Abstract
The synthesis of two sulfur-decorated perylene diimides, the five-membered S-heterocyclic annulated perylene diimide (
Keywords

Introduction
Polycyclic aromatic hydrocarbons (PAHs) have attracted considerable interest from researchers in different fields due to their unique optical and electrochemical properties.1–3 The synthesis, properties, and applications of PAHs have become a rapidly developing scientific field. 4 As a category of PAHs, perylene diimides (PDIs) have high fluorescence quantum yields and excellent photochemical and thermal stabilities. 5 Moreover, they are versatile candidates for applications in organic photovoltaics (OPVs), organic field-effect transistors (OFETs), and organic light-emitting diodes (OLEDs).6–9 Thus, establishing efficient routes for the synthesis of PDIs is very important. Most modification strategies can be classified into two categories. The first involves incorporating functional groups on the aromatic backbone of the imide, ortho, or bay positions of the PDIs.10–12 The second is extending the π-systems by the incorporation of heteroatoms (such as B, N, S, Se, O, and Si) into the frameworks of the aromatic rings. 13 Modifying the imide substituents has a vital effects on the electronic structures and properties of PDIs because the imide nitrogen atoms are the nodal points of frontier orbitals.14,15 Attaching various substitutions at the bay position of PDIs should be another promising strategy for modifying the frontier orbital levels. 16
Fully fused bay-modification of PDIs has been demonstrated to enhance their electronic and optoelectronic properties.17,18 Extensions along the short molecular axis of PDIs cause a hypsochromic shift of absorption spectra and induce a variety of intermolecular interactions. These interactions are essential to achieve highly ordered supramolecular self-assembled structures, which result in excellent device performance.19,20 Introducing heteroatoms to a π-conjugated system is also an easy approach for the construction of intramolecular charge-transfer compounds because the lone pairs of electrons of heteroatoms can be used as electron donors. 21
An alkyl side chain is often selected as a soluble group. Its flexibility and small steric hindrance lead to minor effects on intermolecular approach but cause strong π–π stacking of adjacent perylene molecules. Sometimes, alkyl groups even facilitate the formation of ordered structures of perylene molecules through interdigitated actions. 22 Due to the planar molecular geometry, PDIs with flexible linear alkyl substituents usually have high charge carrier mobility but low solid-state luminescence efficiency, which is a prerequisite for efficient optoelectronic and organic electronic devices. 23 As important structural motifs, sulfoxides have attracted increasing attention in recent years due to their unique properties such as central chirality at the sulfur, strong polarized S–O bond, and high configurational stability. In addition, organic sulfoxides have also been successfully used as steering ligands in transition-metal catalysis, new pharmacophores in drug discovery, and as versatile synthetic intermediates in the generation of natural and biologically active compounds.24,25
In our previous work, two bay unilaterally extended PDIs, a five-membered O-heterocyclic annulated perylene diimide (O-PDI), and five-membered S-heterocyclic annulated PDI (

Synthesis of compounds
Results and discussion
Synthesis and characterization
Scheme 1 shows the chemical structures and synthetic routes toward bay unilaterally extended
Optical properties
The optical properties of compounds

Normalized absorption spectra of

Normalized fluorescence spectra of
Summary of the optical absorption and emission properties of
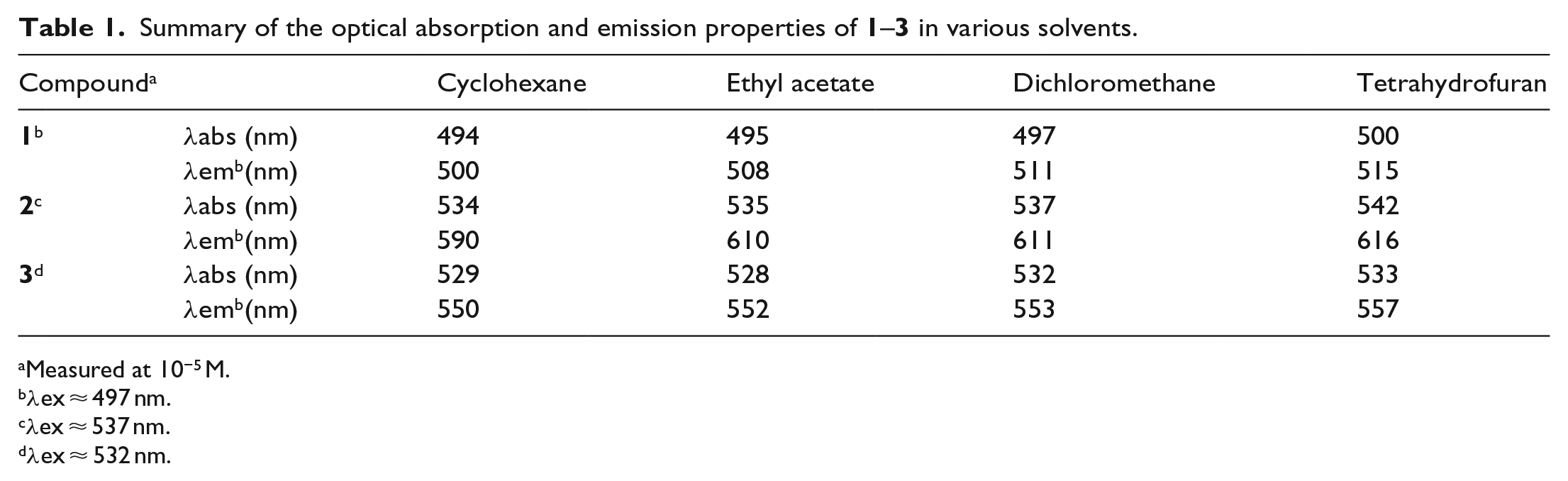
Measured at 10−5 M.
λex ≈ 497 nm.
λex ≈ 537 nm.
λex ≈ 532 nm.
The fluorescence spectra depicted the same structure with a mirror image of the absorption spectra, and the emission peaks appeared at 511 and 542 nm for compounds

Normalized emission spectra of
Electrochemical properties
The cyclic voltammograms of

The cyclic voltammograms of
Electrochemical data of compounds

Measured in a solution of 0.1 M tetrabutylammonium hexafluorophosphate (TBAPF6) in dichloromethane versus Ag/AgNO3 (in Volts).
Calculated from EHOMO = −4.88 − (Eoxd − EFe/Fe+), ELUMO = EHOMO + Egap.
Calculated and experimental parameters for compounds

HOMO: highest occupied molecular orbital; LUMO: lowest unoccupied molecular orbital.
Calculated by density functional theory (DFT)/B3LYP (in eV).
At absorption maxima (Eg = 1240/λmax, in eV).
Quantum chemistry computations
To gain insight into the electronic properties of

Computed frontier orbitals of

Optimized structures of
Powder X-ray diffraction studies
The structures of PDIs

X-ray diffraction patterns of the PDIs
Finally, thermogravimetric analysis (TGA) of
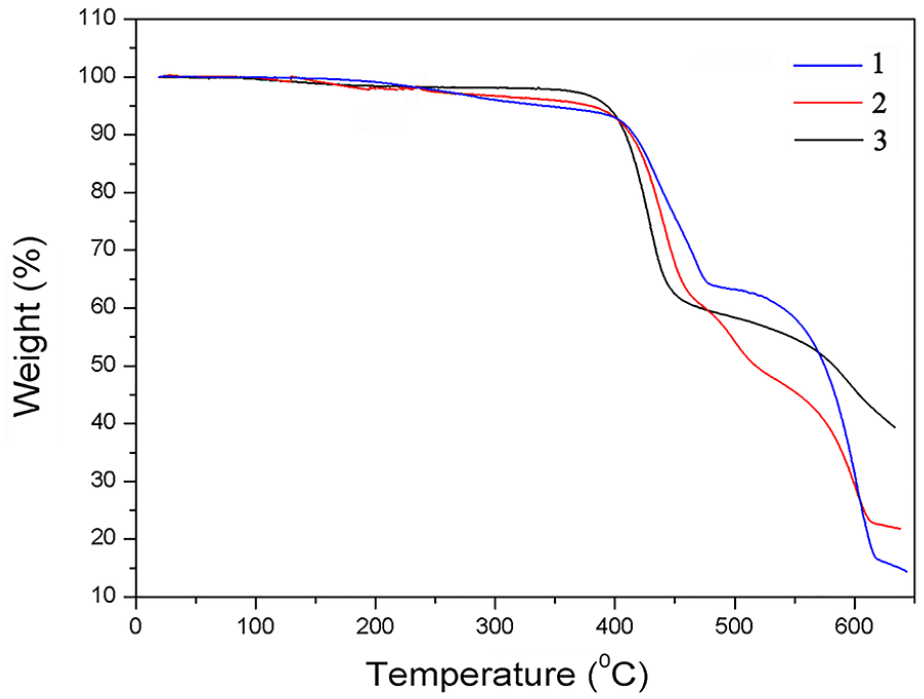
TGA curves of
Conclusion
This study describes the synthesis of three sulfur-decorated PDIs equipped with different substituents and the impact of the structures on their photophysical, electrochemical, aggregation, and thermal properties. The HOMO, LUMO, and energy gaps obtained through DFT calculations are close to the experimental cyclic voltammetry (CV) values. These PDIs possess excellent photooxidative resistance, and demonstrate good solubility, as well as green and red colors in the neutral state derived from absorption in the visible region. Our study may be useful for the development of new PDIs that can be used as organic electronic materials.
Experimental
Materials and equipments
The solvents were purchased from commercial sources and used as received. N,N′-Dicyclohexyl-3,4,9,10-tetracarboxylic acid bisimide (compound
The 1H and 1C NMR spectra were obtained in CDCl3 on a Bruker (Germany) 300 MHz spectrometer. FTIR spectra were measured on a Bruker Tensor-27 spectrophotometer. Mass spectra were measured on a Bruker MaXis UHR-TOF mass spectrometer. The absorption and emission spectra were recorded on a Varian (America) Cary 50 spectrophotometer and a Hitachi (Japan) FL-4500 spectrofluorometer. CV was performed with a CHI760E electrochemical analyzer at a potential rate of 100 mV s−1 in a 0.1 M solution of tetrabutylammonium perchlorate (Bu4NClO4) in dichloromethane. Glassy carbon, platinum, and Ag/AgNO3 electrodes were used as working, counter, and reference electrodes, respectively.
Computation details
Structure optimizations and property calculations employed Becke’s three-parameter gradient-corrected hybrid density function B3LYP method and the standard 6-31G*(d) basis set. 29
Synthesis and characterization
Preparation of compound 1
Compound
Preparation of compound 2
Compound
Preparation of compound 3
Compound
Supplemental Material
Supplementary_Data – Supplemental material for Synthesis and properties of bay unilaterally extended and mono-substituted perylene diimides
Supplemental material, Supplementary_Data for Synthesis and properties of bay unilaterally extended and mono-substituted perylene diimides by Fengxia Zhang, Xianqiang Huang, Xiaofeng Wei, Huixue Ren, Tianyi Jiang, Xuemei Li, Junsen Wu and Yongshan Ma in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Doctoral Foundation of Shandong Jianzhu University (grant nos. XNBS1938 and XNBS1712), the Science and Technology Plan Project of Housing and Urban-Rural Construction Department in Shandong Province (grant nos. 2018-K11-01 and 2018-K7-01), the National Natural Science Foundation of China (grant no. 21871125), the Natural Science Foundation of Shandong Province (grant no. ZR2016EEM01), the National Key R&D Program of China (grant no. 2017YFF0209904), and the Science and Technology Project of MOHURD (grant no. 2015-K7-005).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
