Abstract
7,8,3’,4’,5’-Pentahydroxyisoflavone (8,3’,5’-trihydroxydaidzein) and 6,7,8,3’,4’,5’-hexahydroxyisoflavone (6,8,3’,5’-tetrahydroxydaidzein) are synthesized via a simple and effective method using commercially available daidzein as a raw material. Their structures are confirmed using spectroscopic analyses (infrared, nuclear magnetic resonance, and mass spectrometry), and their purities are determined by high-performance liquid chromatography. The antioxidant capacities are investigated by utilizing 1,1-diphenyl-2-picryl-hydrazyl radical scavenging, superoxide radical scavenging, nitric oxide radical scavenging, reducing power, and phosphomolybdenum assays. The antihypoxia activity is examined using a hypoxia-induced PC12 cell injury model. The antioxidant and antihypoxia abilities of 8,3’,5’-trihydroxydaidzein and 6,8,3’,5’-tetrahydroxydaidzein are greatly improved in comparison with daidzein. These results indicate that 8,3’,5’-trihydroxydaidzein and 6,8,3’,5’-tetrahydroxydaidzein are excellent antioxidant agents and can be used for alleviating injury induced by hypoxia.
Keywords

Introduction
Daidzein (4’,7-dihydroxyisoflavone), Figure 1, commonly called soy isoflavone, is a naturally occurring isoflavone found in large amounts in soybeans and soy-based products. It is reported to have a broad spectrum of pharmacological activities, including anti-inflammatory, 1 antiviral, 2 antidiabetic, 3 cardio-, and neuroprotective.4,5 These beneficial effects can be partly attributed to its antioxidant activities. It is known that the antioxidant ability of flavonoids is due to the phenolic hydroxy, so the number and positions of phenolic hydroxys are directly related to their antioxidant activities. Therefore, introducing more phenolic hydroxy groups to daidzein was expected to increase the antioxidant activity and alter the biological activity. Some hydroxydaidzein derivatives—including 7,3’,4’-, 6,7,4’- and 7,8,4’-trihydroxyisoflavones and 7,8,3’,4’- and 6,7,8,4’-tetrahydroxyisoflavones—have attracted significant attention due to their higher antioxidant, 6 anti-inflammatory, 7 antitumor, 8 and antibacterial 9 activities compared with those of daidzein. As shown in Figure 1, 7,8,3’,4’,5’-pentahydroxyisoflavone (8,3’,5’-trihydroxydaidzein, 8,3’,5’-THD) and 6,7,8,3’,4’,5’-hexahydroxyisoflavone (6,8,3’,5’-tetrahydroxydaidzein, 6,8,3’,5’-FHD) are two hydroxylated daidzein derivatives with five and six phenolic hydroxy groups, respectively, in their structures. In addition, they possess ortho-dihydroxy (catechol) and/or ortho-trihydroxy (pyrogallol) structures, which have a positive effect on enhancing their antioxidative potential. Theoretically, both compounds should have excellent antioxidant activity. However, they have not been isolated from natural sources or obtained by chemical synthesis or microbial biotransformation. Therefore, it is necessary to develop a practical and economical synthetic pathway toward these compounds. The aim of this study was to provide a simple and effective synthetic method for 8,3’,5’-THD and 6,8,3’,5’-FHD. Their antioxidant and antihypoxia activities were also evaluated in this study.
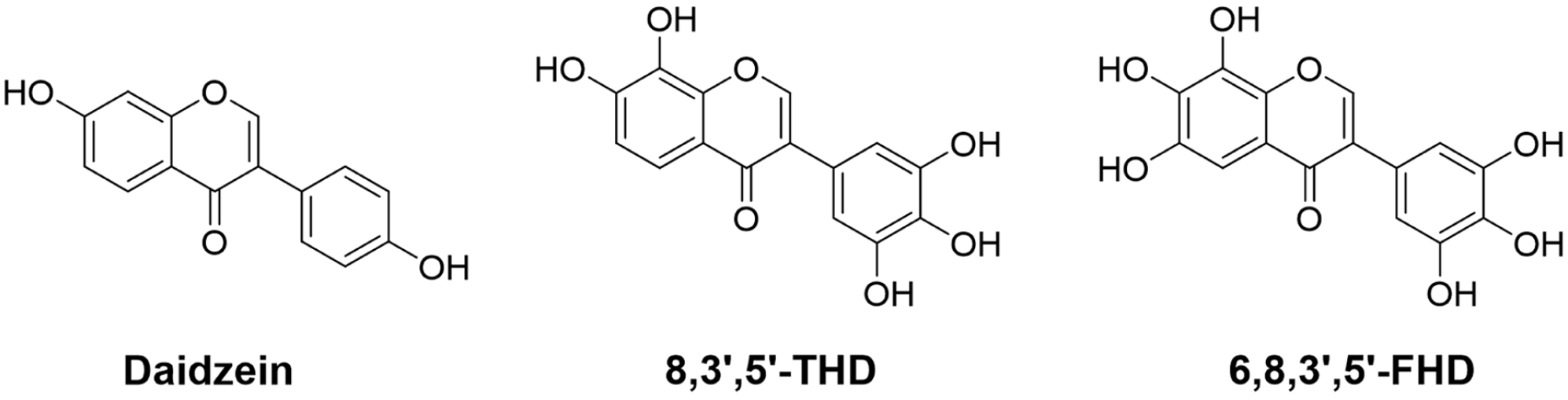
Structures of daidzein, 7,8,3’,4’,5’-pentahydroxyisoflavone (8,3’,5’-THD) and 6,7,8,3’,4’,5’-hexahydroxyisoflavone (6,8,3’,5’-FHD).
Results and discussion
Syntheses of 8,3’,5’-THD and 6,8,3’,5’-FHD
In this study,
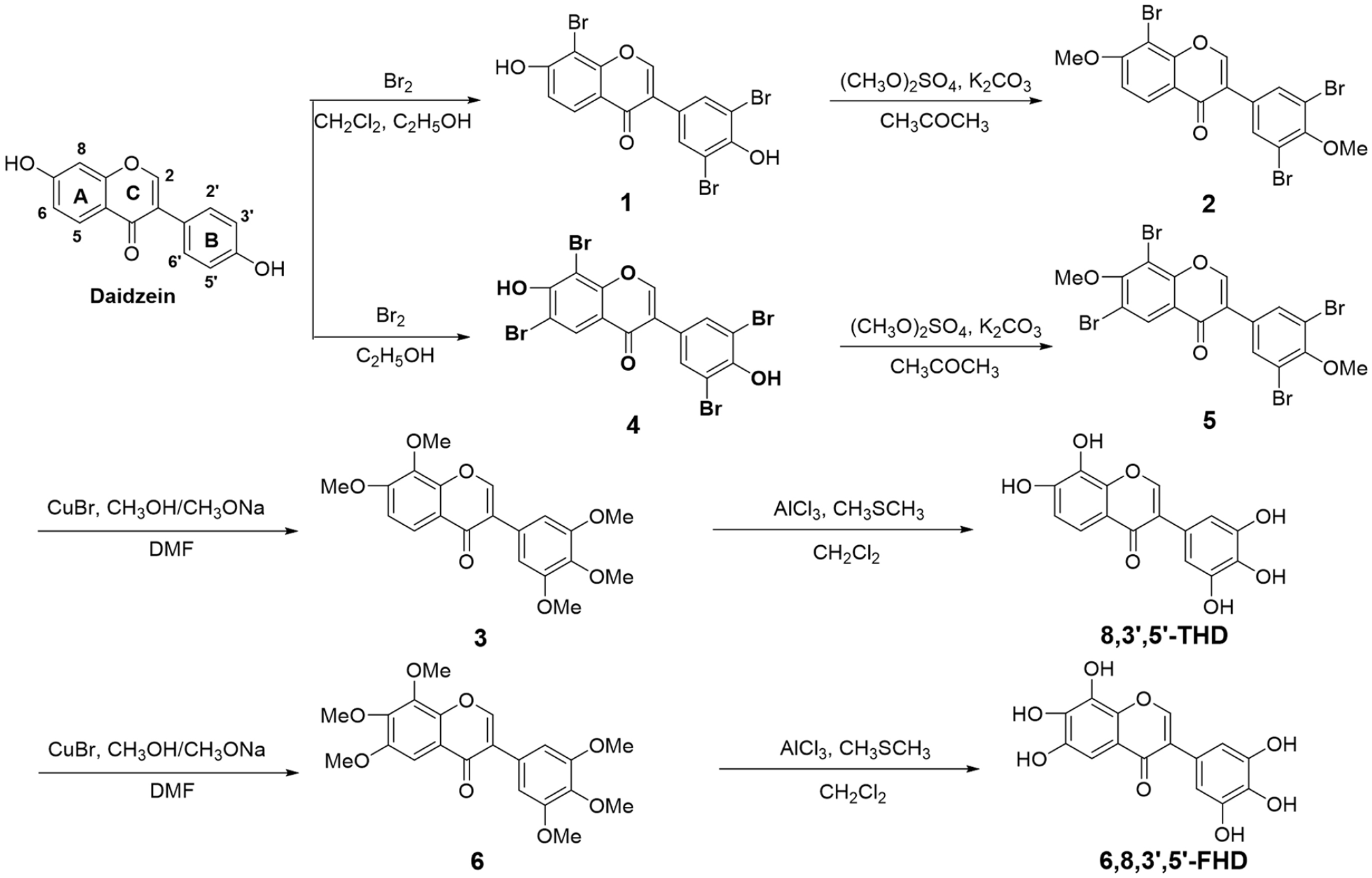
Synthetic route to 8,3’,5’-THD and 6,8,3’,5’-FHD.
The purity of 8,3’,5’-THD and 6,8,3’,5’-FHD
High-performance liquid chromatography (HPLC) chromatograms of 8,3’,5’-THD and 6,8,3’,5’-FHD are shown in Figure 2, with the retention times being 2.86 and 4.25 min, respectively, indicating that they were pure products.

Ultra-high-performance liquid chromatography spectra of 8,3’,5’-THD (a) and 6,8,3’,5’-FHD (b).
Antioxidant activities of 8,3’,5’-THD and 6,8,3’,5’-FHD
Considering that flavonoids have various antioxidant mechanisms, five different in vitro antioxidant assays were performed to fully elucidate the antioxidant activities of 8,3’,5’-THD and 6,8,3’,5’-FHD.
The 1,1-diphenyl-2-picryl-hydrazyl (DPPH) free-radical scavenging assay has been widely used for measuring the ability of compounds to act as free-radical scavengers or hydrogen donors. 12 As shown in Figure 3(a), daidzein exhibited very weak DPPH radical scavenging activity. 6,8,3’,5’-FHD presented the best DPPH radical scavenging activity with an IC50 value of 0.785 ± 0.070 mmol L−1, followed by 8,3’,5’-THD (IC50 = 0.816 ± 0.011 mmol L−1). The IC50 value of the reference compound ascorbic acid (VC) was 1.667 ± 0.009 mmol L−1, indicating that 6,8,3’,5’-FHD and 8,3’,5’-THD exhibited significantly higher scavenging activity toward DPPH compared with that of VC.

Antioxidant activities of 8,3’,5’-THD, 6,8,3’,5’-FHD, and daidzein: (a) DPPH radical scavenging, (b) O2−• radical scavenging, and (c) NO radical scavenging. Vitamin (VC) is used as the reference compound. Data are expressed as the mean value ± SD (n = 3).
Superoxide (O2−•) is the primary reactive oxygen species (ROS) in cells and is also responsible for initiating some highly aggressive ROS, such as hydrogen peroxide (H2O2) and hydroxyl radicals (·OH), that cause oxidative stress and result in cell damage. 13 As shown in Figure 3(b), compared with daidzein (IC50 >2 mmol L−1) and VC (0.487 ± 0.009 mmol L−1), 8,3’,5’-THD and 6,8,3’,5’-FHD exhibited higher O2−• scavenging activity with IC50 values of 0.022 ± 0.001 mmol L−1 and 0.034 ± 0.001 mmol L−1, respectively. Thus, 8,3’,5’-THD and 6,8,3’,5’-FHD present excellent O2−• scavenging activity, even at low concentration.
Nitric oxide (NO) is an important oxidative biological signaling molecule that participates in diverse physiological processes. Nevertheless, high levels of NO can also lead to cell damage. 14 As shown in Figure 3(c), 8,3’,5’-THD and 6,8,3’,5’-FHD exhibited high NO scavenging ratios of 39.66% and 32.93%, respectively, at a low concentration of 0.06125 mmol L−1. While the scavenging ratios of daidzein and VC were only 9.65% and 3.86%, respectively, at the same concentrations. The IC50 value of 6,8,3’,5’-FHD (0.042 ± 0.001 mmol L−1) was slightly lower than that of 8,3’,5’-THD (0.077 ± 0.027 mmol L−1). In addition, 6,8,3’,5’-FHD showed the best NO scavenging ratio at a high concentration of 2 mmol L−1.
The electron-donating abilities of 8,3’,5’-THD and 6,8,3’,5’-FHD were also determined using a reducing power assay and a phosphomolybdenum assay (total antioxidant capacity, TAC). As shown in Figure 4(a), the reducing power of all the samples increased on increasing their concentration. At a dose of 1 mmol L−1, the absorbance values at 700 nm of 8,3’,5’-THD, 6,8,3’,5’-FHD, daidzein, and VC were 2.537 ± 0.005, 2.633 ± 0.009, 0.314 ± 0.021, and 2.052 ± 0.114, respectively. The order of the potency was 6,8,3’,5’-FHD > 8,3’,5’-THD > VC > daidzein. As shown in Figure 4(b), the absorbance values at 695 nm of 6,8,3’,5’-FHD and 8,3’,5’-THD were higher than those of VC and daidzein at the same concentration, indicating that they possessed a stronger TAC than VC and daidzein. The order of the potency was same as in the reducing power assay.

Reducing activities of 8,3’,5’-THD, 6,8,3’,5’-FHD, and daidzein: (a) reducing power and (b) total antioxidant capacity. VC as the reference compound. Data are expressed as the mean value ± SD (n = 3).
Based on the results of these five antioxidant assays, 6,8,3’,5’-FHD and 8,3’,5’-THD, which contain six and five phenolic hydroxy substitutions, respectively, were found to exhibit significantly higher antioxidant activity than daidzein, which only possesses two phenolic hydroxy substitutions. This observation agreed nicely with the theory that the antioxidant capacity of flavonoids is proportional to the number of phenolic hydroxy groups.6,15 Besides, an ortho-trihydroxy group and an ortho-dihydroxy group also have a positive effect on the antioxidant activity. 16 6,8,3’,5’-FHD exhibited the best antioxidant activity, which may be attributed to the presence of the two ortho-trihydroxy groups on rings A and B. 6,8-DHD with an ortho-trihydroxy group on ring A and an ortho-dihydroxy group on ring B also exhibited excellent antioxidant activity.
Antihypoxia activities of 8,3’,5’-THD and 6,8,3’,5’-FHD
ROS play a key role in hypoxia-induced injury, 17 which can be eliminated by the administration of antioxidants. 18 Therefore, we further evaluated the protective effects of 6,8,3’,5’-FHD and 8,3’,5’-THD on hypoxia-induced injury in PC12 cells. As shown in Figure 5(a), treatment of PC12 cells with 6,8,3’,5’-FHD, 8,3’,5’-THD and daidzein at a concentration of 1 μmol L−1 had no obvious effect on the survival rate of the PC12 cells under normoxic conditions, indicating that these compounds showed no cytotoxicity or pro-proliferation activities on PC12 cells at the test concentration. In contrast, the viability of the PC12 cells was significantly decreased to 65.17% following hypoxia exposure for 24 h (Figure 5(b)). On pretreatment with 6,8,3’,5’-FHD, 8,3’,5’-THD, or daidzein, the cell viability significantly increased to 78.22 ± 4.82% (p < 0.01), 75.82 ± 3.80% (p < 0.01) and 72.10 ± 4.30% (p < 0.05), respectively, indicating the positive therapeutic effect of 6,8,3’,5’-FHD, 8,3’,5’-THD, and daidzein against hypoxia-induced damage. Among them, 6,8,3’,5’-FHD had the best cytoprotective activity. 8,3’,5’-THD also exhibited better cytoprotective activity than that of daidzein (p < 0.01).
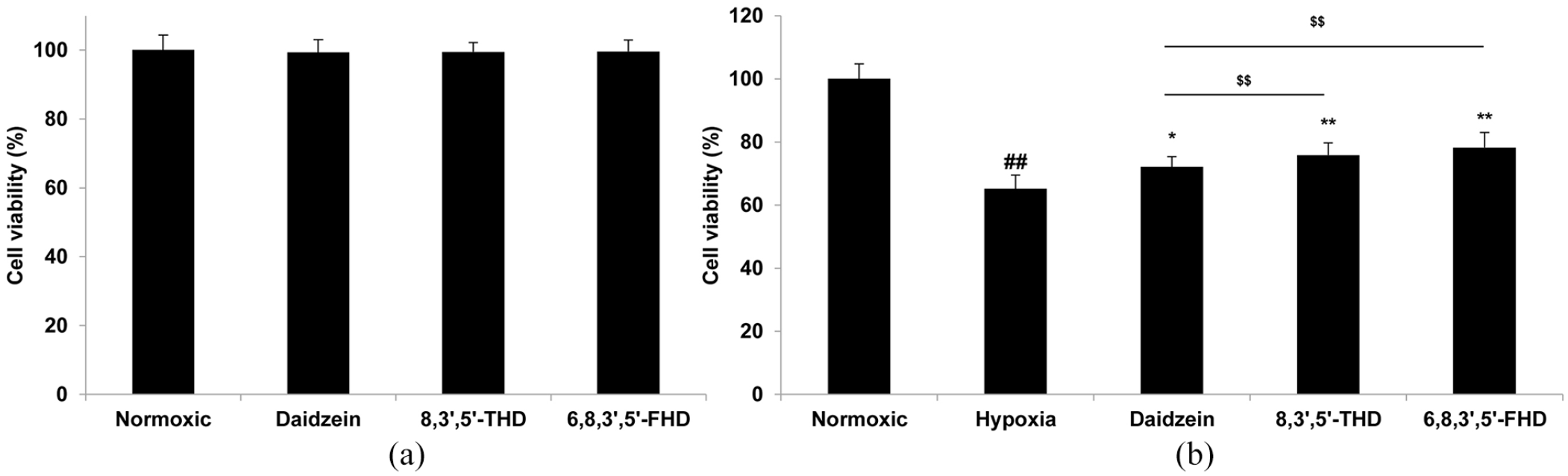
Protective effects on hypoxia-induced PC12 cells. (a) Effects of 8,3’,5’-THD and 6,8,3’,5’-FHD and daidzein at a concentration of 1 μmol L−1 on the cell viability of normal PC12 cells. (b) Effects of 8,3’,5’-THD and 6,8,3’,5’-FHD and daidzein at a concentration of 1 μmol L−1 on the cell viability of hypoxia-induced PC12 cells. Data are expressed as a mean value ± SD (n = 6).
Conclusion
In conclusion, 6,8,3’,5’-FHD and 8,3’,5’-THD have been synthesized for the first time via a simple and effective method using daidzein as the starting material. 6,8,3’,5’-FHD and 8,3’,5’-THD showed stronger antioxidant abilities compared with that of VC. In addition, they exhibited better protective effects on hypoxia-induced injury in PC12 cells in comparison with daidzein. These results indicate that 6,8,3’,5’-FHD and 8,3’,5’-THD are outstanding antioxidant agents and can be used for alleviating injury-induced by hypoxia.
Experimental section
Materials and apparatus
Daidzein (purity >98% by HPLC) was purchased from Ci Yuan Biotechnology Co., Ltd. (Shaanxi, China). Other reagents and solvents were purchased from commercial sources and used without further purification. Melting points were measured using an X-4B melting point apparatus. Flash column chromatography was performed on silica gel (200–300 mesh). Infrared (IR) spectra were recorded on a Bruker ALPHA FTIR spectrophotometer (Billerica, MA, USA). 1H NMR and 13C NMR spectra were recorded on a Bruker Avance III high-definition (HD) spectrometer in CDCl3 or DMSO-d6 with tetramethylsilane (TMS) as the internal standard. HRMS was performed on an Apex II instrument by means of the electrospray ionization (ESI). Low-resolution mass spectra were recorded on a liquid chromatography–mass spectrometry LC-MS-API 3200 mass spectrometer in ESI mode. The purity was analyzed using ultra-high-performance liquid chromatography (UHPLC) with a Thermo Acclaim-C18 column (100 × 2.1 mm, 2.2 µm, Waltham, MA, USA).
Chemistry
Synthesis of 8,3’,5’-tribromo-7,4’-dihydroxyisoflavone (1)
To a stirring solution of daidzein (20 mmol, 5.08 g) in CH2Cl2 (200 mL) and ethanol (20 mL) was added dropwise Br2 (4.1 mL). The reaction mixture was stirred at room temperature (RT, 25 °C) for 2.5 h, followed by treatment with saturated NaHSO3 solution (5 mL) to remove any remaining traces of Br2. The organic solvent was removed under reduced pressure to give a precipitate, which was collected by filtration, washed with distilled water, dried under vacuum, and washed with hot ethyl acetate to afford compound
Synthesis of 8,3’,5’-tribromo-7,4’-dimethoxyisoflavone (2)
To a stirred solution of compound
Synthesis of 7,8,3’,4’,5’-pentamethoxyisoflavone (3)
A solution of 25% sodium methoxide in methanol (70 mL) was added to a suspension of CuBr (2.14 g, 15 mmol) in DMF (5 mL). The mixture was stirred at RT for 1 h and added to a solution of
Synthesis of 7,8,3’,4’,5’-pentahydroxyisoflavone (8,3’,5’-THD)
To a stirred solution of AlCl3 (998 mg, 7.5 mmol) in CH2Cl2 (15 mL) was added dropwise dimethyl sulfide (548 µL) at 5 °C, and then compound
Synthesis of 6,8,3’,5’-tetrabromo-7,4’-dihydroxyisoflavone (4)
To a stirring solution of daidzein (10 mmol, 2.50 g) in ethanol (150 mL) was added dropwise Br2 (3.0 mL) at RT. The resulting mixture was then heated at reflux and stirred for 1 h. Water (200 mL) was added to the resulting mixture under boiling conditions. After cooling to RT, the obtained precipitate was collected by filtration, washed with water, and dried under vacuum to give compound
Synthesis of 6,8,3’,5’-tetrabromo-7,4’-dimethoxyisoflavone (5)
To a stirred solution of compound
Synthesis of 6,7,8,3’,4’,5’-hexamethoxyisoflavone (6)
A solution of 25% sodium methoxide in methanol (50 mL) was added to the suspension of CuBr (1.43 g, 10 mmol) in DMF (4 mL). The mixture was stirred at RT for 1 h and then added to a solution of
Synthesis of 6,7,8,3’,4’,5’-hexahydroxyisoflavone (6,8,3’,5’-FHD)
Dimethyl sulfide (700 µL) was slowly added dropwise to a stirred solution of AlCl3 (1.33 g, 10 mmol) in CH2Cl2 (15 mL) at 5 °C. Compound
Purity test
The purity was analyzed using a Thermo Scientific UltiMate 3000 UHPLC system with a Thermo Acclaim-C18 column (100 × 2.1 mm, 2.2 µm). Isocratic elution was performed using methanol (A) and H2O (0.01% acetic acid, B) with the following gradient combination: 30% A for
Antioxidant activity assay
DPPH radical scavenging assay
The DPPH radical scavenging assay was performed according to the reported method with slight modifications. 21 In brief, 100 μL of the sample in DMSO at various concentrations (0.0625–2.0 mmol L−1) and 100 μL of DPPH solution (100 µM) in methanol were added to a 96-well plate and shaken. After incubation in the dark for 30 min at RT, the absorbance of the resulting solution was recorded at 517 nm using a microplate reader (SpectraMax i3, Molecular Devices, San Jose, CA, USA). The DPPH free-radical scavenging rate was calculated using the equation
where Ab is the absorbance of the blank control (containing all reagents except samples), Ac is the absorbance of the sample control (containing all reagents except DPPH), and As is the absorbance of the sample (containing all reagents with the samples). The antioxidant activity is expressed as the value IC50 value (mmol L−1), which is calculated as the concentration of sample required to scavenge 50% of the DPPH radicals. A low IC50 value corresponded with higher antioxidant activity.
Superoxide anion (O2−•) radical scavenging assay
The O2−• scavenging activity was performed using the phenazine methosulfate (PMS)–nicotinamide adenine dinucleotide (NADH)–nitrotetrazolium blue chloride (NBT) system with slight modifications. 22 In brief, 100 µL of the sample in DMSO at various concentrations (0.015625–1.0 mmol mL−1), 50 µL of NBT solution (0.2 mM) in distilled water, and 50 µL of NADH solution (0.5 mmol L−1) in 0.1 M Tris–HCl, pH 8.0 were added to a 96-well plate. PMS solution (50 µL, 25 µM) in distilled water was added to the mixture, which was shaken and then incubated for 10 min at RT. The absorbance of the resulting solution at 570 nm was recorded using a microplate reader (SpectraMax i3, Molecular Devices). The superoxide scavenging rate was calculated using equation (1), where Ac is the absorbance of the sample control (containing all reagents except PMS), and the other parameters are the same as mentioned above.
NO radical scavenging assay
The NO radical scavenging assay was measured by the Griess reaction with some modifications. 23 In brief, 50 µL of the sample in DMSO at various concentrations (0.0625–2.0 mmol mL−1) and 50 µL of sodium nitroprusside (20 mmol L−1 in phosphate buffer, pH 7.4) were added to a 96-well plate and shaken. After incubation under light for 1.5 h at RT, 50 µL of 0.33% (w/v) sulfanilamide (in 20% glacial acetic acid) and 50 µL of 0.1% (w/v) naphthyl ethylenediamine hydrochloride were added to the resulting solution, which was further incubated for 30 min at RT. The absorbance was recorded at 540 nm in a microplate reader (SpectraMax i3, Molecular Devices). The NO radical scavenging rate was calculated according to equation (1), where Ac is the absorbance of the sample control (containing all reagents except sodium nitroprusside), and the other parameters are the same as mentioned above.
Reducing power assay
The reducing power assay was performed according to a previous reported method with slight modification. 24 In brief, 100 μL of the sample in DMSO at various concentrations (0.03125–1.0 mmol L−1) were mixed with 2.5 mL of 0.2 mol L−1 sodium phosphate buffer (pH 6.6) and 2.5 mL of 1% (w/v) potassium ferricyanide and incubated for 30 min at 50 °C. After cooling to RT, 2.5 mL of 10% trichloroacetic acid (TCA) was added to the mixture, which was then centrifuged at 3000 r/min for 10 min. The upper layer fraction (2.5 mL) was mixed with 2.5 mL of distilled water and 0.5 mL of 0.1% ferric chloride and reacted at RT for 30 min. The absorbance was recorded at 700 nm using a UV-Vis spectrophotometer (NanoPhotometer-NP80, Implen, Westlake Village, CA, USA). A higher absorbance indicated a stronger reducing power.
Phosphomolybdenum assay
The TAC of the sample was determined using the phosphomolybdate method. 25 In brief, 100 µL of the sample in DMSO at various concentrations (0.03125–1.0 mmol L−1) was mixed with 1.0 mL of the reagent solution (0.6 M sulfuric acid, 28 mM sodium phosphate, and 4 mM ammonium molybdate). After incubation at 95 °C for 90 min and then rapid cooling to RT, the absorbance of the resulting mixture was recorded at 695 nm using UV-Vis spectrophotometry (NanoPhotometer-NP80, Implen). A higher absorbance indicates a higher total antioxidant activity. Five assays were performed using Vitamin (VC) as the positive control. All samples were analyzed in triplicate.
Antihypoxia activity
Cell culture and treatment
Rat pheochromocytoma (PC12) cells were obtained from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). As a normoxic control, cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM; Bioss, Beijing, China) supplemented with 10% fetal bovine serum (FBS) (Bioss) and 1% streptomycin–penicillin (Bioss) in a humidified incubator with 95% air and 5% CO2 at 37 °C. To simulate hypoxia injury, the cells were incubated for 24 h at 37 °C in a hypoxic incubator containing 1% O2, 94% N2, and 5% CO2.
Cell viability assay
Cell survival assays were carried out using the cell counting kit-8 (CCK-8) according to the manufacturer’s instructions. Samples were dissolved in DMSO and subsequently diluted in DMEM with the final concentration of DMSO less than 0.1% (v/v). PC12 cells were cultured in a 96-well plate for 24 h and pre-treated with or without samples at a concentration of 1 μmol L−1 for 1 h. Cells were then cultured in normoxic or hypoxic conditions for 24 h. The medium was discarded and then 100 µL of fresh medium containing 10 µL of CCK-8 solution (Bioss, Beijing, China) was added to each well. After incubation for 2 h, the absorbance at 450 nm was obtained using a microplate reader (SpectraMax i3, Molecular Devices). The relative cell viability was calculated as the percentage of normoxic control.
Supplemental Material
sj-docx-1-chl-10.1177_17475198221131528 – Supplemental material for The design, synthesis, antioxidant, and antihypoxia activities of two new hydroxydaidzein derivatives
Supplemental material, sj-docx-1-chl-10.1177_17475198221131528 for The design, synthesis, antioxidant, and antihypoxia activities of two new hydroxydaidzein derivatives by Qingyue Da, Jin Shao, Pengpeng Zhang, Jie Zhang, Huiping Ma and Linlin Jing in Journal of Chemical Research
Footnotes
Date analysis
The values presented in the figures are mean values ± standard deviation (SD). All experiments were performed at least three times. Statistical analysis was performed using SPSS 19.0 software.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (grant nos 81872796, 81202458, and 81571847) and The Logistics Research Program of PLA (grant no. CWH17J010).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

