Abstract
The complexes [Ni(HNOR)2]n·5nH2O (
Complexes [Ni(HNOR)2]n·5nH2O (
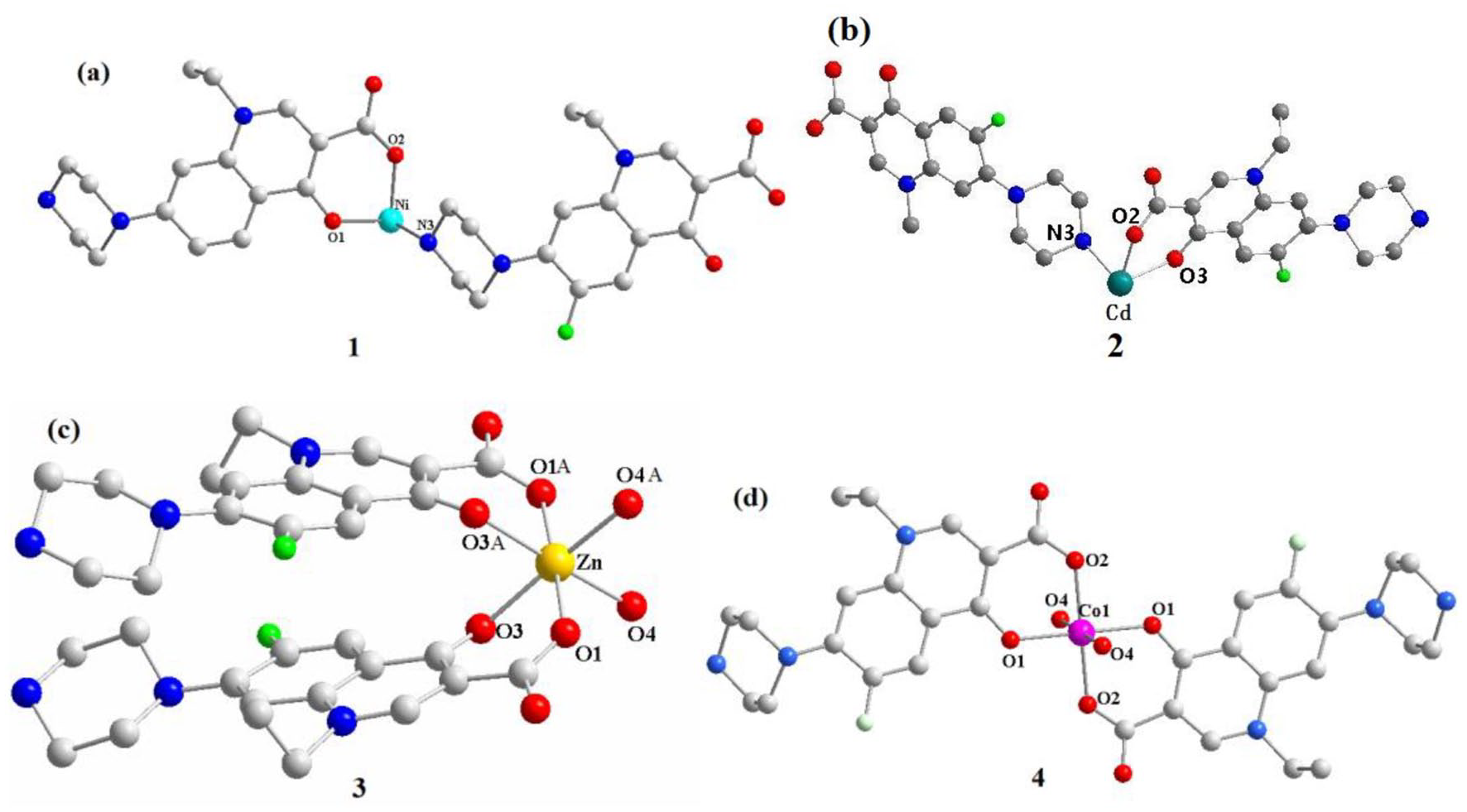
Introduction
Metal complexes are formed by the central atom providing the empty orbital and the coordinating atom providing the lone pair of electrons. In such complexes, oxygen and nitrogen atoms—due to their electronic configuration, electronegativity, and so on—can coordinate with many metals, and there are many different possible bonding modes.1–4 Due to their unique structural modification and composition diversity, metal complexes have a wide range of applications in catalysis, magnetism, luminescence, electrochemical sensing, and so on.5–9
Since 1982, when Lehn first described the photocatalysis of Ru-pyridine and Co-pyridine complexes, catalysis systems involving complexes have aroused wide academic interest and contribute to greener processes.10,11
Metal complexes can be used as catalytic materials, while their metal-directed self-assembly represents a powerful synthetic methodology for supramolecular architectures that have the advantages of flexible regulation at the molecular level and have characteristics of both inorganic and organic compounds.12–14 This ordered coordination improves their visible-light absorption ability and accelerates photo-generated charge mobility.15–18
Norfloxacin as a ligand was studied many years ago because of its multiple coordination sites; although similar complexes have been synthesized, there are some differences in the structures and X-ray crystallographic studies on similar structures have not been previously reported.19,20 Based on the above considerations, we have synthesized a series of complexes: [Ni(HNOR)2]n·5nH2O (
Results and discussion
Description of crystal structures
[Ni(HNOR)2]n·5nH2O (1)
Single-crystal X-ray diffraction analysis showed that complex

The asymmetric units of complexes
Selected bond lengths (Å) and angles (°) for complexes


Octahedral structures of complexes
Hydrogen bonds play an important role in the process of molecular crystal formation due to their electrostatic and directional properties. Oxygen atoms with large electronegativity and a small radius in the HNOR− ligands and in water molecules easily form hydrogen bonds (Table 2). The metal–ligand interactions and the hydrogen bond interactions turn complex
Hydrogen bonds (Å) and angles (°) for complexes 1 and 4.
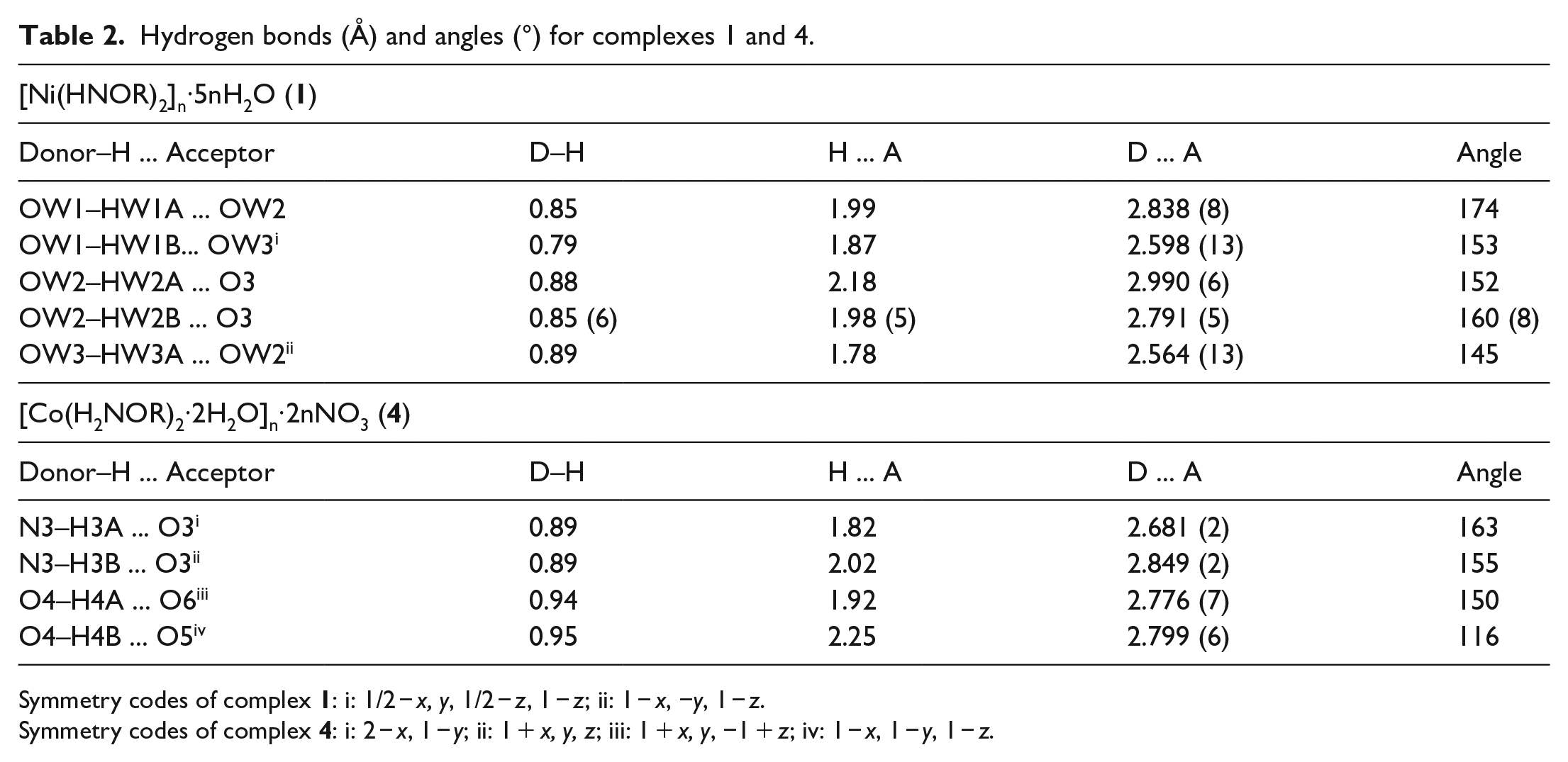
Symmetry codes of complex
Symmetry codes of complex

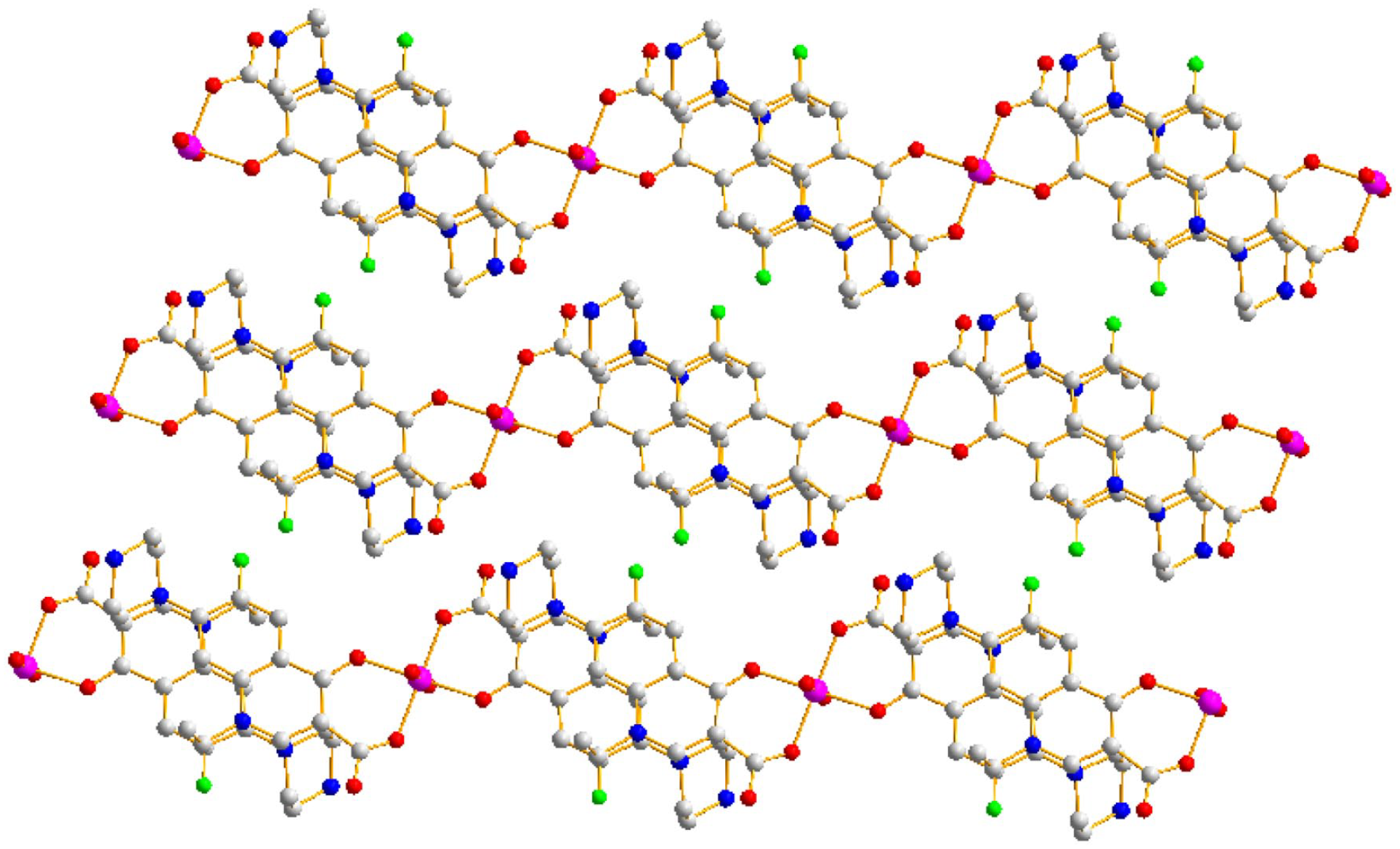
Two-dimensional layered structure of complex
[Cd(HNOR)2]n (2)
Single-crystal X-ray diffraction analysis revealed that the complex [Cd(HNOR)2]n (
The bond lengths of complex
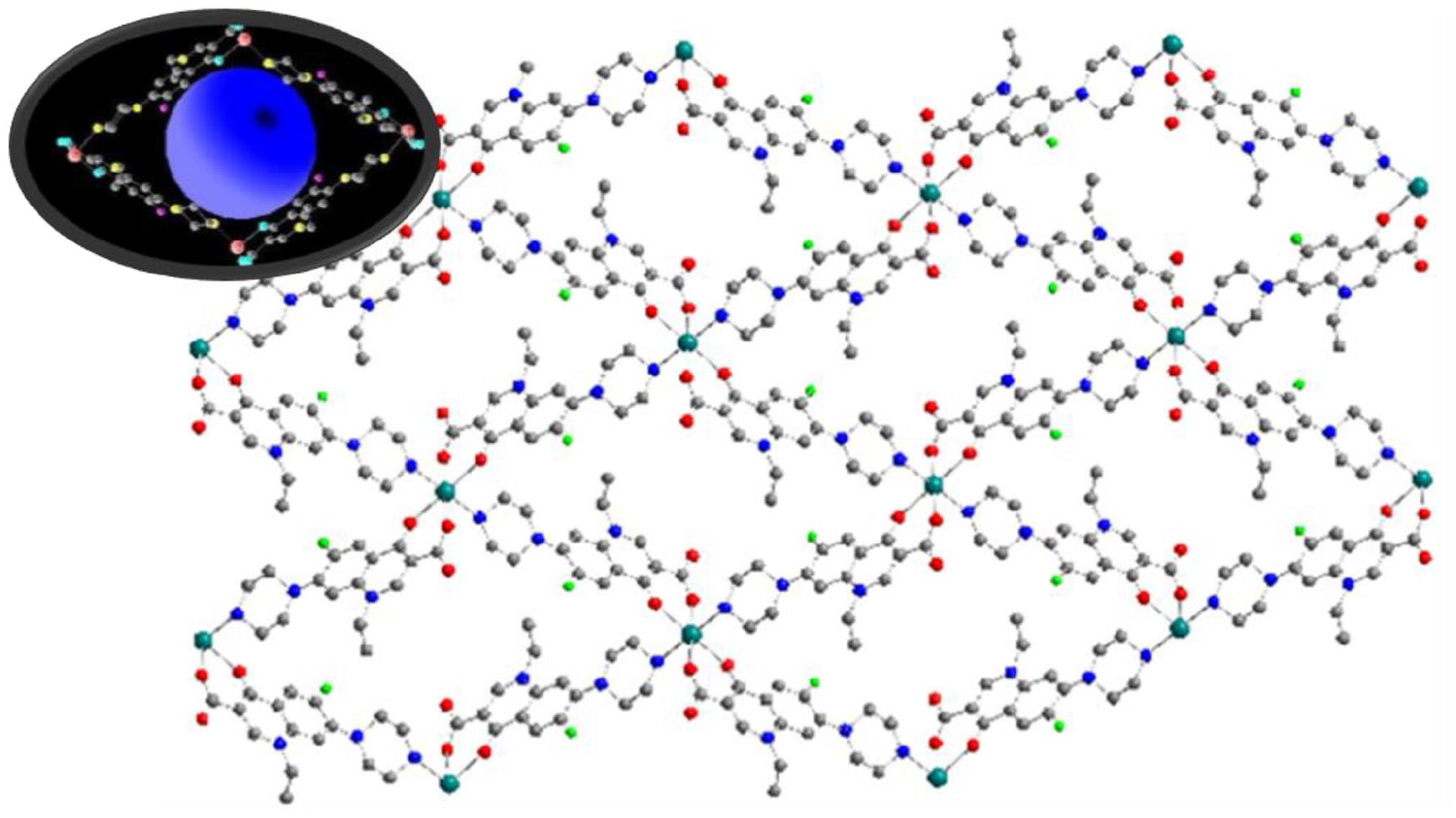
The two-dimensional porous structure of complex
[Zn(HNOR)2·2H2O] (3)
Complex
The π–π stacking interaction is an important spatial arrangement of aromatic compounds, which occurs in the ligands and symmetry two adjacent equivalent ligands, π–π stacking interaction is weak interaction between aromatic rings (Table 3). Through such π–π stacking interactions, complex
Relative parameters of intermolecular π–π stacking in complexes

Defined rings and symbol explanations of complex
Symmetry codes: i: 1 − x, −y, 2 − z.
Defined rings and symbol explanations of complex
Symmetry codes: i: 2 − x, −y, 1 − z.

The two-dimensional porous structure of complex
[Co(H2NOR)2·2H2O]n·2nNO3 (4)
Complex
Two-dimensional layered structure of complex
Powder X-ray diffraction and infrared spectra
The crystallinity of the materials was analyzed by X-ray powder diffraction (XRD). The samples were milled and mounted on the sampling plate. The XRD patterns were recorded on an Advance Bruker X-ray diffractometer with the Cu Kα radiation (λ = 1.5406 Å, 40 kV, 40 mA), a step size of 0.02°, and a scanning rate of 1 step/0.8 s in the range of 10°–80°. Figure 7(a) shows the complexes
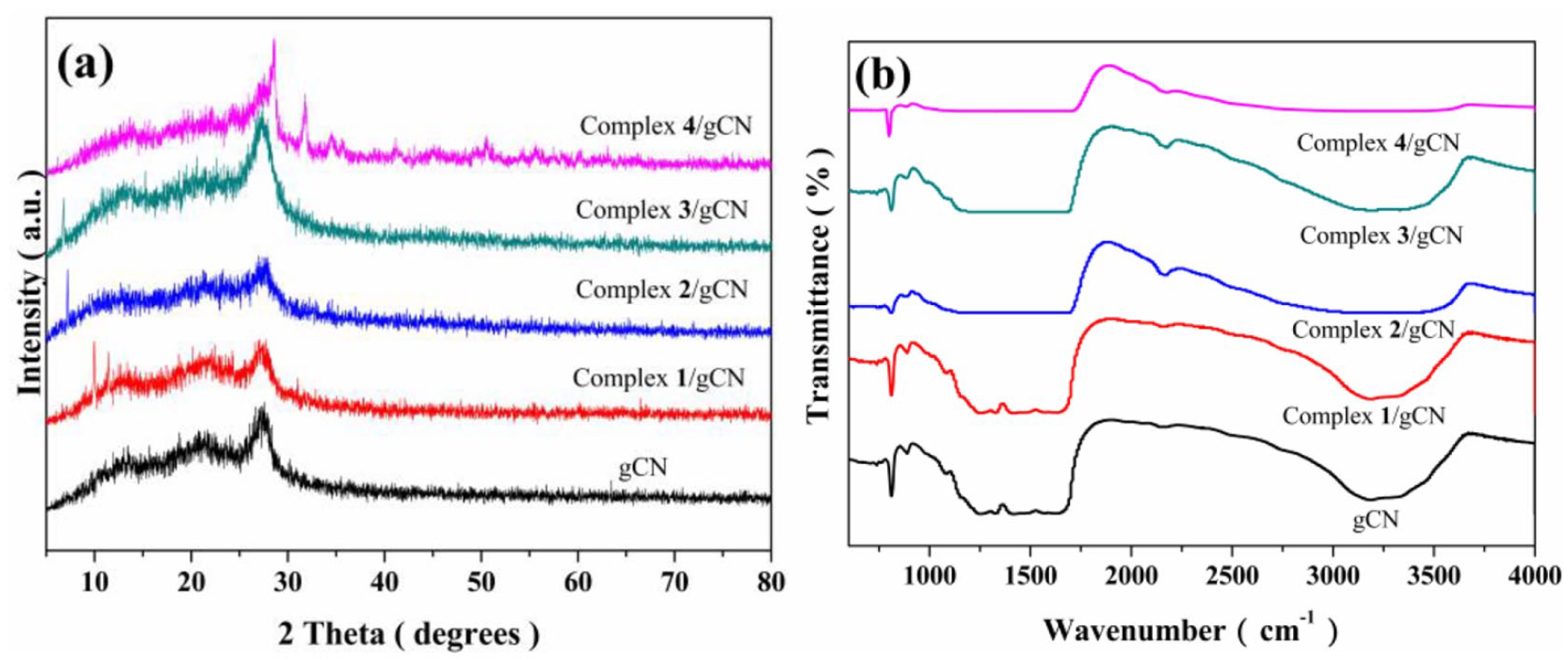
XRD patterns and IR spectra of gCN and complexes 1
In addition, the bonding vibrations of the materials were observed by infrared (IR). The samples were mixed with KBr before molding to afford a round pill with 99 wt% of KBr. The IR spectra were recorded on a Nicolet 170SX spectrometer, with wavenumbers in the range of 4000–400 cm−1, and with a step size of 1 cm−1. The interactions between the complexes
UV–vis diffuse reflection Solid spectra and the Tauc’s plots
The influence of the addition of the complex on the optical properties of the as-prepared materials was observed via UV-Vis diffuse reflection solid (DRS). UV–vis DRS spectra of gCN and complexes 1–4/gCN shown in Figure 8(a), due to the modification of the complexes, all the composite photocatalysts showed a wider visible-light absorption range compared to gCN, leading to enhanced light energy absorption. Moreover, the direct band gaps (Eg) of complexes

UV-Vis DRS spectra and Tauc’s plots of gCN and complexes
Migration and separation efficiency of photo-generated charges
To evaluate the separation of e− and h+ in the materials prepared, photoluminescence (PL) and electrochemical impedance spectroscopy (EIS) were recorded. Figure 9(a) shows the PL spectra of the composite photocatalysts under light excitation at a wavelength of 350 nm. It has been reported that a lower PL intensity is specific to a lower rate of recombination of photo-generated electron–hole pairs resulting in higher photocatalytic performance.22,23 The fluorescence intensity of complex

PL spectra and EIS spectra of complexes
The electron–hole pair recombination of the materials was further investigated by EIS spectra. Typically, a material will have a higher current and a smaller resistance, in other words, at the interface and the surface if the frequency of the EIS Nyquist semicircles becomes smaller.24,25 Figure 9(b) indicates that the arc radii of complexes
Photocatalytic hydrogen production
Photocatalytic hydrogen production experiments were performed using a Labsolar-6A photocatalytic apparatus connected to a gas chromatograph. The samples (0.05 g) were dispersed in deionized water with triethanolamine (20 vol%, 100 mL) and Pt (H2PtCl6·6H2O) (3.0 wt%). The reactor was then placed under a Xenon lamp (300 W, λ ⩾ 420 nm) for testing. During the photocatalytic process, the temperature of the reaction system was kept at 15 °C by circulating water and the resulting gas was analyzed by gas chromatography using high purity nitrogen as the carrier gas. As can be seen from Figure 10, complexes

Time courses of the photocatalytic hydrogen production using complexes

The photocatalytic rate of hydrogen production.
Conclusion
In summary, we have prepared four complexes, [Ni(HNOR)2]n·5nH2O (
Experimental
Materials
Analytical grade urea, norfloxacin, NiCl2·6H2O, CdSO4·8H2O, ZnSO4·7H2O, Co(NO3)2·6H2O, and sodium hydroxide were obtained from Shanghai Tengzhun Biotechnology Corporation (Shanghai, China), and were used as received.
Instruments and measurements
The IR spectra were recorded on a Nicolet FTIR 170SX spectrometer. Elemental analysis was carried out with a Perkin-Elmer 240C analyzer. Photocatalytic hydrogen production was accomplished using a Labsolar-6A. PL was recorded on an F4500 PL detector. EIS was performed on a CHI760E electrochemical workstation.
Syntheses and measurements
Synthesis of [Ni(HNOR)2]n·5nH2O (1)
A mixture of NiCl2·6H2O (0.2377 g, 1.0 mmol) and norfloxacin (0.6387 g, 2.0 mmol) in H2O (20 mL) at room temperature was stirred thoroughly while stirring. The four complexes all use the ligand–metal ratio of 2:1. When the mixture and the pH was adjusted to about 8.0 with NaOH. The obtained cloudy solution was transferred into a 30-mL Teflon-lined autoclave under autogenous pressure and heated at 110 °C for 6 days (Scheme 1). After cooling to room temperature, the resulting green block-shaped crystals of complex

Preparation of complexes
Synthesis of [Cd(HNOR)2]n (2)
The preparation of complex
Synthesis of [Zn(HNOR)2·2H2O] (3)
Complex
Synthesis of [Co(H2NOR)2·2H2O]n·2nNO3 (4)
Complex
Synthesis of gCN and the composite photocatalysts
gCN was obtained by calcining urea in a muffle furnace at 450 °C for 2 h (5 °C/min) in ambient air and the composite photocatalysts were obtained by calcining urea and [Ni(HNOR)2]n·5nH2O (
Crystal structure determination and physical measurements
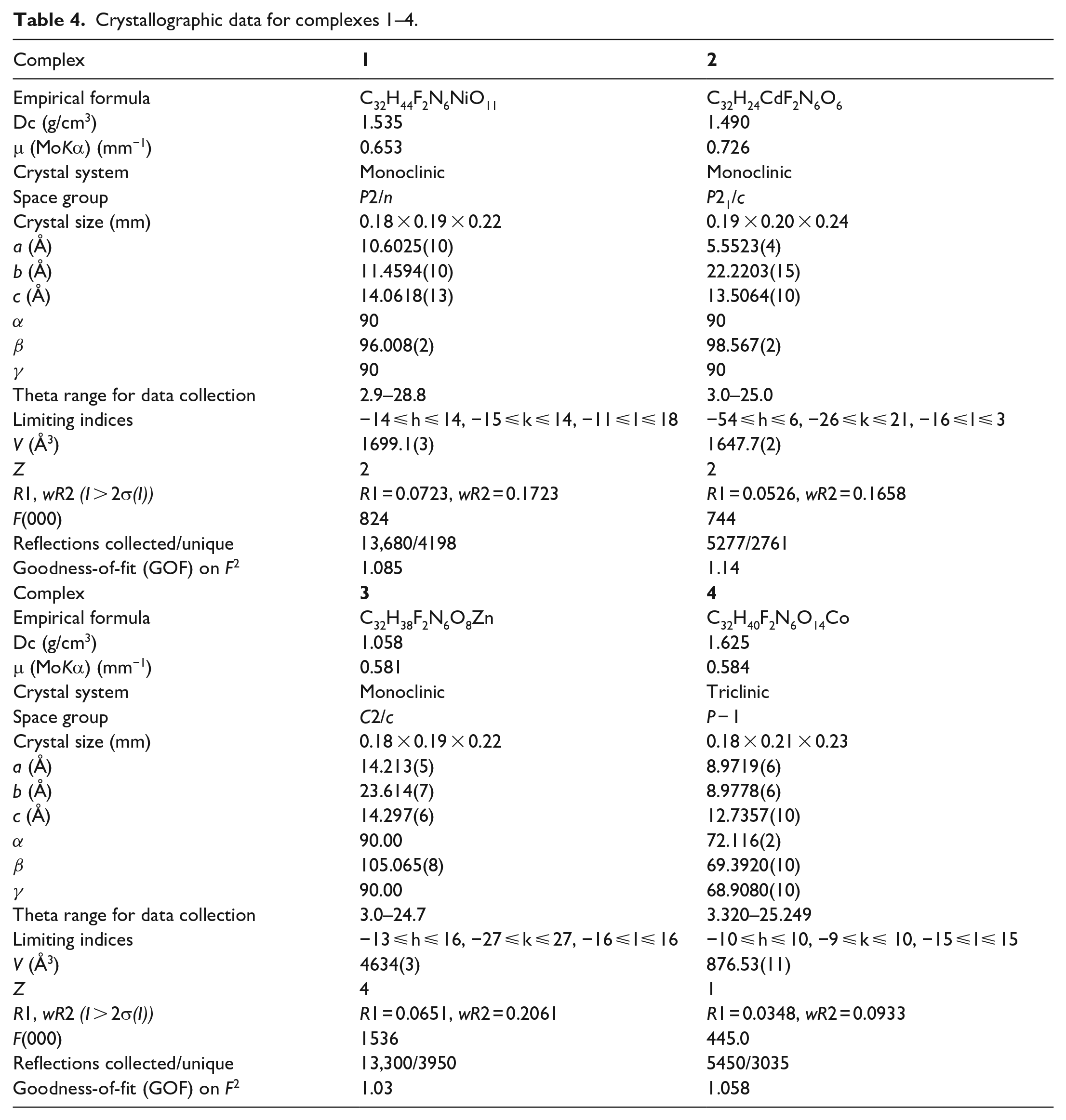
The crystallographic data for these four complexes were collected on a Bruker SMART X-ray diffractometer with a CCD area detector using a graphite-monochromated MoKa radiation (λ = 0.71073 Å). The structures of the complexes were solved by direct and subsequent Fourier and differential Fourier synthesis methods, respectively, and then by full-matrix least-squares techniques on F2 using the program SHELXL. The crystallographic data for the four complexes are summarized in Table 4.
Crystallographic data for complexes 1–4.
Footnotes
Author contributions
Y.H. contributed to the material characterization, research, and writing. J.W. contributed to synthesis of complexes and complex/carbon nitride photocatalysts. J.Z. contributed to the structural analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank the Education Department of Jilin Province (JJKH20210541KJ) and the Innovation and Entrepreneurship Training Program for college students of Tonghua Normal University (s202010202071).
Availability of data and material
CCDC 1984305-1984307 and 2106398 contain the supplementary crystallographic data for the complexes [Ni(HNOR)2]n·5nH2O (
