Abstract
Fifty-five 8-hydroxyquinoline (8-HQ) derivatives are synthesized and the corresponding aluminum(III), cadmium(II), copper(II), and zinc(II) metal complexes are prepared and their fluorescent activities are evaluated. The results indicate that the aluminum complexes have the best fluorescence properties, followed by zinc and cadmium complexes, while almost no fluorescence is observed with the copper complexes. The relationship between the fluorescence properties and complex structure is summarized and a predictive three-dimensional quantitative structure–property relationship model is established using the multifit molecular alignment rule of Sybyl program. With the introduction of groups at the C-2 position or electron-withdrawing groups to the 8-hydroxyquinoline framework, fluorescence wavelength blue shifts are observed with the zinc, aluminum, and cadmium complexes. At the same time, a red shift of the fluorescence emission wavelength is detected for the aluminum and zinc complexes when C-5 of 8-hydroxyquinoline was substituted with halogens or a methyl group. Moreover, the zinc, cadmium, and aluminum complexes with 2,4-dimethyl substituents on the 8-hydroxyquinoline all show good fluorescence properties. The highest occupied molecular orbital and lowest unoccupied molecular orbital energies of the complexes are also calculated and the fluorescence properties of the metal complexes are analyzed from the viewpoint of molecular orbitals.
Keywords

Introduction
Organic light-emitting diodes (OLEDs) are highly sought-after by researchers in the monitor and lighting fields, and developing new luminescent materials with good properties has become an important area of research.1–5 8-Hydroxyquinoline, which possesses a heterocyclic conjugated planar structure, was used in multiple areas because of its chelating performance, luminescence properties, and coordination abilities. 6 Despite many efforts having been made on the preparation of 8-hydroxyquinoline derivatives and their metal complexes,7–12 only a limited number of comparative studies are available on the influence of different substituents on the 8-hydroxyquinoline framework and different metals on the fluorescence properties of the corresponding complexes. Recently, we reported the synthesis of 55 different substituted 8-hydroxyquinolines (Figure 1) along with preliminary studies on the fluorescent properties of their zinc complexes. 13

The structures of the previously prepared 8-hydroxyquinoline derivatives.
For a comparative study on the fluorescence properties of quinolines with different substituents and different central metals, a series of 8-hydroxyquinoline complexes with aluminum(III), cadmium(II), zinc(II), and copper(II) was prepared using the substituted 8-hydroxyquinolines shown above as ligands (Scheme 1).

Preparation of substituted 8-hydroxyquinoline Al(III), Cu(II), Cd(II), and Zn(II) complexes.
The luminescence properties of the aluminum(III), cadmium(II), zinc(II), and copper(II) complexes were determined by fluorescence spectroscopy. The experimental results demonstrated that different substituents on the quinoline ring and different central metals had diverse influences on the fluorescence properties.
To our knowledge, there are no previous studies on the theoretical and experimental fluorescence properties of metal complexes of 8-hydroxyquinoline with different substituents. In the present work, a combined experimental and theoretical method was used to investigate the fluorescence spectral properties of 8-hydroxyquinoline derivatives, and provides a useful tool in designing metal complexes of 8-hydroxyquinoline by correlating theoretical optical properties with their experimental values.
Results and discussion
In general, the fluorescence intensities of the 8-hydroxyquinoline aluminum complexes were the highest. Those of the zinc and cadmium complexes were very similar and weaker than those of the corresponding aluminum complexes. Fluorescence was barely observed for the copper complexes of the 8-hydroxyquinoline.
The fluorescence emissions of the 55 aluminum compounds were measured, 5 of which showed better fluorescence intensity than 8-hydroxyquinoline with fluorescence quantum yields (ɸf) above 0.171. Among the 22 complexes in which fluorescence was observed, the emission wavelengths of eight compounds were red-shifted and the wavelengths of 14 compounds were blue-shifted. Compared with aluminum complexes, nine zinc complexes exhibited improved fluorescence intensities in comparison to 8-hydroxyquinoline. Among the 16 zinc complexes in which fluorescence was observed, the emission wavelengths of three compounds were red-shifted and those of the remaining 13 compounds were blue-shifted. The fluorescence properties of the cadmium complexes were very similar to zinc, albeit the fluorescence intensity was weaker. There were eight cadmium complexes which exhibited improved fluorescence intensity compared to 8-hydroxyquinoline, and the emission wavelengths of them all were blue-shifted. Fluorescence was barely observed with the copper complexes, even with 8-hydroxyquinoline (8-HQ). The influence of different central metals and different substituents on the 8-hydroxyquinoline on the fluorescence emission wavelengths and intensities are discussed below.
The effects of substituents on 8-hydroxyquinoline on the fluorescence intensity
The fluorescence data of the aluminum complexes with
The fluorescence and UV data of the aluminum complexes with

8-HQ: 8-hydroxyquinoline.
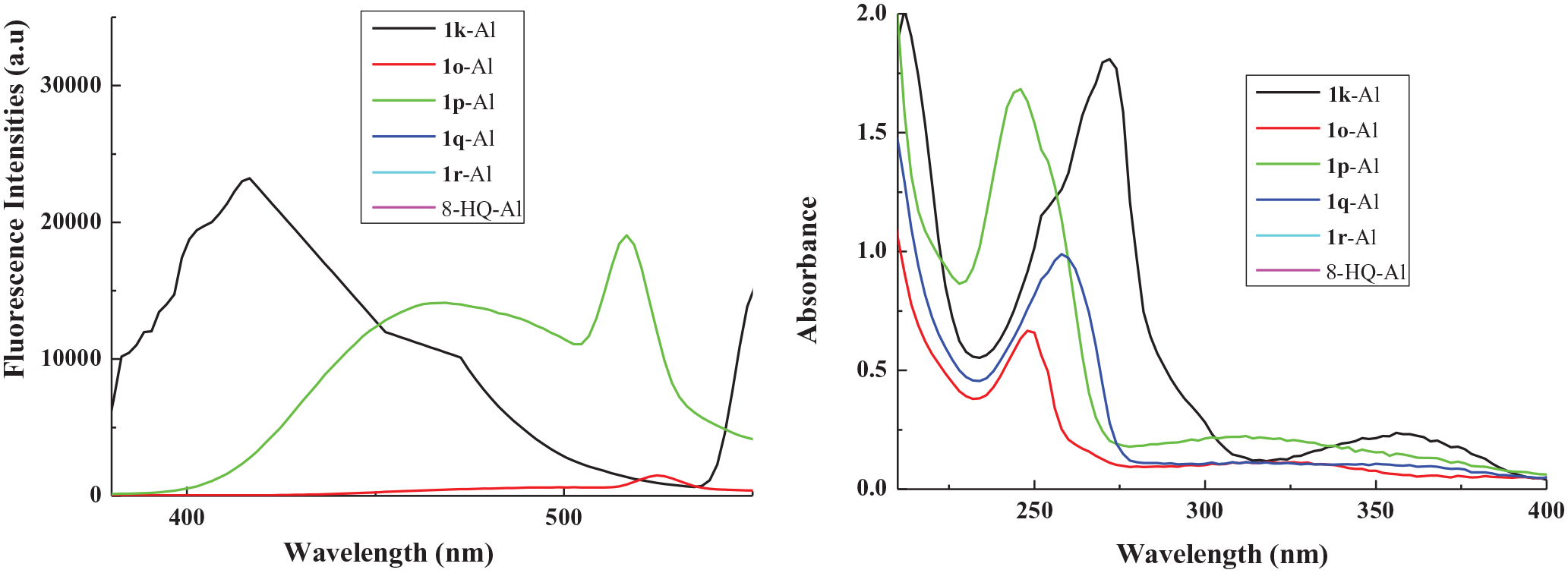
Fluorescence (left) and UV (right) spectra of aluminum complexes with
The fluorescence intensities of the aluminum complexes were
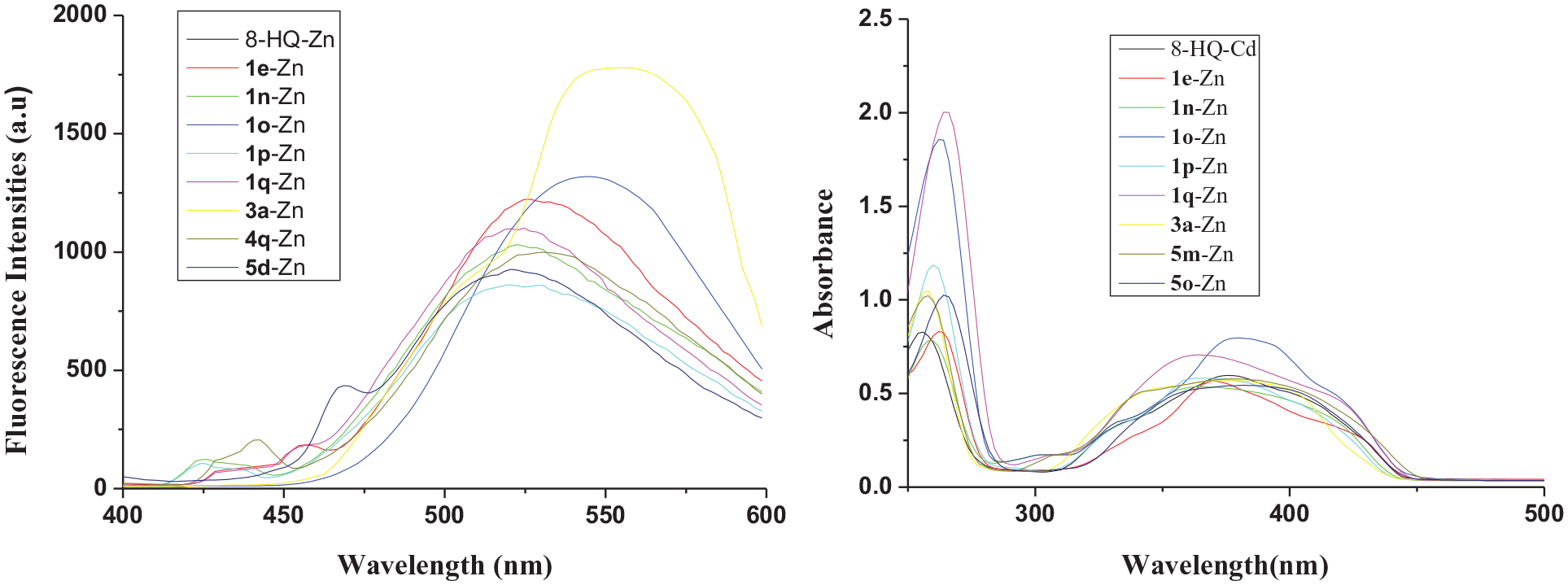
There were nine 8-HQ derivatives of zinc complexes possessing high fluorescence intensities and these were also characterized by UV spectroscopy analysis (Figure 3). The fluorescence spectra and UV spectra of their zinc complexes are shown below.

Fluorescence (left) and UV (right) spectra of 8-hydroxyquinoline derivative zinc complexes with high fluorescence intensities in MeOH at 2 × 10−5 mol L−1.
Furthermore, the data in Table 2 reveal that the ɸf values of complexes
The fluorescence and UV data of zinc complexes with

8-HQ: 8-hydroxyquinoline.
There were eight fluorescent 8-hydroxyquinoline cadmium complexes that were characterized by UV spectroscopic analysis using the 8-hydroxyquinolone cadmium complex for comparison (Figure 4). The fluorescence and UV spectra of the cadmium complexes are as follows.
Fluorescence (left) and UV (right) spectra of 8-hydroxyquinoline derivative cadmium complexes with high fluorescence intensity in MeOH at 2 × 10−5 mol L−1.
The fluorescence intensities of the cadmium complexes of 8-hydroxyquinoline derivatives and 8-HQ were much lower than those of the corresponding aluminum and zinc complexes (Table 3). The complexes of
The fluorescence and UV data of cadmium complexes of

8-HQ: 8-hydroxyquinoline.
As almost all copper complexes of the 8-hydroxyquinoline derivatives were not fluorescence active, no further studies are discussed herein.
There are three compounds—
Some zinc and cadmium complexes—such as those of
However, with compound
Three-dimensional quantitative structure–property relationship of the metal complexes
Three-dimensional quantitative structure–property relationship (3D-QSPR) analysis is widely applied due to its well-established predictive power.14–16 In particular, the comparative molecular field analysis (CoMFA) is an effective method based on statistical techniques. 17
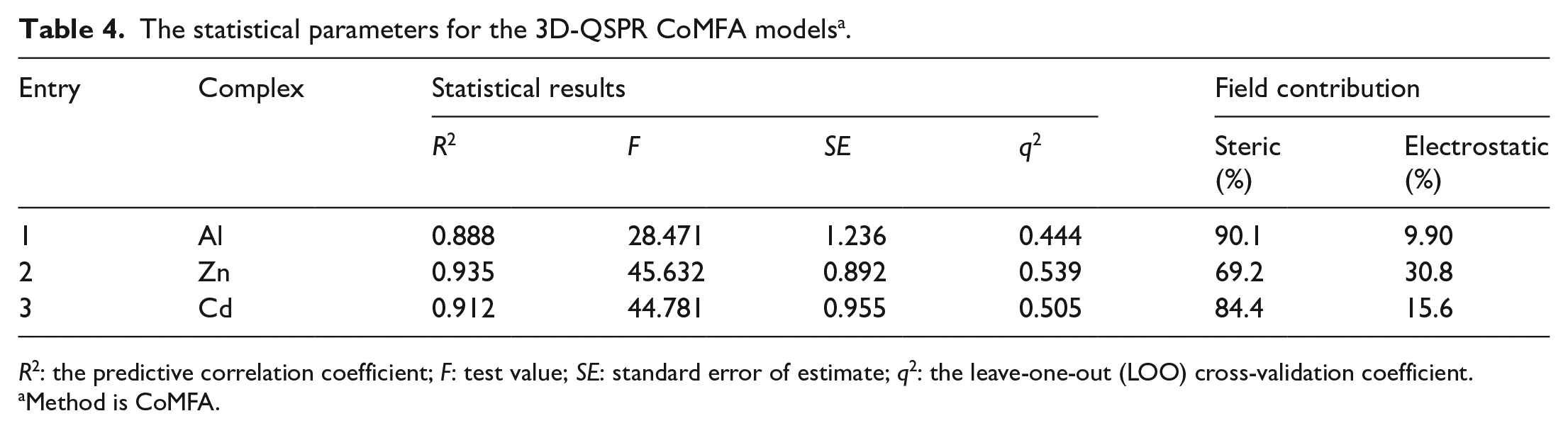
In order to further modify the molecule, 3D-QSPR studies were applied on the ɸf using the CoMFA protocol for the first time. The data-based fitting procedure was finally adopted through careful comparison. Each metal complex, in which fluorescence could be observed, was superimposed to the template based on the common substructure of aluminum(III), cadmium(II), zinc(II), and copper(II) complexes of 8-hydroxyquinoline. The aligned molecules are illustrated in Figure 5, and the statistical parameters are listed in Table 4.

3D views of metal complex congeners by RMSD-based fitting.
The statistical parameters for the 3D-QSPR CoMFA models a .
Method is CoMFA.
For metal complexes, as shown in Table 4, better predictions are obtained with 3D-QSPR/CoMFA for the absorption properties. The model has a high
The contribution of the steric field of aluminum(III), zinc(II), and cadmium(II) is 90.1% (Table 4, entry 1), 69.2% (Table 4, entry 2), and 84.4% (Table 4, entry 3) and the electrostatic field of aluminum(III), zinc(II), and cadmium(II) is 9.90% (Table 4, entry 1), 30.8% (Table 4, entry 2), and 15.6% (Table 4, entry 3), respectively. This showed that the steric hindrance has a significant influence on the fluorescence properties, especially the fluorescence properties of aluminum complexes which are almost determined by the steric factors. The plots of the predicted versus actual ɸf value for the QSPR/CoMFA model are displayed in Figure 6, which shows a good correlation. The electrostatic field is the main factor influencing the maximum absorption.

Plots of experimental data versus the corresponding predicted data from the QSPR/CoMFA models.
To view the field effect on the target property, CoMFA contour maps were generated and shown in Figure 7. For steric fields, green means that a bulky group is favorable and yellow indicates that a bulky group is unfavorable. Similarly, blue means that an electropositive charge is favorable and red indicates that an electronegative charge is favorable.

Stereo views of CoMFA steric field and electrostatic field of metal complexes.
For example, for the aluminum complexes, the steric field of the complexes has green contours (75.80%) indicating sterically favored regions for fluorescence properties, and the yellow contours (24.20%) highlight the sterically unfavorable regions. The electrostatic field has blue contours (13.23%) representing electropositively preferred regions to better fluorescence performance, and red contours (86.77%) indicate regions where more electronegative substituents are favored to increase fluorescence. The data for the CoMFA steric field and electrostatic field of the cadmium and zinc complexes of 8-hydroxyquinoline are also shown in Figure 7.
The substituents on 8-hydroxyquinoline resulted in a blue shift of λ emission examples (λem)
The
Data for the aluminum, cadmium, and zinc complexes both with blue shifted of
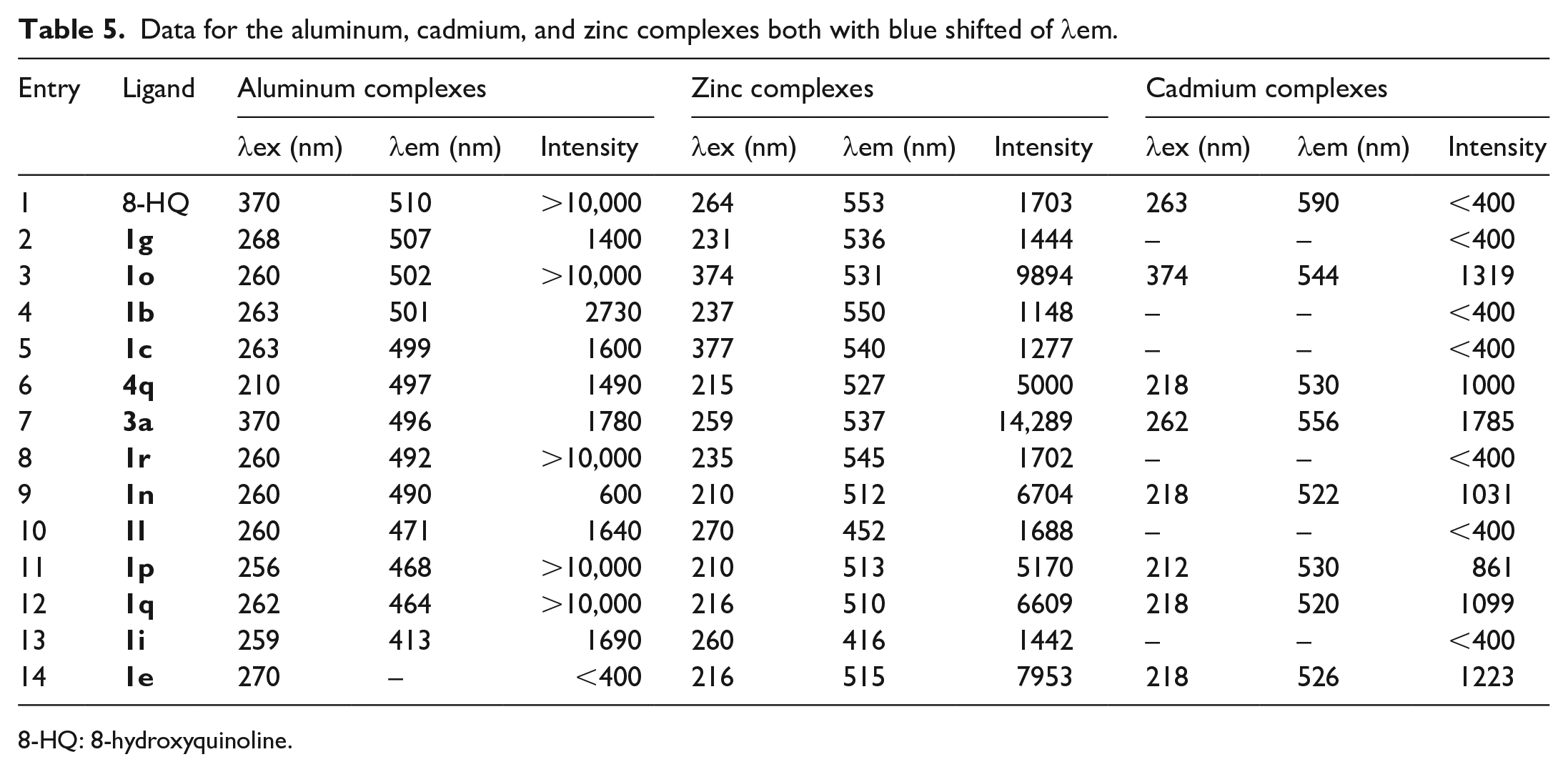
8-HQ: 8-hydroxyquinoline.
The blue shift of complex
The maximum emission indicated that the electron-donating and electron-withdrawing nature of the substituents on the 8-HQ and the emission wavelengths had a good correlation.
18
There were two factors causing the blue shifts of the aluminum, cadmium, and zinc complexes, which were the introduction of electron-donating groups on the pyridine ring and electron-withdrawing groups on the benzene ring.19,20 The addition of substituents at C-2 of the pyridine ring, such as in ligands
In the same way, the
The substituents on 8-hydroxyquinoline resulted in a red shift of λ emission examples (λem)
Since no red shift was observed in the cadmium complexes, only zinc and aluminum complexes were compared and discussed. The emission wavelength of nine aluminum complexes was red-shifted, while three of the nine zinc complexes were red-shifted. The wavelengths of the aluminum and zinc complexes of
Data for the aluminum and zinc complexes both red-shifted
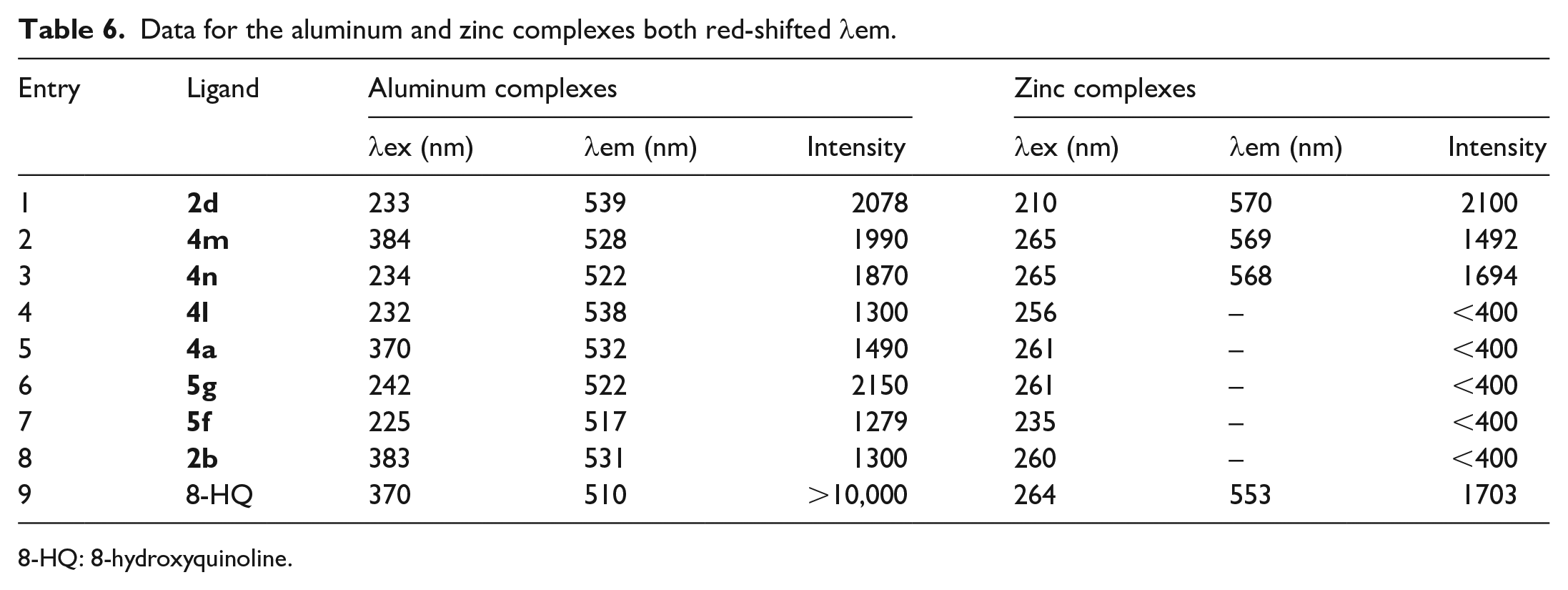
8-HQ: 8-hydroxyquinoline.
When halogens or methyl groups were introduced at the C-5 position of 8-hydroxyquinoline, the fluorescence emission wavelength of the corresponding metal aluminum and zinc complexes was red-shifted. Compared with the
The fluorescence wavelengths of the aluminum complexes of
Analysis of the effect of the structure on the fluorescence properties in terms of molecular orbitals
There are three compounds—
Energies of the molecular orbitals and the ɸf values of

The Hartree was used as the energy unit; energies in subsequent tables are also in Hartrees.

The HOMOs and LUMOs of metal complexes
The effects of the different energies of the molecular orbitals on the fluorescence properties of the corresponding aluminum(III), cadmium(II), copper(II), and zinc(II) metal complexes are summarized as follows: the electron density of the pyridine ring was improved by the introduction of methyl groups at C-2 and C-4 of 8-HQ and would thus change the LUMO and HOMO values of the complexes.19,21 At the same time, the
Another phenomenon was that the
The addition of substituents at the C-2 position of the pyridine ring, such as in ligands
However, the regularity of the wavelength changing with the Egap is not as good as that of ɸf. In order to analyze the cause of the blue shift of the metal complexes from the viewpoint of molecular orbitals, the HOMOs and LUMOs of complexes with similar structures were calculated and compared, and the results are shown in Figure 9 and Table 8.

The HOMOs and LUMOs of metal complexes
Energies of molecular orbitals and the

8-HQ: 8-hydroxyquinoline.
The ligands, which possess different electron-withdrawing groups, such as sulfonic acid, could induce deflection of the electron cloud, which leads to an increase of the HOMO value of the quinoline benzene ring. As a result, the energy gap between the HOMO and LUMO is increased, and the fluorescence emission wavelength of compounds had a blue shift, such as in compounds
The luminescence of the 8-hydroxyquinoline metal complex was caused by the electron π–π* transition from the HOMO of the phenol ring to the LUMO located on the pyridyl ring.
23
The highest electron density of the HOMO of the 8-hydroxyquinoline metal complex was at the phenol oxygen and the C-5, C-7, and C-8 positions. Thus, it can be predicted that electron-withdrawing groups or electron-donating groups at these positions would lead to blue or red shifts of the absorption and fluorescence spectra.
24
There are three compounds,
Energies of the molecular orbitals and the
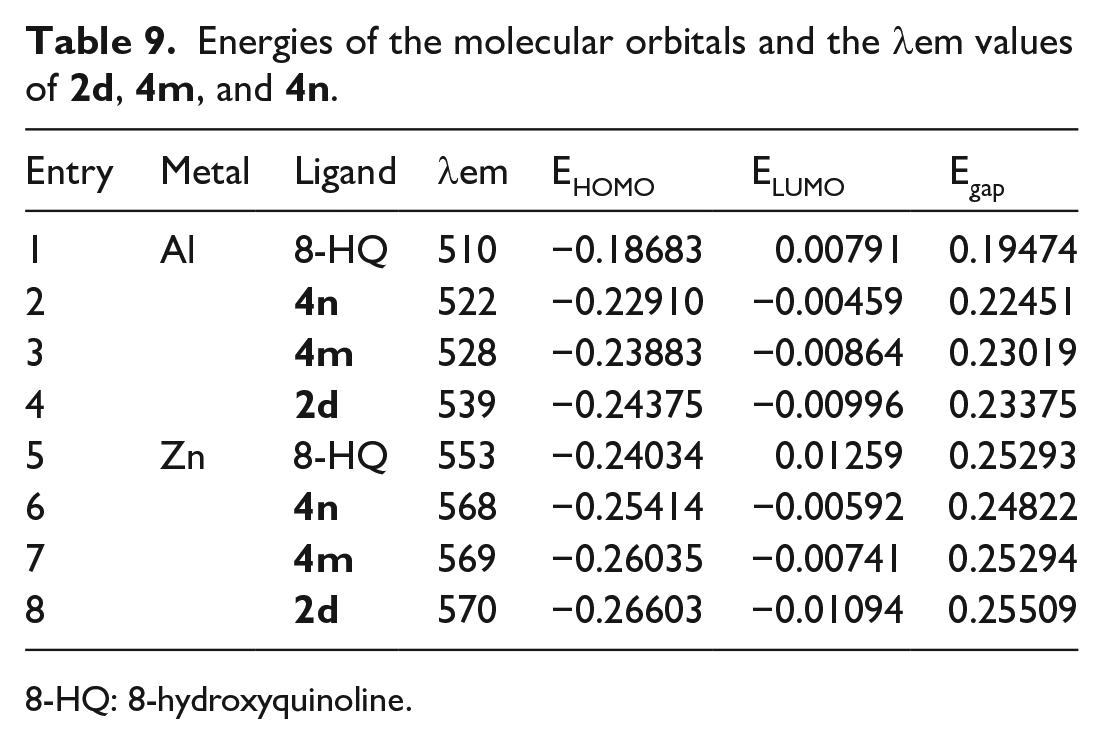
8-HQ: 8-hydroxyquinoline.

The HOMO and LUMOs of metal complexes
The Egap of most of the red-shifted complexes was also larger than that of 8-hydroxyquinoline, and was irregular with the wavelength changes. However, we found that the lower the HOMO value of the complexes, the greater the red-shifted fluorescence emission wavelength. For example, the HOMO of the aluminum complexes decreases from
When the C-5 position of the 8-hydroxyquinoline benzene ring was substituted with a halogen, such as in
Conclusion
Fifty-five 8-hydroxyquinoline (8-HQ) derivatives were synthesized and the corresponding aluminum(III), cadmium(II), copper(II), and zinc(II) metal complexes were prepared and their fluorescent activities were evaluated. The aluminum complexes had the best fluorescence properties, followed by zinc. The fluorescence can also be observed in cadmium complexes, while almost no fluorescence was observed in copper complexes. The fluorescence properties of the few aluminum, zinc, and cadmium complexes were different. The 3D-QSPR model was proposed using CoMFA based on the molecular simulation, which also exhibited good stability and good prediction ability. The optimum models were statistically significant with cross-validated coefficients (
The relationship between the fluorescence properties and the complex structure has been summarized. The effects of the structure and energies of the molecular orbitals on the fluorescence properties were analyzed. The effect of the ligand substituents on the fluorescence properties of 8-hydroxyquinoline zinc, cadmium, and aluminum complexes shows some common points: first, the introduction of a halogen or a methyl group at C-5 of the 8-hydroxyquinoline ring results in a red shift of the fluorescence emission wavelength of aluminum complexes due to the increased conjugation effect and the decreased EHOMO value. Second, when electron-withdrawing groups were introduced on the 8-hydroxyquinoline ring, the conjugation effect of the quinoline ring was decreased and the Egap value increased, which led to a blue shift of the fluorescence emission wavelength and a weak fluorescence intensity. Third, a stronger intensity of the fluorescence would be achieved by introducing a methyl groups at position of 2 and 4 of the 8-hydroxyquinoline ring, which led to steric issues and a bigger gap between the HOMO and LUMO.
Experimental
Details of the synthesis and characterization of ligands are provided in the supporting material. The procedures for the preparation of the zinc complexes and recording the fluorescence measurements are consistent with those of previous our report. 13
General procedure for the preparation of the aluminum complexes
The 8-hydroxyquinoline ligand (1.0 mmol) was added to anhydrous methanol at room temperature, and then the temperature was raised to 70 °C. An aqueous solution of aluminum sulfate (1.0 mmol) was slowly added dropwise, and the mixture was maintained at 70 °C for 20 h. The pH of the mixture was adjusted with ammonia solution to about 7–8. After stirring for 4 h, the obtained precipitate was filtered and recrystallized from anhydrous methanol to give the pure product.
General procedure for the preparation of the cadmium complexes
A mixture of CdNO3 (0.5 mol), 8-hydroxyquinoline ligand (1.0 mmol), water, and MeOH in a capped vial was heated at 80 °C for 1 day. Colored block–like crystals were collected by filtration, washed with ether, and dried to give the desired product.
General procedure for the preparation of the copper complexes
A pale green solution of CuCl2·2H2O (0.5 mmol) dissolved in methanol was added slowly to a solution of the 8-hydroxyquinoline ligand (1.0 mmol) in methanol. After stirring for 30 min, the solid was filtered, washed with ether, and dried to give the desired product.
Photoluminescence quantum yields
Quantum yields of fluorescence (ɸf) can be measured in two different ways: relative to a fluorescent standard material with a known ɸf or as an absolute quantity. The measurement of relative fluorescence quantum yields has exclusively been done by optical methods, most prominently using a spectrofluorometer and the comparative method as proposed by Parker and Rees. 25 In this paper, the ɸf was calculated by a comparative method using the following equation6,26,27

In this experiment,
Computational details
All computations of optimized geometry were carried out using the Gaussian 09 28 computer software package. The electronic descriptors were obtained from a single-point calculation at the B3LYP/6-311+g (d) level. The structural parameters—including the energy of highest occupied molecular orbital (EHOMO), the energy of the lowest unoccupied molecular orbital (ELUMO), and the energy difference between the LUMO and HOMO (Egap)—were also calculated. 3D-QSPR studies on the metal complexes were performed using the literature procedure reported by us using CoMFA performed on the Sybyl 8.0 package. 29
Supplemental Material
sj-pdf-1-chl-10.1177_1747519820973601 – Supplemental material for Theoretical and experimental studies on the fluorescence properties of aluminum(III), cadmium(II), zinc(II), and copper(II) complexes of substituted 8-hydroxyquinoline
Supplemental material, sj-pdf-1-chl-10.1177_1747519820973601 for Theoretical and experimental studies on the fluorescence properties of aluminum(III), cadmium(II), zinc(II), and copper(II) complexes of substituted 8-hydroxyquinoline by Zhang Yuanyuan, Cheng Hongrui, Sun Qingrong, Chen Hongli, Yang Weiqing and Ma Menglin in Journal of Chemical Research
Supplemental Material
sj-pdf-2-chl-10.1177_1747519820973601 – Supplemental material for Theoretical and experimental studies on the fluorescence properties of aluminum(III), cadmium(II), zinc(II), and copper(II) complexes of substituted 8-hydroxyquinoline
Supplemental material, sj-pdf-2-chl-10.1177_1747519820973601 for Theoretical and experimental studies on the fluorescence properties of aluminum(III), cadmium(II), zinc(II), and copper(II) complexes of substituted 8-hydroxyquinoline by Zhang Yuanyuan, Cheng Hongrui, Sun Qingrong, Chen Hongli, Yang Weiqing and Ma Menglin in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Ministry of Education, Chunhui Project of China (no. 192635).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
