Abstract
Silver (Ag) amidinate metal organic decomposition precursors of the type: [Ag2((ArN)2C(H))2] (Ar = 2,6-dimethylphenyl (
Introduction
Silver (Ag) is a multi-faceted element which has found uses in various fields. 1 One of the most common uses of Ag is in the electronics industry where it is utilized for its high conductivity to manufacture electrical circuitry for a wide range of electronic devices. Conventional methods of manufacturing conductive Ag circuits, which consume substantial energy and resources, are rapidly being replaced by techniques that allow low-temperature, and less energy intensive manufacture. 2 These new processes rely largely on Ag inks which are deposited onto various substrates and sintered to obtain conductive Ag tracks. Ag nanoparticle (NP) inks are one category of inks that have garnered substantial interest and are becoming a popular choice for deposition of conductive Ag films. 3 Another prevailing use of Ag is in its nanoparticulate form for the manufacture of optical/biological sensors. The localized surface plasmon resonance of the NPs is susceptible to the NP environment; 4 for Ag NPs, a robust surface plasmon resonance is observed, which is not seen in the spectrum of the bulk metal. 5 This property of the nanoparticulate nature of the metal is utilized in the fabrication of these sensors. In addition, the noble nature of the metal offers stability to these deposited Ag NPs which make the metal competent for this use. Both these applications, however, involve the synthesis of Ag NPs with a narrow size distribution which is challenging.6,7 NP aggregation is a constant hindrance in both these applications which affects the functionality of the final device, while also reducing the shelf life of the inks/complexes. The deposition of NP inks for use in printed electronics also faces issues with printer clogging, high temperature sintering and the need for additives, all of which are detrimental to the final device. 8
Metal-organic decomposition (MOD) inks are an encouraging alternative to the NP counterparts. MOD inks are composed of MOD precursors in a liquid vehicle that is easy to deposit. 9 The precursors consist of the desired oxidized metal ion bound to suitable ligands. When these precursors are sintered (usually thermally), the compound breaks down to elemental metal (in nanoparticulate form) while the ligands decompose as volatile compounds. Often, the ligands aid the reduction process of the central metal ion to its elemental form. 10 These precursors and subsequent inks are easy to synthesize and store, do not face issues with agglomeration or printer clogging and provide a solution to the issues faced during the synthesis and use of NPs and NP inks.
Ag MOD inks, although not as vastly studied as their NP alternative, have been successfully used to deposit conductive metal. Some of the noticeable work in Ag MOD inks for deposition to conductive metal includes that by Walker and Lewis 11 and Knapp et al. 3 who reported the room temperature deposition of the same ink using plasma sintering to obtain conductive Ag on paper. Other notable works include those by Vaseem et al. 12 and Bhat et al. 13 who synthesized an Ag MOD ink with amines and aminoalcohols as ligands that successfully deposited Ag metal for use in electronics.
Other desired properties of an MOD ink include clean decomposition to target material without residual contaminants, 14 good solubility and long shelf life. 15 Additional pre-requisites for an MOD ink are highlighted in our recent review. 16 All these properties largely depend on the precursor design and ligand selection,17,18 making these crucial processes for the design of suitable MOD inks.
An understudied group of ligands for the synthesis of Ag MOD precursors is amidines. A series of Ag-amidinate complexes synthesized for use in catalysis showed impressive thermal properties, with decomposition at ~150 °C, which make them highly promising candidates for MOD precursors. 19 In 2011, Whitehorne et al. 20 were the first to report deposition of Ag using aliphatic amidinates complexed to Ag (Figure 1) via chemical vapour deposition and atomic layer deposition. Thermal gravimetric analysis (TGA) of these compounds showed the reduced metal in the TGA pan with no volatilization occurring. Two potential decomposition pathways were proposed: β-hydrogen elimination and carbodiimide de-insertion.

Silver-amidinate complexes synthesized by Whitehorne et al. 20
MOD inks are often criticized for their low metal weight percentage loading due to the presence of the bulky organic ligands. However, they present the opportunity to allow metal deposition for a range of uses. This work reports the synthesis and characterization of three low metal weight percentage loading Ag-amidinate complexes which have then been used to formulate inks to deposit metallic Ag. Given the low weight percentage of Ag in these inks, single-layer depositions have been used to deposit distinct NPs of Ag for use in sensors, while multiple layer depositions were carried out to yield films with a well-connected network of Ag particles, displaying properties similar to that of bulk Ag.
Results and discussion
Precursor synthesis
The amidine ligands were synthesized via modified literature procedures,21,22 with the 2:1 reaction of 2,6-dimethylphenyl aniline, 2,6-diethylphenyl aniline and 2,6-diisopropylphenyl aniline with triethylorthoformate forming ligands
The stoichiometric reaction of

Synthetic route for the synthesis of Ag(I) amidinate complexes
The 1H NMR spectra of the complexes all show a single ligand environment, indicating that the ligand is conformationally locked upon coordination. Compound

Schematic displaying the overlapping of splitting due to 3JAg-H coupling.
Crystals of

Crystal structure of
TGA
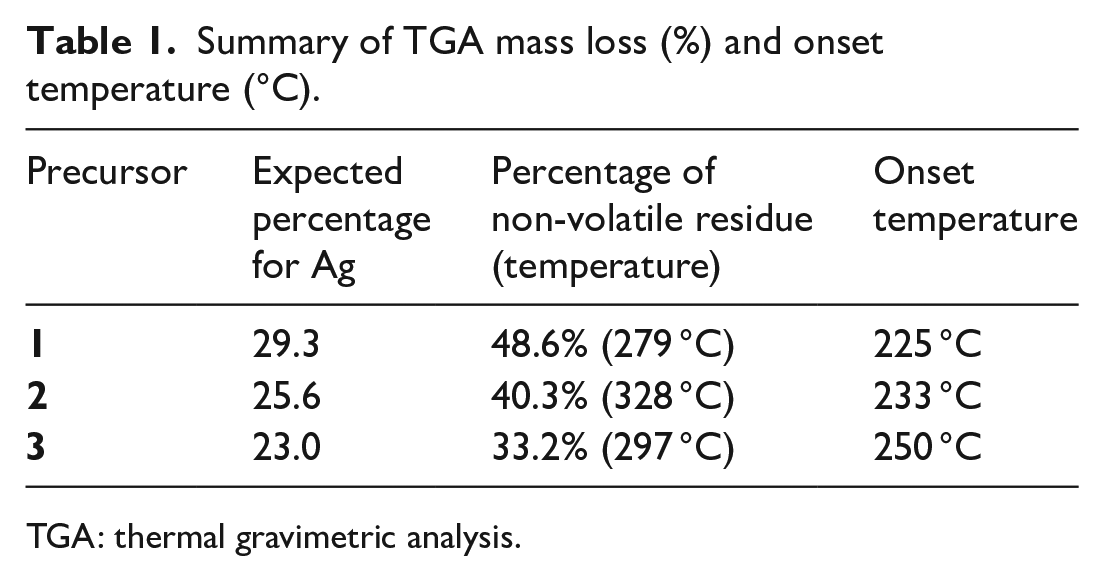
To assess the suitability of precursors

TGA curves and decomposition data for
Summary of TGA mass loss (%) and onset temperature (°C).
TGA: thermal gravimetric analysis.
Ink preparation and thin film deposition
To prepare the Ag MOD inks, the solubility of compounds
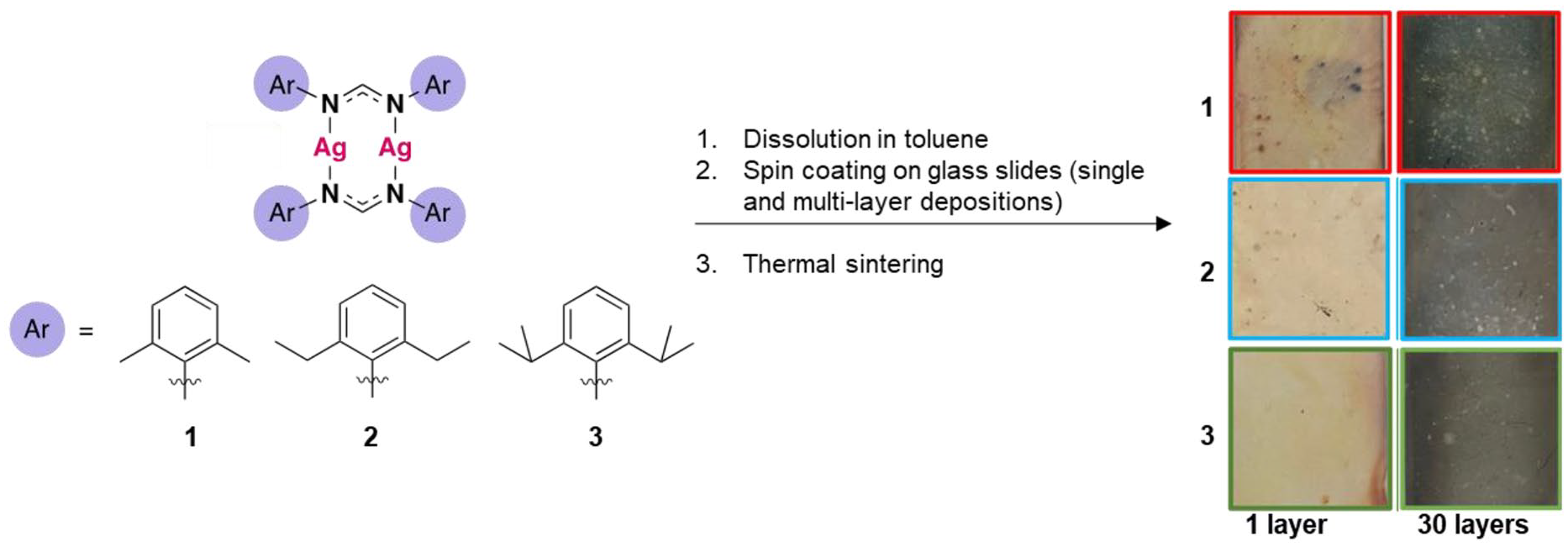
Application of one layer of each ink resulted in transparent deposits, suggesting NP formation rather than aggregated Ag thin film deposition. Scanning electron microscopy (SEM) confirmed the presence of NPs which were not connected, and therefore, the deposits were not conductive (Figure 5). UV-Vis absorption spectra of films deposited from inks

SEM images of single-layer deposits of inks (a)

Absorbance spectra of single-layer deposits obtained using inks

Transmittance spectra for single-layered, 5-layered and 10-layered films from (a)
Following the single-layer depositions of nanoparticulate Ag, multiple layer depositions were undertaken to fabricate Ag films that could be used in electronics. Inks were deposited in single layers and sintered, with films prepared and analysed between 1 and 30 layers (Due to poor solubility at room temperature, inks
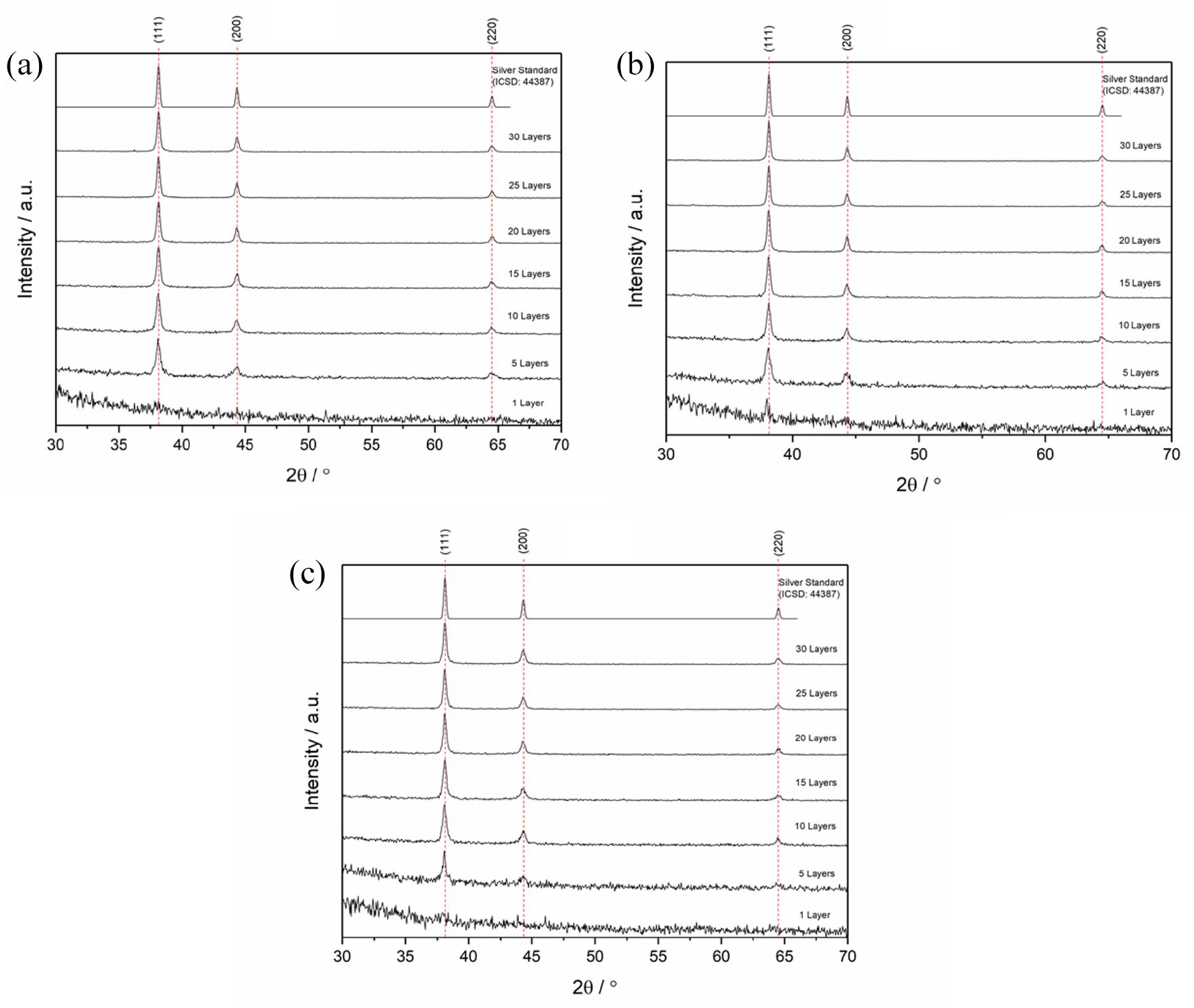
XRD plots for multi-layered Ag films deposited using (a)

XPS data for 30-layered films deposited using
Resistance measurements (detailed in the Supplementary Information) showed a decrease in resistance values as the number of layers was increased, with the lowest detectable resistance of 0.1 Ω obtained from 30-layer depositions of

SEM images of 30-layered films obtained from (a)
Side-on SEMs of these films were obtained (Supplementary Information) and were used to calculate the resistivities of the 30-layered films, with resistivity values of 0.916–1.83 × 10−6, 1.575–3.15 × 10−6 and 7.25–14.5 × 10−6 Ωm from inks

Images of films obtained from inks
Conclusion
This work has shown that Ag-amidinate MOD precursors are a versatile choice for metallic silver deposition: single-layer depositions produce highly transparent (>90% transmittance) Ag NP coatings which are reflective in the UV region, while layering can produce conductive Ag films (ρ = 0.916–1.83 × 10−6 Ωm), which have potential for use in electronic materials. Inks were produced from dissolving MOD precursors of the type: [Ag2((ArN)2C(H))2] (Ar = 2,6-dimethylphenyl (
Experimental
General procedures
All chemicals were obtained from Sigma-Aldrich and used without further purification. All syntheses with Ag(I) compounds were carried out in the absence of light. Nuclear Magnetic Resonance (NMR) data were collected using Bruker Avance III 400 MHz and Avance III 600 MHz instruments. Fourier transformation infra-red (FTIR) spectroscopy data were collected from 500–4000 cm−1 using an ALPHA II FTIR spectrometer. TGA data were collected using a Netzsch STA 449C Jupiter instrument, with graphs being calibrated to 100%. Precursor samples were placed in an Al crucible and measurements were recorded from 20 to 400 °C. Single crystal X-ray diffraction (SCXRD) data were collected using a SuperNova Atlas (Dual) diffractometer using Cu Kα radiation at a wavelength of 1.54184 Å. A suitable crystal was chosen, mounted on a nylon loop and kept at 150 K during data collection (CCDC 2126901). Electrospray ionization (ESI) and electron ionization (EI) mass spectra were attempted using Waters LTC Premier XE ESI Q-TOF mass spectrometer. Elemental analysis (EA) was collected by the Geosciences Department at the University of Edinburgh. Thin films were analysed as deposited.
L 1
H :
Triethyl orthoformate (4.16 mL, 25 mmol), 2,6-dimethylaniline (0.29 mL, 5 mmol) and glacial acetic acid (0.29 mL, 5 mmol) were combined together and heated at 160 °C for 16 h. The resulting solution was dried in vacuo to yield a solid which was dissolved in diethyl ether (130 mL) and neutralized with sodium carbonate (20 mL). The organic layer was extracted, dried with Na2SO4 and the solvent removed in vacuo. The resulting solid was recrystallized from acetone at −18 °C, yielding
L 2
H :
Triethyl orthoformate (3.33 mL, 20 mmol), 2,6-diethylaniline (6.59 mL, 40 mmol) and glacial acetic acid (0.23 mL, 4 mmol) were combined together and heated at 160 °C for 16 h. The resulting mixture was dried in vacuo to yield a solid which was dissolved in diethyl ether (130 mL) and neutralized with sodium carbonate (20 mL). The organic layer was extracted, dried with Na2SO4, and recrystallized at −18 °C, yielding
L 3
H :
Triethyl orthoformate (3.24 mL, 19.5 mmol), 2,6-diisopropylaniline (7.9 mL, 39 mmol) and glacial acetic acid (0.21 mL, 3.9 mmol) were combined together and heated at 160 °C for 16 h. The resulting mixture was dried in vacuo to yield a yellow solid. The solid was dissolved in diethyl ether (150 mL) and neutralized with sodium carbonate (50 mL). The organic layer was extracted, dried with Na2SO4, and recrystallized at −18 °C, yielding
Synthesis of Ag(I) amidinate complexes
1
2
3
Ink preparation and thin film deposition
Physical measurements
Thin films were analysed as deposited. Thin-film XRD patterns were collected using a Bruker AXS D8 discovery Lynx Eye diffractometer. The X-rays were generated using a Cu Kα (40 kV, 40 mA), λ = 1.54056 Å. The incident beam angle was kept at 1° and data were collected between 2θ = 30° and 70° with a step size of 0.05° at 4.0 s/step. XPS measurements were collected using a Thermo Scientific K-alpha photoelectron spectrometer with a monochromatic Al Kα source. These data were calibrated against C(1s) adventitious carbon (284.6 eV) for charge correction using CasaXPS software. UV-Vis data were collected between 200 and 800 nm using UV-2600 UV-Vis spectrophotometer. SEM images were taken using a JEOL 6301 filament scanning electron microscope. To minimize charging effects, these were taken at ranges from 3 to 5 kV. Before the images were taken, a fine layer of gold was coated on the sample to stop charging on the surface. Energy-dispersive X-ray spectroscopy (EDS) was collected on a Philips XL30SEM instrument. Resistance was measured using a Kewtech KT116 Digital Multimeter.
Inks
Supplemental Material
sj-docx-1-chl-10.1177_17475198221075301 – Supplemental material for Deposition of metallic silver from versatile amidinate precursors for use in functional materials
Supplemental material, sj-docx-1-chl-10.1177_17475198221075301 for Deposition of metallic silver from versatile amidinate precursors for use in functional materials by Shreya Mrig, Martha A Jennings, Malavika A Bhide, Clare Bakewell and Caroline E Knapp in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The authors thank the EPSRC (EP/R513143/1) and the Ramsay Memorial Trust for funding.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
