Abstract
Medicinal plants have been used for the treatment of various diseases and still play an important role in covering the basic health needs in the developing countries like Ethiopia. In this study, successive extraction, partial purification using column chromatograpy, phytochemical screening, determination of the total phenolic and flavonoid content, and the antioxidant and antibacterial activities of the petroleum ether, chloroform, and ethanol stem bark extracts and fractions of Clematis simensis are reported. The results show the presence of different valuable phytochemicals. The total phenolic content is 54.744, 68.933, and 87.522 mg g–1 of gallic acid equivalent for the petroleum ether, chloroform, and ethanol extracts, respectively. The total flavonoid content of the petroleum ether, chloroform, and ethanol extracts are 22.770, 37.223, and 52.289 mgCE g–1, respectively. This could lead to potential biological activity of the plant extract. Besides, the ethanol extract exhibits higher antioxidant activity than all the tested extracts. Likewise, the extracts have promising antibacterial activity with that of the ethanol extract being higher than those of the chloroform and petroleum extracts as well as isolated fractions.

Introduction
Medicinal plants have been used for the treatment of various diseases since ancient time and still play important roles to cover the basic health needs in developing countries. A majority of people in developing countries use medicinal plants for the treatment of infectious diseases. Medicinal plants are used in spiritual therapies and manual techniques and are applied to treat, diagnose, or prevent illness. 1 Even in developed countries, people seek alternative herbal medicines because of the side effects that are common in strong modern drugs. According to the World Health Organization (WHO), herbal medicines serve the health needs of about 80% of the world population, especially for the millions of people in the vast rural areas of developing countries. Thus recently, there has been significant interest in the development of new drugs from plants. 2 Medicinal plants have the capacity to produce a combined effect that surpasses the total activity of the individual constituents synergistically. Several investigations have examined the antimicrobial effects of herbal plants extracts, including roots, stem, leaves, and flowers. 3
In Ethiopia, the majority of the population use traditional medicines for the treatment of various types of infections and disorders. However, traditional practitioners in Ethiopia use the plants without scientific dose optimization. Reports indicate that different parts of the plant are used traditionally to treat different types of infections. According to recent reports, 25% of modern drugs are derived from extracts of medicinal plants. 4 Plants are therefore important sources of active natural products, including phytochemicals like flavonoids, alkaloids, tannins, and other polyphenolic compounds, which have different structures, biological properties, and mechanisms of action. 5
Clematis simensis is one of the common medicinal plants in use in Ethiopian society. C. simensis (Figure 1) is a genus of clematis within the family Ranunculaceae. A number of ethnobotanical studies have reported that the leaves of C. simensis are used in folk remedies for the treatment of hemorrhoids, itching, leishmania, coughs, eczema, ringworm, tropical ulcers and fever, and also as an insecticide and rubefacient. In addition, the leaves are crushed and tied on to the affected area for the treatment of swollen legs. 6 In Ethiopia, the stem bark of C. simensis is used for treatment of toothache and cancer. 7

Stem bark and aerial parts of Clematis simensis.
Ethno-veterinary medicine is very important for animal health care in most developing countries, and research are still going on toward the production of low-cost, resistance-free, and locally available antimicrobial drugs. 8 However, phytochemical investigations and biological activity studies of the stem bark of C. simensis have not been well studied in Ethiopia. Therefore, the aim of this study was to investigate the phytochemical compositions of the crude substances and isolated fractions, and to assess the antibacterial effects and antioxidant activity of the stem bark of C. simensis grown in Ethiopia by using different solvent systems.
Results and discussion
Isolation and characterization
Before performing column chromatography, different solvent systems were used to obtain good resolution. Finally, petroleum ether, dichloromethane, and ethyl acetate with different ratios were used as solvent systems for thin-layer chromatography (TLC) studies of the chloroform extract of the stem bark of C. simensis. Solvent system I consisted of petroleum ether / ethyl acetate (10:0), which gave no separately visible detected spots and its Rf value was 0. In solvent system II, which consists of petroleum ether / ethyl acetate (8:2), five spots with the Rf values of 0.2, 0.28, 0.3, 0.43, and 0.5 were collected. In solvent system III, petroleum ether / ethyl acetate (7:3), five spots were detected with Rf values of 0.25, 0.35, 0.45, 0.6, and 0.80. In solvent system IV, dichloromethane / petroleum ether (8:2), three spots were observed having Rf values of 0.09, 0.15, and 0.21. In solvent system V, dichloromethane / petroleum ether (10:0), four spots were observed having Rf values of 0.1, 0.25, 0.4, and 0.92. In solvent system VI: dichloromethane / acetone (8:2), five spots were detected with Rf values of 0.25, 0.3, 0.6, 0.79, and 0.81 (Figure 2).

TLC analysis of the first elution process.
The TLC profiles in chloroform were impressive and indicate the presence of a number of phytochemicals. Different phytochemicals have different Rf values in different solvent systems. This variation in Rf values of the phytochemicals provides a very important clue in understanding their polarity and also helps in the selection of appropriate solvent systems for the separation of pure compounds by column chromatography. The selection of an appropriate solvent system for particular plant extracts was achieved by analyzing the Rf values of the compounds in different solvent systems. The chloroform extract of C. simensis was subjected to column chromatography over silica gel (150 g). The column was eluted with solvents of increasing polarity, and a total of 10 fractions were collected. The purity of each fraction was checked by TLC. Fractions 6, 7, 8, 9, and 10 show the Rf values given in Table 1. The Rf values of fractions 6 and 7 were the same and those of fractions 8, 9, and 10 were the same. Next, the same fractions of 6 and 7 were eluted further by column chromatography to give one single spot denoted as F1 with an Rf value of 0.8, also fractions 8, 9, and 10 were eluted further to give one single spot denoted as F2 with an Rf value of 0.7 (Figure 3).
TLC Rf values for the chloroform extract of Clematis simensis after elution.

TLC: thin-layer chromatography.

TLC analysis of isolated pure fractions.
Qualitative phytochemical analysis
Phytochemical screening was carried out on C. simensis extracts to test the presence of the most relevant bioactive compounds used for medication. 9 The confirmation of the presence or absence of phytochemical constituents in the stem bark of C. simensis was determined via one test method used for each natural compound. This study reveals the presence of various bioactive secondary metabolites that might be responsible for the medicinal attributes. This test confirmed the presence of flavonoids and steroids in all the solvent extracts, and phenol and tannin being present in the ethanol extract. Terpenoids and cardiac glycosides were present in the chloroform extract and tannin was also present in the chloroform extract. Cardiac glycoside was present in fraction 1 (F1), and the terpenoid was present in fraction 2 (F2). These results suggest that the phytochemical properties of the plant can be used for curing various ailments and also possess potential antioxidant activity. Table 2 shows the various phytochemical identification tests performed on the C. simensis stems bark crude extracts in petroleum ether, chloroform, and ethanol and of isolated fractions.
Phytochemical test results of the crude extracts and the fraction of stem bark of C. simensis..

PE: petroleum ether extract; ChE: chloroform extract; EE: ethanol extract; F1: isolated fraction 1; F2: isolated fraction 2; +: the presence of phytochemicals; −: the absence of phytochemicals in the crude extract.
Spectroscopic analysis (infrared and ultraviolet)
After isolation, pure fractions F1 and F2 were tested by using Fourier transform infrared (FTIR) and ultraviolet (UV) instruments. A broad IR absorption band at 3412 cm–1 indicates the existence of a hydroxy group, while the absorption band at 1612 cm–1 is attributed to a C=C alkene functional group.
The absorption bands observed at 1418 and 1099 cm–1 are attributed to a C–H bending system and the C–O stretching vibration of the hydroxy attached to the carbon, respectively. Furthermore, the IR absorptions at 2861 and 2923 cm–1 suggest sp3 C–H stretching and sp2 C–H stretching vibrations, respectively. The UV spectrum showed characteristic absorption bands at λmax 412 and 280 nm, due to conjugation of n →π* and π →π* transitions.
According to the spectroscopic analysis and more importantly, the phytochemical screening test, the isolated fractions could be grouped within classes of natural compounds. For example, fraction 1 was classified under cardiac glycoside, and fraction 2 was classified under the terpenoid class of natural compounds. However, further studies are required to ascertain the precise identities of the compounds.
Quantitative analysis
Determination of the total phenolic content
The total phenolic content of the petroleum ether, chloroform, and ethanol extracts were estimated by Folin Ciocalteu’s method. Gallic acid was used as the standard and the total phenolic content is expressed as mg gallic acid equivalence (mg GAE g–1). In this study, the total phenolic contents of the C. simensis stem bark crude extracts were determined based on the calibration curve of gallic acid (Figure 4), with the absorbance recorded at 760 nm. A set of standard solutions of gallic acid (1, 6, 12, 18, and 24 mg L–1) was used to construct the calibration curve. The calibration curve was constructed by plotting absorbance versus concentration and a straight line with an equation of y = 0.068X – 0.068, and a linear regression coefficient (R2) of 0.999 was obtained. The total phenolic content of the crude extracts was solvent-dependent. The higher the polarity of the solvent, the greater the total phenolic and flavonoid content in the extract.

Calibration curve for phenolic determination.
In the present study, the amount of total phenolic content in extracts of different polarities varied, ranging from 54.744, 68.933, and 87.522 mg g–1 of GAE. The ethanol extract exhibited the highest total phenolic content (87.522 mg GAE g–1), followed by chloroform (68.933 mg GAE g–1) and petroleum ether (54.744 mg GAE g–1), as indicated in Table 3.
Total phenolic content of the stem bark extract of Clematis simensis.

Total flavonoid content determination
The total flavonoid content (TFC) of C. simensis stem bark extract was determined by the aluminum chloride calorimetric method. 10 The absorbance of each sample was measured, and then the standard calibration curve was constructed (Figure 5). Concentrations of 2.4, 4.7, 9.4, 33.9, and 50.9 ppm of catechin were used to construct the calibration curve. The calibration curve was constructed by plotting absorbance versus concentration and a straight line with an equation of y = 0.08 + 0.005x, and a linear regression coefficient (R2) of 0.998 was obtained.

Calibration curve of catechin as a standard for total flavonoid determination.
The TFC of the samples was determined in terms of the number of catechin equivalents mgCE per g using equation 2, and the results are given in Table 4. The flavonoid content was higher in the ethanol extract (52.289 mg g–1) and the lowest TFC (22.770 mg g–1) was recorded in the least polar solvent (petroleum ether). The present study agrees with the result reported by Mostafa and co-workers. According to this report, the highest amount of TFC was derived from the acetone extract and the lowest amount was recorded in the water extract of C. hirusta. Hence, the presence of flavonoids and phenolic compounds in the ethanol stem bark extracts of C. simensis confirm the traditional use of the plant for folk remedies. Since it contains a high proportion of polyphenols and flavonoids, it is reasonable to postulate that it possesses antioxidant and anticancer activities. This increased level of total phenolics might be due to the presence of phenolic compounds such as coumarins, flavonoids, lignans, neolignans, lignins, and phenylpropenes in C. simensis. 11
Total flavonoid content of each solvent extract of C. simensis stem bark.

Antioxidant capacity assay
DPPH radical scavenging activity
The antioxidant activity of C. simensis stem bark extracts was determined using an ethanol solution of the 2,2-dyphenyl-1-picrylhydrazyl (DPPH) reagent. A freshly prepared DPPH solution exhibits a deep purple color with an absorption maximum at 517 nm. This purple color generally fades when antioxidant molecules quench DPPH free radicals (i.e. by providing hydrogen atoms or by electron donation, conceivably via free-radical attack on the DPPH molecule) and convert them into a colorless / bleached product, which leads to a decrease in the absorbance band at 517 nm. 12 The concentration of each extract required to inhibit 50% of the DPPH free radical at 519 nm was calculated to quantify the antioxidant activities (IC50 values) shown in Table 5. The IC50 values were found to be 63.522, 45.159, 42.35, 50.928, 53.277, and 25.805 mg L–1 for the petroleum ether, chloroform, and ethanol extract and the isolated fractions of F1 and F2 and ascorbic acid, respectively (Figure 6).
IC50 values of stem bark extracts of Clematis simensis toward DPPH scavenging activity.

DPPH: 2,2-dyphenyl-1-picrylhydrazyl.

Inhibition (%) versus concentration plots of C. simensis stem bark extracts and ascorbic acid toward DPPH scavenging activity.
Among the studied extracts, the ethanol extract showed the highest scavenging activity, followed by chloroform and petroleum ether, but all extracts IC50 values were lower compared to the standard sample of ascorbic acid as shown in Table 5. The scavenging activity of pure fractions, such as fraction 1 and fraction 2, were lower compared to those of the ethanol and chloroform crude extracts but, were higher than the petroleum ether extract. The higher scavenging activity of the chloroform crude extract, compared to the pure fractions is due to a synergetic effect. The highest scavenging activity of the ethanol extract, among all the extracts, indicates that it has a number of bioactive phytochemical constituents. Table 6 shows that the stem bark extract exhibits significant DPPH free-radical scavenging effects compared to standard ascorbic acid.
Absorbance values of the stem bark extracts of Clematis simensis from DPPH assays.

DPPH: 2,2-dyphenyl-1-picrylhydrazyl.
Reducing power assay/ferric reducing antioxidant power
In vitro antioxidant assays for the petroleum ether, chloroform, and ethanol extracts and isolated fractions F1 and F2 were determined by the ferric reducing antioxidant power method using ascorbic acid as the standard. The reducing power is a measure of the ability of the extract to reduce Fe3+ to Fe2+. Substances with reduction potentials react with potassium ferrocyanide (Fe3+) to form potassium ferrocyanide (Fe2+), and can then react with ferric chloride to form a ferric-ferrous complex. 13 Ascorbic acid solutions of concentration 20–100 mg L–1 and concentrations of 40, 60, 80, and 100 mg L–1 of each extract and isolated fractions were used.
Table 7 shows that the reducing powers of the petroleum ether, ethanol, and chloroform extracts and those of isolated fractions F1 and F2 were weaker than the positive control of ascorbic acid. However, the reducing capacity of the ethanol extract was higher followed by the chloroform extract; fractions F1 and F2, and petroleum ether extract had lower capacities. The results of the percent reducing power were proportional with the DPPH assay. From the above results, the ethanol extract has the highest antioxidant activity compared to the chloroform and petroleum ether extract (Figure 7).
Absorbance of the stem bark extracts of Clematis simensis stem bark obtained from reducing power assays.


Reducing power (%) versus concentration plots of C. simensis stem bark extracts and ascorbic acid in reducing power assays.
Antibacterial activity
The antibacterial activity of the petroleum ether, chloroform, and ethanol stem bark extracts of C. simensis were evaluated by using the Agar disk diffusion method. Different bacteria were used for the determinations: two were Gram-negative bacteria (Escherichia coli and Klebsiella pneumonia) and two were Gram-positive bacteria (Staphylococcus aurous and Streptococcus pyrogens). A concentration of 1 mg mL–1 for each extract and 0.2 mg mL–1 gentamicin were prepared and diffused into incubated plates in which bacteria had been cultured, and the inhibition zone values were recorded. As shown in Table 8, the ethanol extract had higher antimicrobial activity than the petroleum ether and chloroform extracts, and exhibited maximum zones of inhibition against both Gram-negative and Gram-positive bacteria. The present study shows that the different extracts of C. simensis stem bark possess significant antibacterial activity and provide a possible rationalization for the traditional medicinal use of the plant. Some of the phytochemical groups (see Table 2) detected in the different extracts may be responsible for the antibacterial activities.
Zones of inhibition of different extracts of C. simensis stem bark against both Gram-positive and Gram-negative bacteria.

Bold significance is used in order to magnify the results.
The present results show strong antibacterial activities against the tested bacteria with the ethanol extract exhibiting significantly (p < 0.05) better zones of inhibition ranging from 13 to 20 mm than those of the chloroform (12–17 mm) and petroleum ether extracts (10–11 mm) at a concentration of 1 mg mL–1 against the tested bacteria (Figure 8). Figure 9 shows that the isolated fractions have also remarkable antibacterial activity. In particular, F1 showed a zone of inhibition comparable with the chloroform and ethanol extracts, being even greater than that of the petroleum ether extract. Typically, Gram-positive bacterial strains are more susceptible to C. simensis stem bark extracts than Gram-negative bacteria at concentrations of 1 mg mL–1. This is because Gram-negative bacterial strains contain a cell wall covered by a strong phospholipid bilayer system.
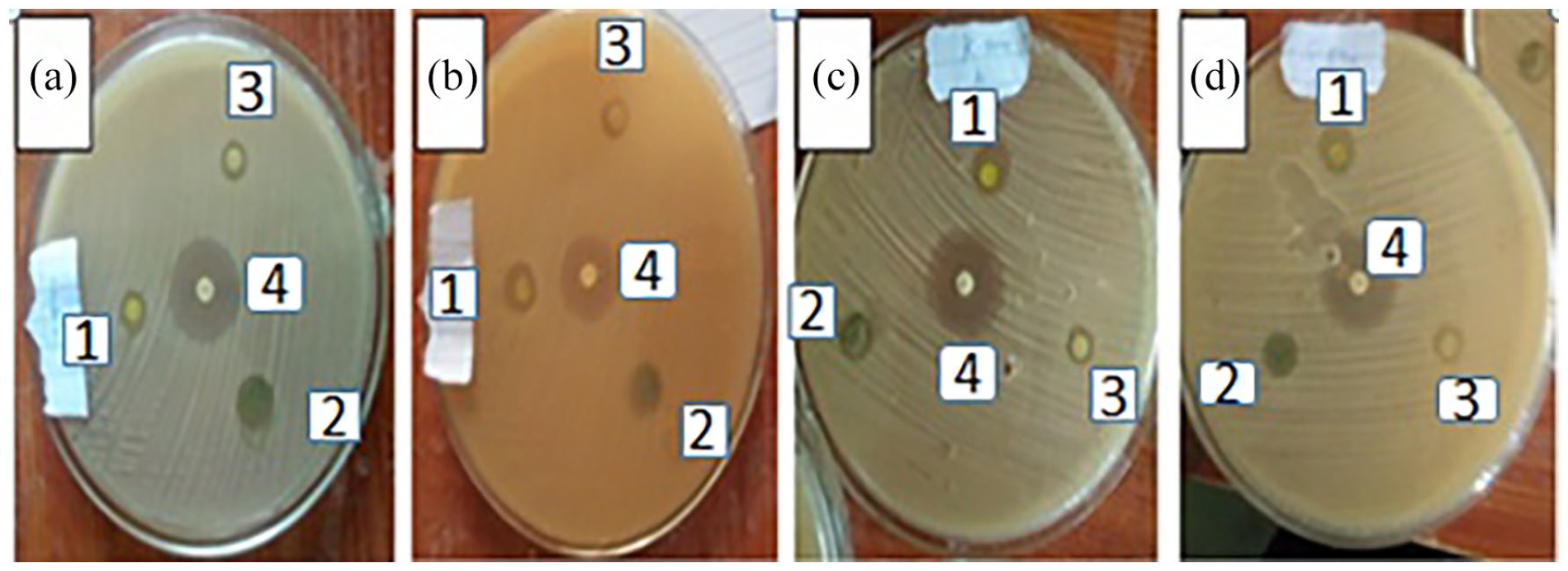
Zones of inhibition of 1 mg mL–1 ethanol, chloroform, and petroleum ether extracts of C. simensis and 0.2 mg mL–1 of gentamicin against the tested bacterial strains. (a) S. aureus, (b) E. coli, (c) K. pinnata, and (d) S. pyogenes.

Zones of inhibition of 1, 0.5, 0.25, and 0.125 mg mL–1 concentrations of each isolated fraction of F1 and F2 of C. simensis against the tested bacterial strains. (a) S. aureus, (b) E. coli, (c) K. pinnata, and (d) S. pyogenes.
Conclusion
C. simensis, a well-known medicinal plant has been widely used in many countries for traditional medicinal purposes. In the present study, the powdered stem bark of C. simensis was successively extracted with petroleum ether, chloroform, and ethanol to obtain the phytochemical constituents of phenols, tannins, steroids, and flavonoids with high antioxidant activity. The chloroform extract was subjected to further purification to give two pure fractions F1 and F2. The fractions were classified as cardiac glycoside and steroid functional groups, respectively. The results of phytochemical analysis revealed the presence of phenols, flavonoids, tannins, terpenoids, steroids, cardiac glycosides, and glycosides in different extracts of the plant.
Based on the present study, it can be concluded that the stem bark extracts of C. simensis exhibited different levels of antioxidant activity and contained potential antimicrobial components that may be of use in the pharmaceutical industry as alternative therapies against bacterial diseases and to formulate new antimicrobial and antioxidant.
However, further studies are necessary to isolate and elucidate the structures of the bioactive compounds of the plant extracts and to determine the pharmaceutical potential of the plant as a medicine. Also, this plant should be studied more extensively to explore its antimicrobial activities by using other bacteria, fungi, and viruses on the crude extracts as well as the isolated compounds.
Experimental section
Study area and study design
The study was conducted in Bahir Dar city situated on the southern shore of Lake Tana, which is located North Western part of Ethiopia approximately 565 km from Addis Ababa, being the capital of Amhara National Regional State (ANRS). The experiment was conducted at Bahir Dar University Department of Chemistry Postgraduate Research Laboratory.
Chemicals and reagents
The chemicals and reagents used for this study were chloroform, dichloromethane, ethanol, petroleum ether, and ethyl acetate. Standards of phenolic acids (gallic acid) and of flavonoids (catechin), and ascorbic acid for DPPH, Folin-Ciocalteu’s phenol reagent, and aluminum chloride (AlCl3) were used. Distilled water or deionized water, 10% ferric chloride, Wagner’s reagent (Iodine in potassium iodide), sodium nitrite (NaNO2), sodium carbonate (Na2CO3), hydrochloric acid (HCl), sulfuric acid (H2SO4), sodium hydroxide (NaOH), iodine, trichloroacetic acid (TCA), potassium hexacyanoferrate(II), ammonia solution, dimethylsulfoxide (DMSO), silica gel for column chromatography, a standard antibiotic (gentamicin), and Mueller-Hinton agar (MHA) as culture medium were employed.
Plant materials
The fresh stem bark of C. simensis was collected from Bahir Dar Bezawit Maryam Forest, 565 km northwest of Addis Ababa, Ethiopia, in December 2020. The plant was identified by a botanist in Department of Biology at Bahir Dar University. The bark was cut into small pieces and dried in air for several days. After drying, the material was ground into a fine powder and stored at Organic Chemistry Postgraduate Research Laboratory, Bahir Dar University.
Extraction and isolation
The powdered plant sample (1 kg) was successively extracted with petroleum ether (2 L), chloroform (2 L), and ethanol (2 L), using the maceration extraction technique. The flasks containing powdered plant sample were sealed with a cotton plug and aluminum foil and stored for 96 h at room temperature with frequent shaking on an automatic shaker. The samples were subsequently filtered through Whatman (number 1) filter paper. The filtrates were concentrated to furnish 4 g (0.4%), 7 g (0.7%), and 9 g (0.9%), respectively. The concentrated extracts were obtained as crude extract. The chloroform crude extract was isolated by column chromatography. The chloroform extract (4.5 g) was adsorbed on an equal amount of silica gel and fractionated over silica gel (120 g) by column chromatography. The column was eluted with petroleum ether / dichloromethane / ethyl acetate of increasing polarities to furnish 10 fractions. A volume of 50 mL of each fraction was collected. The first fraction (F) was collected with 100% petroleum ether. F2 to F10 were eluted with petroleum ether / ethyl acetate with ratios 9:1, 8:2, 7:3, 3:2, 3:7, 1:4, 1:9, and 0:10, respectively. Fractions that showed similar Rf values and characteristic colors on TLC were combined and repurified to give pure fractions, which were subjected to biological activity testing. 14
Qualitative phytochemical analysis
Preliminary phytochemical analyses of the extracts were carried out using standard literature protocols with slight modifications.15–18
Determination of the total phenolic content
The total phenolic content was determined by the Folin-Ciocalteu method using spectrophotometry. 19 The reaction mixture consists of 1 mL of each extracts taken from the concentration of 1 mg mL–1 separately and 2 mL of distilled water was added. The mixtures were treated in 1 mL of Folin–Ciocalteu phenol reagent and shaken vigorously. After 5 min, the mixtures were treated with 2 mL of 7% Na2CO3 solution.10 The volumes were made up to 6 mL. A set of standard solutions of gallic acid (1.2, 6, 12, 18, and 24 mg L–1) were prepared.
The absorbance of the test and standard solutions were determined against a reagent blank at 765 nm with UV/VIS spectrophotometer. Quantification was performed on the basis of a standard curve of gallic acid based on the measured absorbance. The concentration of gallic acid was read (mg L–1) from the calibration line. Next, the content of phenols in the extracts was expressed in terms of gallic acid equivalents (mg of GA g–1 of extract).
Determination of the TFC
The TFC of the extracts of C. simensis was determined by using the aluminum chloride colorimetric method. 20 In this method, catechin was used to record the calibration curve; 10 mg of catechin was dissolved in methanol and then diluted to 2.4, 4.7, 9.2, 34, and 51 mg L–1. A calibration curve was generated by measuring the absorbance of the solutions at 510 nm (λmax of catechin) with a UV spectrophotometer. Aluminum chloride (1%) and NaNO2 (1 M) solutions were prepared. A 1 mL sample of each extract (0.1 mL of aluminum chloride, 0.1 mL of sodium nitrite solution, 2.8 mL of distilled water and 2 mL of 1 mol L–1 NaOH) was added and mixed well. A sample blank was prepared in a similar way by replacing aluminum chloride with distilled water. Similarly, a blank and samples of all three extracts were prepared and their absorbances were measured at 510 nm. All the prepared solutions were filtered through Whatman filter paper before recording the measurement. The concentration values of the extracts were obtained from the catechin standard curve by interpolating to the x-axis. Finally, the TFCs were determined based on the calibration curve.
Antioxidant capacity assay
DPPH radical scavenging assay
The determination of the free-radical-scavenging activity of the petroleum ether, chloroform, and ethanol crude extracts, and the isolated fractions were carried out using the DPPH assay.9 Concentrations (20, 40, 60, 80, and 100 mg L–1) of ascorbic acid as a standards and samples (40, 60, 80, and 100 mg L–1) of each extract were prepared. One (1) milliliter of 0.004% DPPH in ethanol was added to 4.0 mL of both the extracts and standard. The final concentrations (33.3, 50, 66.6, and 86.6 mg mL–1) of extract were obtained and allowed to incubate for 30 min. The UV absorbance of the resulting solution was measured at 517 nm.9 The procedure was accomplished by using ascorbic acid as the reference compound. The decrease in absorbance was then converted into percentage inhibition using equation (3) and IC50 values were determined by using equation (5).
Reducing power assay
The reducing power of the extracts of petroleum ether, chloroform, and ethanol and the isolated fractions of C. simensis stem bark were quantified by a described method with slight modification. 21 To 2 mL of the sample of different concentrations (40, 60, 80, and 100 mg L–1) were added 2.5 mL of phosphate buffer (0.2 M, pH 6.6) and 2.5 mL of (1%) potassium ferricyanide (K3Fe (CN)6), and the mixture were incubated at 50 °C for 20 min. After cooling, the reaction was terminated by adding 2.5 mL of TCA solution (10%) and centrifuged at 3000 r/min for 10 min. The supernatant (2.5 mL) was then mixed with 2.5 mL of distilled water and 0.5 mL of 0.1% ferric chloride. The absorbance of the reaction mixture was recorded at 700 nm. Ascorbic acid was used as a positive control. The increased absorbance of the reaction mixture indicated a higher reducing power of the plant extract. 22
Antibacterial activity testing
Antibacterial assay
The petroleum ether, chloroform, and ethanol extracts and isolated fractions of C. simensis stem bark were evaluated in antibacterial assays by using the agar disk diffusion method. The antibacterial activities of all samples were investigated using four different species of bacteria from American-type culture collection (ATCC), which includes two Gram-positive bacteria Staphylococcus aureus and Streptococcus pyogenes and two Gram-negative bacteria, Escherichia coli and Klebsiella pneumonia, using MHA medium. 23 All the bacterial species were obtained from the Department of Biology, Bahir Dar University.
Preparation of inoculums
The tested bacterial species were transferred from the stock cultures and streaked on MHA plates and incubated for 24 h. Well-separated bacterial colonies were then used as inoculums. Bacteria were transferred using a bacteriological loop to autoclaved MHA that had been warmed to about 45 °C in a water bath and mixed by gentle swirling. The medium was then poured into a sterile Petri dish, allowed to solidify and used for biological test. 23
Preparation of the test solution for assessing the antibacterial activity
The petroleum ether, chloroform, and ethanol crude extracts of 1 mg mL–1 concentration and isolated fractions were prepared in different concentrations (1, 0.5, 0.25, and 0.125 mg mL–1) in DMSO.
Testing for antibacterial activity
Sterilized filter paper disks (6 mm) were transferred to the MHA plates seeded with bacteria and incubated at 37 °C for 24 h. All the tests were performed in triplicate. The stem bark crude extracts and isolated fractions were used to test the sensitivity toward four bacteria. The standard drug gentamicin was used as a positive control and the corresponding solvent was used as a negative control (such as DMSO). After incubation, the zones of inhibition were recorded as the diameter of growth-free zones measured in mm using a ruler.
Method of data analysis
The total phenolic contents of the extracts of petroleum ether, chloroform, and ethanol were calculated as gallic acid equivalents (mgGAE g–1) by using the formula given below

where, GAE is the gallic acid equivalence (mg mL–1); V is the volume extract (mL), and m is the weight (g) of the pure plant extract.
The TFC of the extracts of petroleum ether, chloroform, and ethanol were calculated as catechin equivalents (mgCE g–1) by using the formula given below

where C = concentration of catechin from the standard curve
V = volume of extract, W =weight of plant extract.
The free-radical scavenging activity of each extract and the standard ascorbic acid were calculated from the following formula from the absorbance at 519 nm. The percentage scavenging activity of DPPH was calculated by using equation (3)

where A is the absorbance of DPPH; B is the absorbance of the extract/sample + DPPH.
The reducing power assay was also calculated by using equation (4)

where, Asample is the absorbance of sample and Ablank is the absorbance of blank

Statistical data analysis
The data for the quantitative studies, antioxidant and antibacterial activities are expressed as the average of three measurements, and all the remaining data are expressed as mean ± standard deviations of triplicates using MS Excel 2010, Origin 8.
Footnotes
Author contributions
Conception and design of study: B. B. Beyene, Y. G. Mehari
Acquisition of data: Y. G. Mehari, M. T. Ayana.
Analysis and/or interpretation of data: B. B. Beyene, Y. G. Mehari, and M. T. Ayana
Drafting the manuscript: M. T. Ayana, B. B. Beyene.
Revising the manuscript critically for important intellectual content: M. T. Ayana, B. B. Beyene.
All authors revised the manuscript.
Consent for publication
All authors consent to the publication.
Data availability statement
All data sets associated with this paper are included within the paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Using plants without scientific knowledge is sometimes hazardous because of unbalanced dose. The process used in this study does not affect humans, animals, and plants.
