Abstract
A new antibacterial complex C4H15CuN5O7S, which is derived from the non-insulin-dependent diabetes mellitus drug metformin hydrochloride (MH) and cupric sulfate pentahydrate, is prepared and characterized. The complex shows that a three-dimensional supramolecular structure and the intermolecular interactions are analyzed by the Hirshfeld surface analysis. More importantly, compared with the MH free ligand, the present complex shows excellent antimicrobial activity, especially for Gram-negative bacteria.
Keywords

Introduction
The development of metal–drug complexes has attracted significant attention over the past decades. Metal–drug complexes show high diversity of possible structures due to their dynamic nature,1,2 and the wide application of coordination of metal ions to biologically active agents in many fields has inspired numerous scientific studies toward constructing drugs.3,4 Research on metal–drug complexes mainly focuses on anticancer, antimicrobial, and antiviral activity, and altering the biological and pharmacological properties of the free drugs.5–9 Furthermore, the utilization of metal–drug complexes has been considered as a promising method to treat challenging diseases,10,11 including cancer and HIV.12,13 Metformin hydrochloride (MH), as an example of a bioligand containing a biguanide skeleton, is a first-line drug for the treatment of non-insulin-dependent diabetes mellitus (NIDDM) throughout the world.14,15 MH has a relative 50%–60% bioavailability and its short and variable biological half-life is 0.9–2.6 h. 16 The absorption of MH is limited to the upper gastrointestinal (GI) tract. In the pharmaceutical field, it is also found that metal complexes of MH show obvious antimicrobial activity bial activity compared to the free bioligand, due to its good chelating properties because of the close proximity of its two =NH functional groups.
The free drug of MH has two different polymorphs, both of which have monoclinic structures: stable form I and metastable form II.17,18 The structure of MH indicates that it can coordinate with cations or anions. Herein, we are concerned with the attractive properties of MH coordinated with metal ions and the new promising opportunities they may bring in the development of metal–drug complexes. This study constructs a novel three-dimensional (3D) metal–drug complex by self-assembly of MH with hydrated copper sulfate. The crystal structure was analyzed by means of single-crystal X-ray diffraction, and molecular Hirshfeld surface analysis was performed for the purpose of investigating intermolecular interactions. In addition, evaluation of the antimicrobial activity of the title compound has been performed against Gram-positive and Gram-negative bacteria (e.g. Staphylococcus aureus, Escherichia coli, and Pseudomonas aeruginosa).
Results and discussion
Description of the crystal structure of complex 1
Single-crystal X-ray diffraction analysis revealed that complex

Crystal structure of complex
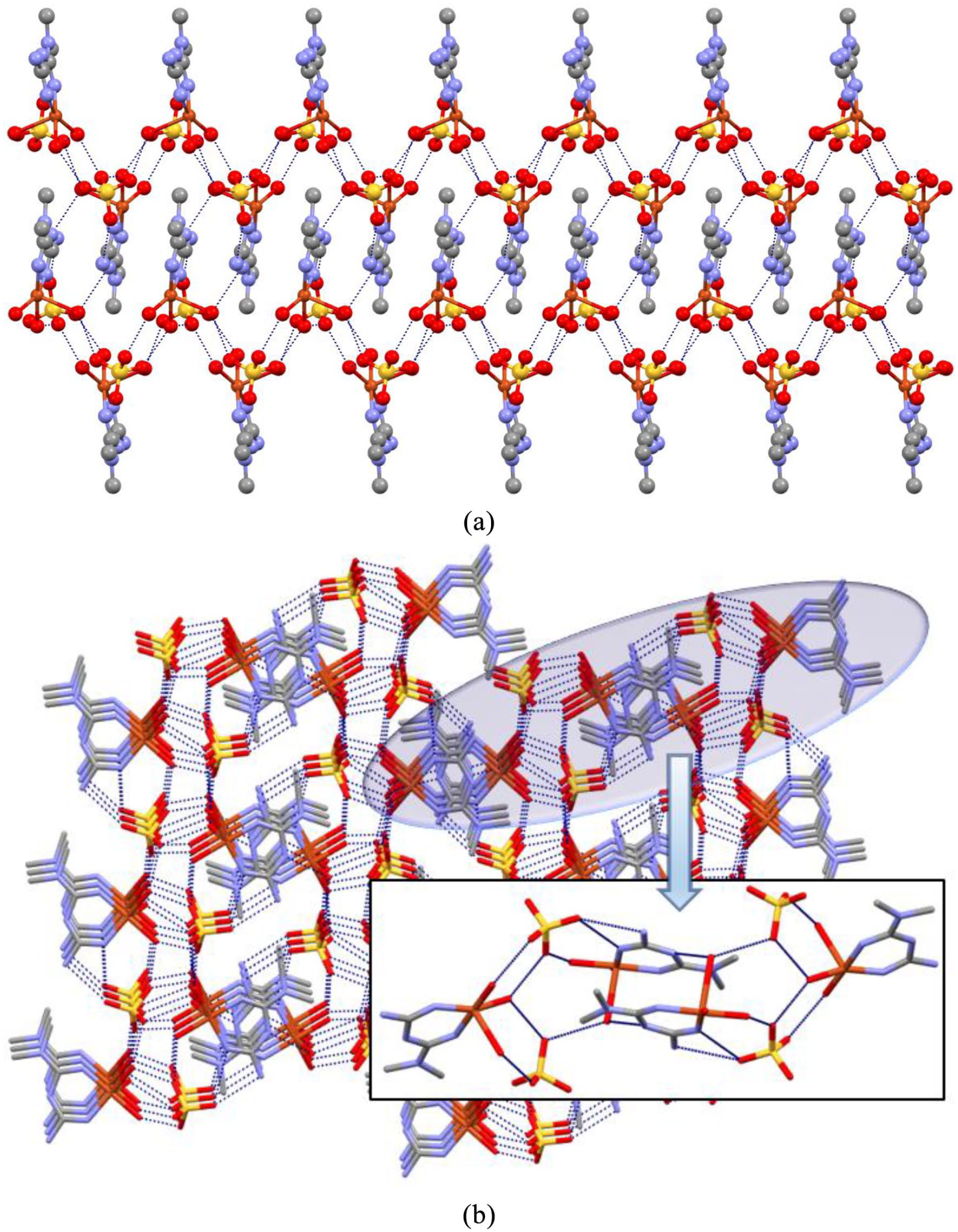
(a) 2D and (b) 3D crystal structures of complex
Crystal data and structure refinement for complex

Selected bond lengths (Å) and angles (°) for complex

Hirshfeld surface analysis
The Hirshfeld surface analysis is a method for the study of the intermolecular interactions as illustrated by the colours red, blue, and white represented on the surfaces. The color intensity can be used for visualizing the strength of the interactions, with the three colors representing the relative strengths of the intermolecular interactions. Red regions represent the hydrogen bond interaction, blue regions represent the longer contacts, and white regions represent the contacts exactly equal to the van der Waals separation.
The Hirshfeld dnorm surfaces, shape index, and curvedness of asymmetric unit complex

Hirshfeld dnorm surfaces, shape index, and curvedness for complex
The two-dimensional (2D) fingerprint plot of complex

Fingerprint plots for complex
In vitro antimicrobial studies
The antibacterial activity of metal–drug complexes has been the focus of continued attention.19–21 Thus, we have investigated complex
MIC values of complex

MH: metformin hydrochloride.
Conclusion
A novel 3D Cu(II) metal–drug complex based on an MH ligand has been synthesized. We have investigated the crystal information through structural investigations. The space group was determined to be monoclinic space of P21/n by single-crystal X-ray diffraction. The crystal packing was studied by the Hirshfeld surface analysis, which revealed the supramolecular structure and hydrogen bonding networks. In addition, we have studied the antibacterial properties of complex
Experimental
Materials and methods
The MH ligand was obtained from Shanghai Macklin Biochemical Co., Ltd and used as received. All other chemicals were of reagent grade quality obtained from commercial sources and were used without further purification. Single-crystal X-ray diffraction data of the product crystals were measured using monochromated MoKα radiation (λ = 0.071073) at 298 K on a Rigaku SCXmini diffractometer. The crystal data were collected and processed using SAINT, and the structures were solved and refined by full-matrix least-squares on F2 using SHELXL. The structure of the title complex was resolved with the MERCURY program. 22
Synthesis of complex 1
The ligand MH (166 mg and 10 mmol) was dissolved in methanol 10 mL and was adjusted to pH 6 with triethylamine, and then slowly added dropwise to a 10-mL aqueous solution containing CuSO4·5H2O (500 mg and 20 mmol) and stirred for 30 min and filtered. Blue block transparent crystals of complex
Single-crystal X-ray diffraction
The single-crystal X-ray diffraction data of complex
Hirshfeld surface analysis
Molecular Hirshfeld surface calculations were mapped using the CrystalExplorer by reading the cif files of the complexes. During the analysis, all bond lengths to hydrogen were automatically modified to typical standard neutron values (C–H = 0.93 Å and N–H = 0.86 Å). The normalized contact distance (dnorm) is defined as de and di. In this study, the Hirshfeld surfaces were generated using a standard (high) surface resolution. The 3D dnorm surfaces are mapped over a fixed color scale of 0.42 (red) to 1.6 Å (blue). The 2D fingerprint plots are displayed using the standard 0.6–2.6 Å view with the de and di distance scales displayed on the graph axes.
In vitro antibacterial activity studies
The antibacterial activity of the complex
The MIC was the lowest concentration of antimicrobial agent showing complete inhibition of growth, which was studied by the liquid microdilution method, and performed by preparing twofold dilutions of complex
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was based on the work supported by the Natural Science Research Foundation of Jiangsu Higher Education Institutions of China (grant no. 19KJD230002) and the Scientific Research Foundation of the Graduate School of Jiangsu Agri-Animal Husbandry Vocational College, China (grant no. NSFHP201902).
