Abstract
The 3C-like protease (also known as Mpro) plays a key role in SARS-CoV-2 replication and has similar substrates across mutant coronaviruses, making it an ideal drug target. We synthesized 19 thiazolidinedione derivatives via the Knoevenagel condensations and Mitsunobu reactions as potential 3C-like protease inhibitors. The activity of these inhibitors is screened in vitro by employing the enzymatic screening model of 3C-like protease using fluorescence resonance energy transfer. Dithiothreitol is included in the enzymatic reaction system to avoid non-specific enzymatic inhibition. Active inhibitors with diverse activity are found in this series of compounds, and two representative inhibitors with potent inhibitory activity are highlighted.
New thiazolidinedione derivatives were designed and synthesized. Two of these compounds were proved to have potent inhibitory activity against SARS-CoV-2 Mpro.

Introduction
At the end of 2019, COVID-19, caused by SARS-CoV-2, led to a global health crisis, with more than six hundred million people cumulatively diagnosed with SARS-CoV-2 and more than six and a half million deaths according to the World Health Organization (WHO). SARS-CoV-2 is an enveloped, positive single-stranded RNA virus belonging to the subgenus Sarbecovirus of the genus Betacoronavirus, 1 the genome of which consists of about 30,000 nucleotides. 2 SARS-CoV-2 has high homology with SARS-CoV and Middle East respiratory syndrome coronavirus (MERS-CoV). After the infection of host cells by this virus, the ORF1a/b of SARS-CoV-2 is first translated and expressed with two polyprotein precursors (pp1a and pp1ab) with the help of the host cell. 3 The polyprotein precursors undergo intramolecular cleavage by the action of the main protease (Mpro for short). Mpro, a papain-like protease, is also known as a 3C-like protease (3CLpro), 4 because its cleavage site specificity is similar to that of the 3C protease of picornavirus, and both are referred to as Mpro/3CLpro. The non-structural proteins produced by the polyprotein precursors are involved in the production of viral subgenomic RNA and four structural proteins (envelope/E, membrane/M, spike/S, and nucleocapsid/N), which, in turn, are responsible for the reproduction and release of the offspring virus. Since the Mpro protease plays a crucial role in the virus's life cycle and there is no homologous protein in the human body, the Mpro master protease is an ideal target for developing antiviral drugs. 5
Since the SARS outbreak in 2003, several anti-SARS drugs based on the Mpro structure have been investigated. Anti-HIV drugs such as lopinavir and ritonavir also act on similar protease targets and have been well-validated in clinical practice. 6 Meanwhile, the screened Mpro inhibitors have a certain degree of broad-spectrum anti-coronavirus ability and may even be used for the treatment of other animal diseases caused by coronaviruses (porcine coronavirus, for example), because Mpro is highly conserved in beta coronavirus. 7 SARS-CoV-2 Mpro/3CLpro differs from SARS-CoV Mpro/3CLpro by only 12 amino acids, and the homology is greater than 96%. 8 Also according to the Global Initiative on Sharing Avian Influenza Data (GISAID), SARS-CoV-2 Mpro is highly conserved. The mutation rate of its binding domain is less than 0.001. 9 Therefore, mutations do not broadly affect the efficacy of SARS-CoV-2 Mpro inhibitors. As its key roles are in mediating viral replication and transcription, Mpro is viewed as an ideal drug target for viruses. 10
Jin et al. 11 screened about 10,000 compounds using fluorescence resonance energy transfer (FRET) analysis and selected seven compounds with IC50 values in the range of 0.67–21.4 µM, including tideglusib and TDZD-8 (Figure 1). Tideglusib and TDZD-8 are novel 3CL protease inhibitors possessing a thiadiazolidinedione structure. However, recent research results suggest that the thiadiazolidinedione structure of tideglusib suffers from instability. Inhibition of 3CL protease activity by tideglusib is significantly reduced when dithiothreitol (DTT) is added to the enzymatic assay of cysteine protease. The results of this experiment show that tideglusib is sensitive to DTT and is a non-specific inhibitor of SARS-CoV-2 Mpro. 12 We hypothesize that the sulfur-nitrogen bond in the structure of the thiadiazolidinedione is a high-energy bond. The addition of DTT breaks the sulfur-nitrogen bond leading to ring opening. Thus, tideglusib has significant issues as a drug.
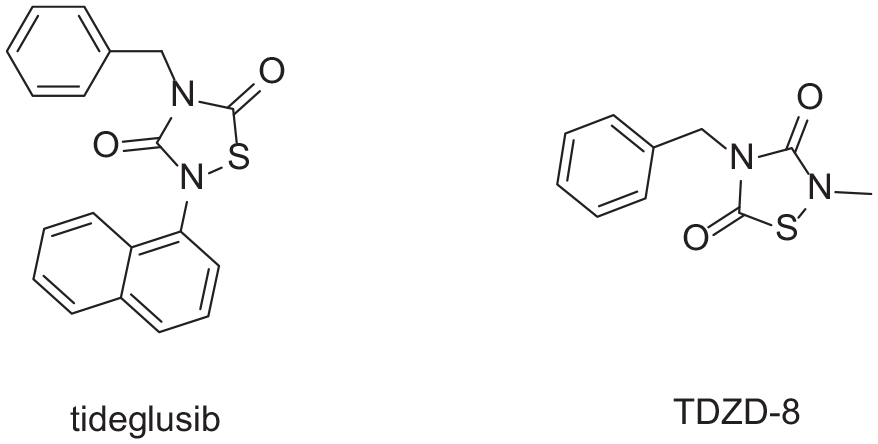
Novel 3CL protease inhibitors possessing a thiadiazolidinedione structure.
Computer simulations show that when tideglusib is bound to the substrate protease, the two carbonyl groups of the thiadiazolidinedione structure form hydrogen bonds with the protease. Two side chains occupy the pocket to dock COVID-19 virus Mpro. 11 Thus, we designed their bioisostere structures. Thiazolidinediones also have two carbonyl groups and two side chains, and one of the nitrogen atoms has been replaced with a methylene group to improve structural stability. Thiazolidines can be used as dual inhibitors of aldose reductase and protein tyrosine phosphatase 1B, which are potential agents for the treatment of type 2 diabetes mellitus and its complications, 13 as tyrosinase inhibitors. 14 It also has an inhibitory effect on RNA viruses, 15 so it was hypothesized that it might have an inhibitory effect on 3CL protease.
The enzyme activity was measured by inhibition assays using FRET (Figure 2). The MCA-AVLQSGFR-Lys(Dnp)-Lys-NH2 substrate peptide was derived from residues P4-P5’ of the SARS-CoV-2 Mpro N-terminal autoprocessing site with the AVLQSGFRK sequence. The enzymatic cleavage between Q4 and S5 of AVLQSGFR-Lys(Dnp)-Lys-NH2 releases a highly fluorescent 7-methoxycoumarin (MCA) moiety that is affected by the internal bursting agent of 2,4-dinitrophenol (DNP), resulting in a large increase in fluorescence intensity. The substrate is cleaved by 3CL/Mpro to produce MCA fluorescent group, which has a maximum excitation at 320 nm and a maximum emission wavelength of 405 nm. The activity of 3CL Mpro is detected by measuring the fluorescence intensity. A potent 3CL Mpro inhibitor, GC376 serves as a positive control for the experiments. 16

Principle of FRET.
Results and discussion
The intermediates
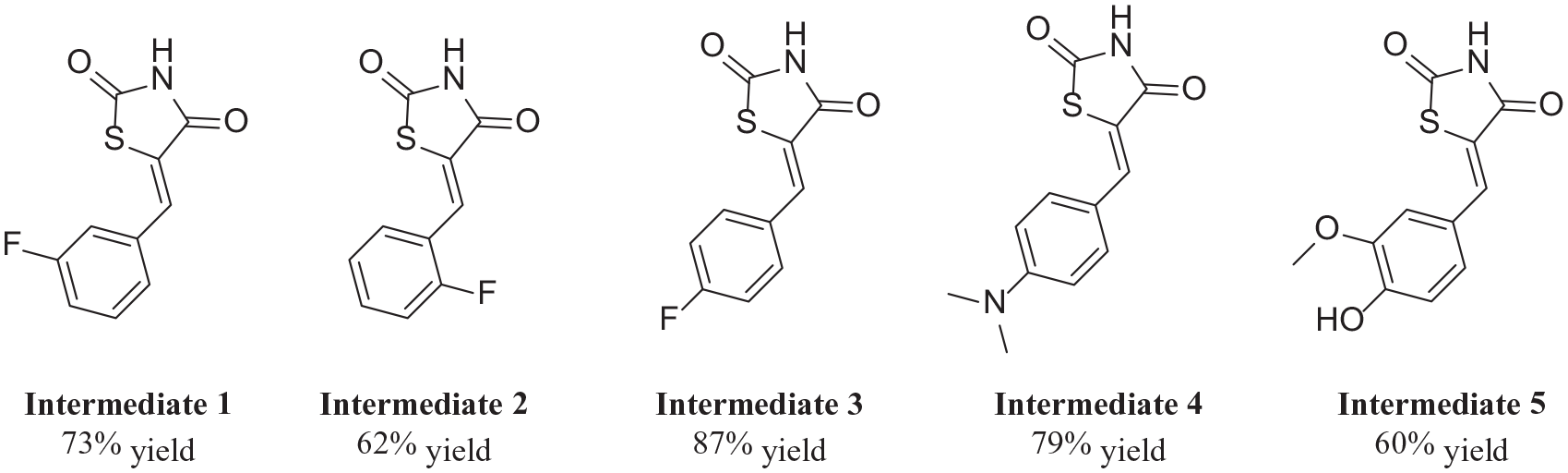
Structures of intermediates

Synthesis of intermediates

Synthesis of
Compounds
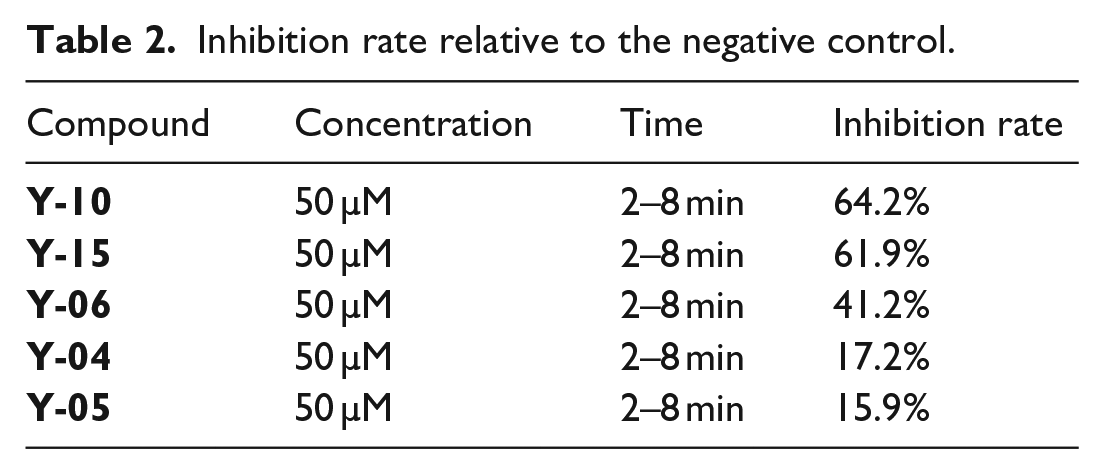
In vitro tests of the inhibition activity were carried out on the 19 compounds synthesized (final concentration of the reaction system 50 µM) and the positive control GC376 (final concentration of the reaction system 0.2 μM). Of these, 64.2% inhibition was achieved for
Inhibition rate relative to the negative control.
The IC50 values of the selected compounds inhibiting SARS-CoV-2 3CL Mpro were calculated and plotted. Reference group GC376 has an IC50 value of 60.04 nM. Compound

IC50 values of compounds
Conclusion
All the 1,2,4-thiazolidine-3,5-dione derivatives synthesized demonstrated inhibitory effects against recombinant 3CL Mpro. With the addition of DTT,
Experimental
Materials and reagents
All reagents used were analytically pure reagents from commercially available companies such as Lakin, Adamas, and Titan, and reactions were monitored by thin-layer chromatography (silica gel HSGF254 from Yantai Jiangyou Silicone Development Co., Ltd., China). Spots were observed by ultraviolet (UV) irradiation (254 nm), and compounds were purified by silica gel column chromatography (100–200 mesh). Melting points were determined on a melting point determination apparatus RY-1 (Tianjin, China). 1H NMR, 13C NMR, and 19F NMR spectra were recorded using a Bruker 600 MHz NMR spectrometer with DMSO-d6 (D = 99.9% + TMS = 0.03%) as the solvent. Mass spectra were obtained on an Agilent 6120 Ion Trap LC/MS 500 analysis system (Santa Clara, CA, USA). Elemental analyses were obtained on an Elementar Vario EL III elemental analyzer (Langenselbold, Germany).
MCA-AVLQSGFR-Lys (Dnp)-Lys-NH2 and SARS-CoV-2 Main Protease Mpro were obtained from Beyotime Biotechnology Inc. (China). GC376 was purchased from TargetMol (Wellesley Hills, MA, USA). Costar® 96-well assay plates (Cat no. 3925) was purchased from Coring Inc. (Corning, NY, USA).
Enzymatic activity assays
Briefly, the 3CL Mpro of 30 nM was mixed with serial dilutions of each compound in reaction buffer (50 mM Tris-HCl, pH 7.3, 1 mM ethylenediaminetetraacetic acid (EDTA), 0.01% TritonX-100; 1 mM DTT), and incubated at 37 °C for 10 min. Next, a final concentration of 20 μM fluorogenic substrate MCA-AVLQSGFR-Lys(Dnp)-Lys-NH2 was added. The fluorescence intensity was immediately measured at the wavelengths of 320 nm (excitation) and 405 nm (emission) every 3 min for 30 min using a TECAN infinite 200PRO plate reader (Männedorf, Switzerland). GC376 (100 nM) was used as a positive control.
The compounds with potent inhibition observed in preliminary inhibition assays were selected for further IC50 assays. The IC50 values against 3CL Mpro were measured with 9–11 concentration sets, and three independent experiments were performed. Data were analyzed with GraphPad Prism software.
Intermediates 1–5 general procedure
To a solution of 2,4-thiazolidinedione (20 mmol) and 3-fluorobenzaldehyde (20 mmol) in anhydrous ethanol (20 mL), added dropwise piperidine (4 mL) and glacial acetic acid (2 mL). The resulting mixture was heated to 110 °C for 3 h and stirred overnight at 90 °C. After cooling to room temperature, the reaction mixture was poured into ice water (100 mL), and a large amount of yellow solid precipitated. The crude product was filtered off, recrystallized from ethanol, and dried in vacuo to afford intermediate
(Z)-5-(3-Fluorobenzylidene)thiazolidine-2,4-dione (intermediate
(Z)-5-(2-Fluorobenzylidene)thiazolidine-2,4-dione (intermediate
(Z)-5-(4-Fluorobenzylidene)thiazolidine-2,4-dione (intermediate
(Z)-5-(4-(Dimethylamino)benzylidene)thiazolidine-2,4-dione (intermediate
(Z)-5-(4-Hydroxy-3-methoxybenzylidene)thiazolidine-2,4-dione (intermediate
Compounds Y-01–Y-06
To a stirred mixture of intermediate
(Z)-5-(3-Fluorobenzylidene)-3-(2-phenoxyethyl)thiazolidine-2,4-dione (Y-01): Light yellow solid, 76% yield. m.p. 102 °C. 1H NMR (600 MHz, DMSO-d6): δ 7.95 (s, 1H), 7.59 (dd, J = 14.2, 8.0 Hz, 1H), 7.48 (d, J = 10.0 Hz, 1H), 7.45 (d, J = 7.9 Hz, 1H), 7.34 (td, J = 8.4, 2.3 Hz, 1H), 7.27 (dd, J = 8.6, 7.4 Hz, 2H), 6.93 (t, J = 7.3 Hz, 1H), 6.90 (d, J = 7.8 Hz, 2H), 4.21 (t, J = 5.7 Hz, 2H), 4.04 (t, J = 5.7 Hz, 2H). 13C NMR (151 MHz, DMSO-d6): δ 167.4 (s), 165.9 (s), 162.7 (d, J = 245.0 Hz), 158.4 (s), 135.7 (d, J = 8.2 Hz), 132.2 (d, J = 2.6 Hz), 131.9 (d, J = 8.5 Hz), 130.0 (s), 126.1 (d, J = 2.7 Hz), 123.2 (s), 121.4 (s), 117.9 (d, J = 21.2 Hz), 117.4 (d, J = 22.5 Hz), 115.0 (s), 64.1 (s), 41.4 (s). 19F NMR (565 MHz, DMSO-d6): δ −111.7 (s). ESIMS: m/z (relative intensity) 366.0 [M + Na]+ (100). Anal. calcd for C18H14FNO3S: C, 62.96%; H, 4.11%; N, 4.08%; found: C, 63.16%; H, 4.09%; N, 3.98%.
(Z)-5-(3-Fluorobenzylidene)-3-(2-(pyridin-2-yl)ethyl)thiazolidine-2,4-dione (
(Z)-5-(3-Fluorobenzylidene)-3-(pyridin-3-ylmethyl)thiazolidine-2,4-dione (
(Z)-5-(3-Fluorobenzylidene)-3-(2-methoxyethyl)thiazolidine-2,4-dione (
(Z)-3-(2-Ethoxyethyl)-5-(3-fluorobenzylidene)thiazolidine-2,4-dione (
(Z)-3-(2-(2-Ethoxyethoxy)ethyl)-5-(3-fluorobenzylidene)thiazolidine-2,4-dione (
Compounds Y-07–Y-13
To a stirred mixture of /intermediate
(Z)-5-(2-Fluorobenzylidene)-3-(2-phenoxyethyl)thiazolidine-2,4-dione (
(Z)-5-(2-Fluorobenzylidene)-3-(2-(pyridin-2-yl)ethyl)thiazolidine-2,4-dione (
(Z)-5-(2-Fluorobenzylidene)-3-(pyridin-3-ylmethyl)thiazolidine-2,4-dione (
(Z)-5-(2-Fluorobenzylidene)-3-(2-methoxyethyl)thiazolidine-2,4-dione (
(Z)-3-(2-Ethoxyethyl)-5-(2-fluorobenzylidene)thiazolidine-2,4-dione (
(Z)-5-(2-Fluorobenzylidene)-3-(2-morpholinoethyl)thiazolidine-2,4-dione (
(Z)-3-(2-(2-Ethoxyethoxy)ethyl)-5-(2-fluorobenzylidene)thiazolidine-2,4-dione (
Compounds Y-14–Y-16
To a stirred mixture of intermediate
(Z)-5-(4-Fluorobenzylidene)-3-(2-(pyridin-2-yl)ethyl)thiazolidine-2,4-dione (
(Z)-5-(4-Fluorobenzylidene)-3-(2-morpholinoethyl)thiazolidine-2,4-dione (
(Z)-5-(4-Fluorobenzylidene)-3-(2-(pyrrolidin-1-yl)ethyl)thiazolidine-2,4-dione (
Compound Y-17
To a stirred mixture of intermediate
(Z)-5-(4-(Dimethylamino)benzylidene)-3-(2-phenoxyethyl)thiazolidine-2,4-dione (
Compound Y-18
To a mixture of intermediate
(Z)-5-(4-(Dimethylamino)benzylidene)-3-(2-(pyridin-2-yl)ethyl)thiazolidine-2,4-dione (
Compound Y-19
To a stirred mixture of intermediate
(Z)-5-(4-Hydroxy-3-methoxybenzylidene)-3-(2-(pyridin-2-yl)ethyl)thiazolidine-2,4-dione (
Supplemental Material
sj-docx-1-chl-10.1177_17475198231152556 – Supplemental material for Synthesis and enzymatic inhibition effects of thiazolidinedione 3C-like protease inhibitors
Supplemental material, sj-docx-1-chl-10.1177_17475198231152556 for Synthesis and enzymatic inhibition effects of thiazolidinedione 3C-like protease inhibitors by Xin Ye, Yuhua Li, Lina Guo, Yiling Yao, Rouyu Zhu, Shuang Wei, Hua Diao and Zhiyu Shao in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Commission of Shanghai Municipality (grant no. 22S11901000).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
