dinitrobenzene with amines in dimethylformamide or EtOH
Supplemental material, sj-pdf-1-chl-10.1177_17475198231154812 for Reactions of 4,5-difluoro-1,2-dinitrobenzene with amines in dimethylformamide or EtOH by M John Plater and William TA Harrison in Journal of Chemical Research
Footnotes
Acknowledgements
We thank the UK EPSRC National Mass Spectrometry Service Centre for mass spectrometric data and the UK National Crystallography Centre (University of Southampton) for the X-ray data collections. MJ Plater performed all synthesis and obtained the characterisation data and WTA Harrison solved the crystallographic data sets. Data sets were obtained free of charge from the National Crystallography Centre, Southampton University.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
