Abstract
A heterogeneous Cu(II)-AOFs catalyst, Cu(II) supported on amidoxime fibers (AOFs), is successfully applied to the cross-dehydrogenative coupling of aromatic terminal alkynes and N,N-dimethylanilines to form propargylamines. The Cu(II)-AOFs catalyst shows high catalytic activity with the yields of the corresponding propargylamines reaching 90% at 70 °C for 4 h without the protection of an inert gas. The Cu(II)-AOFs coordinate with the imine ion intermediate generated during the reaction, making the coupling facile. X-ray photoelectron spectrometer, scanning electron microscope, and energy dispersive X-ray spectroscopy results show that Cu(II) successfully coordinates with N and O atoms on the surface of the fibers with the mutual conversion between monovalent and divalent Cu in the Cu-AOFs being the catalytically active center. The catalyst can be recycled more than four times, and the catalytic activity is not obviously reduced. This process represents a new pathway for the formation of propargylamines using a Cu(II)-AOFs catalyst.
Keywords

Introduction
Propargylamines, are the components of many bioactive intermediates, 1 and are widely used for the preparation of nitrogen-containing compounds, such as pyrazines, oxazoles, and pyrroles. 2 The preparation of propargylamines has attracted interest in the fields of medicinal chemistry and organic synthesis.3,4 Traditional synthetic methods toward propargylamines are reactions of propargyl halides with amines and functional amines with metal acetylides and the one-pot, three component reactions of aldehydes/dihalomethanes, amines, and alkynes. 5 However, these methods generally require a leaving functional group or pre-functionalized aldehydes and amines.
Recently, numerous cross-dehydrogenative coupling (CDC) methods have attracted significant interest via transition-metal activation of the sp 3 C–H bond adjacent to a nitrogen atom to form directly the propargylamines. In 2004, it was first reported that CuBr catalyzed the synthesis of propargylamines from anilines and terminal alkynes in the presence of tert-butyl hydroperoxide (TBHP), and the yields of 12%–82% were obtained at 100 °C in 3 h. 6 When (t-BuO)2 is used as the oxidant, FeCl2 can catalyze the coupling reaction of tertiary amines with terminal alkynes. 7 It has been reported that a Cu(acac)2 catalyst promoted the reaction of tertiary amine oxide with terminal alkynes at 110 °C under an N2 atmosphere and provided 53%–89% yields of the corresponding propargylamines. 8 The reaction of N-phenyltetrahydroisoquinoline with phenylacetylene was realized with a dual catalyst composed of the photooxidation catalyst [Ru(bpy)2(dtbbpy)](PF6)2 and the metal salt (MeCN)4CuPF6, with the desired product being obtained in 84% yield under visible light over 16 h. 9 Other copper-catalyzed methods for the synthesis of propargylamines have also been developed by several research groups.10,11 However, most of the above CDC reactions involve homogeneous catalysts, which are difficult to recover and separate after the reaction.
In order to solve these problems, various heterogeneous catalysts have been developed and applied in CDC experiments. For example, using Y-type zeolite-supported copper nanoparticles (CuNPs/ZY) as the catalyst led to the successful reaction between anilines and terminal alkynes to obtain the desired products in moderate to excellent yields at 70 °C over 20 h. 12 The ZnBr2 and manganese oxide–based octahedral molecular sieves composite catalyst (Zn2+-OMS-2) promoted the CDC reaction of aliphatic amines and terminal alkynes, and when cyclopentyl methyl ether (CPME) was used as the solvent and oxygen as the oxidant, the yields of the corresponding products were 13%–83% at 100 °C over 8 h. 13 A metal-organic framework catalyst, MOF-199, was used to catalyze the alkynylation reaction of N,N-dimethylaniline and phenylacetylene in 79% yield at 120 °C over 3 h. 14 In addition, the nanocatalyst Cu6Se4.5NPs was applied in the reaction of N,N-dimethylbenzylamine and phenylacetylene, and the desired product was obtained in 62% yield at 100 °C in 3 h in the presence of N2. 15 Later, copper(I) ligated with 4,5-bis[(phenylthio/seleno)/methyl] acridine catalysis was reported to catalyze the same reaction of N,N-dimethylbenzylamine and phenylacetylene to afford a 73% isolated yield of the products at 60 °C over 6 h under inert gas protection. 16 However, after reaction with a Zn2+-OMS-2 catalyst, only OMS-2 molecular sieves were recovered, which needed to be reloaded by adding ZnBr2 for the next experiment. The process for the preparation of the nanocatalyst is not only lengthy and complicated but also needs a harsh environment without water or oxygen. In addition, flammable and explosive lithium powder or easily oxidized trioctylphosphine is indispensable experimental raw materials in the preparation of such systems.
In this research, we prepared a metal cation–supported amidoxime fiber (AOFs) catalyst using cheap raw materials via a simple preparative method. The catalyst demonstrated good performance CDC reactions of N,N-dimethylanilines and terminal alkynes. The AOFs (involving [H2N–C=N–OH] side chain group) with abundant and coordinating –NH2, –OH functional groups, were obtained by modifying the –C≡N functional group in the polyacrylonitrile (PAN) fiber with NH2OH. When the AOFs were reacted with copper bromide solution at room temperature, the N and O atoms on the surface of the AOFs, which have lone pairs of electrons, can coordinate with the empty orbitals of Cu(II) to give Cu(II)-AOFs. The Cu(II)-AOFs catalyst shows high catalytic activity and selectivity for the CDC reaction of N,N-dimethylaniline and phenylacetylene to form the propargylamine at 70 °C over 4 h in air. The coupling product is obtained in 90% yield, and the alkyne selectively couples with a methyl group of the N,N-dimethylaniline. In addition, the Cu(II)-AOFs is an effective and reusable heterogeneous catalyst for the CDC reaction.
Results and discussion
Analysis of scanning electron microscope and energy-dispersive X-ray for the Cu-AOFs
The morphology of the Cu(II)-AOFs before the reaction was characterized by scanning electron microscope (SEM). As shown in Figure 1(a), the surface of the Cu(II)-AOFs before the reaction was very smooth, the energy-dispersive X-ray (EDX) analysis (Figure 1(b)) revealed the presence of a large amount of Cu in the Cu-AOFs catalyst.

(a) SEM and (b) EDX patterns of the Cu(II)-AOFs.
Analysis of X-ray photoelectron spectrometer for the Cu-AOFs
In order to clarify the change of the valence state of the copper during the reaction, X-ray photoelectron spectrometer (XPS) measurements of the Cu-AOFs before and after the reaction were carried out. The XPS of Cu(II)-AOFs before reaction showed that the catalyst was composed of five elements: C, N, O, Cu, and Br, as shown in Figure 2(a). Figure 2(b) shows that there is only one valence state of Cu(II) in the Cu(II)-AOFs before the reaction, showing one peak at 933.11 eV. 17 However, the copper was found to exist in two different valence states in the Cu-AOFs after the reaction. As shown in Figure 2(c), the peak at 934.48 eV is due to Cu(II) 2p3/2 and the peak at 932.42 eV is attributed to Cu(I) 2p3/2, 18 which can also be proved by the CuL3M45M45 Auger spectroscopy binding energy located at 571 eV (see the Supporting Information). 19 Subsequently, the reacted Cu-AOFs were placed in air at room temperature for 1 week and then studied again by XPS. The results are shown in Figure 2(d). Unexpectedly, the peak intensity of Cu(II) becomes higher and the peak intensity of Cu(I) becomes lower, which suggests that Cu(I) in the Cu-AOFs is unstable, and most are oxidized to Cu(II) by air. Therefore, the above XPS data indicate there may be a mutual conversion between monovalent and divalent Cu during the Cu-AOFs catalytic reaction. 12 At the same time, this feature shows promise for the reuse of the catalyst. Figure 2(e) and (f) shows the characteristic lines of N and O. As shown in Figure 2(e), the peaks due to N1s in the AOFs appear at 399.08 and 399.75 eV, respectively. 19 However, the two peaks of N1s increased to 399.52 and 400.56 eV in Cu(II)-AOFs. The increase of binding energy is mainly due to the combination of Cu(II) and N atoms in the Cu(II)-AOFs forming a N–Cu bond. The binding energies at 531.42 and 532.61 eV shown in Figure 2(f) are the characteristic of O1s in AOFs. Compared with the binding energies of O1s in Cu(II)-AOFs, these two peaks are changed to 531.97 and 532.99 eV, respectively, which are ascribed to the O–Cu bond between Cu(II) and O atoms. In summary, the above results prove Cu(II) has successfully coordinated with the AOFs during preparation of the catalyst.

XPS survey spectra of (a) Cu(II)-AOFs (unreacted), (b) Cu2p XPS spectra of Cu(II)-AOFs (unreacted), (c) Cu2p XPS spectra of Cu-AOFs (reacted), (d) Cu2p XPS spectra of Cu-AOFs after standing for 1 week, and (e) and (f) peak fitting of the O1s and N1s spectra in AOFs and Cu(II)-AOFs (unreacted).
Optimization of the reaction conditions
In order to obtain high yields, the conditions for the reaction of N,N-dimethylaniline with phenylacetylene were optimized. As shown in Table 1, the desired coupling product is not obtained in the absence of copper catalyst (entry 1).
Optimization of the reaction conditions. a
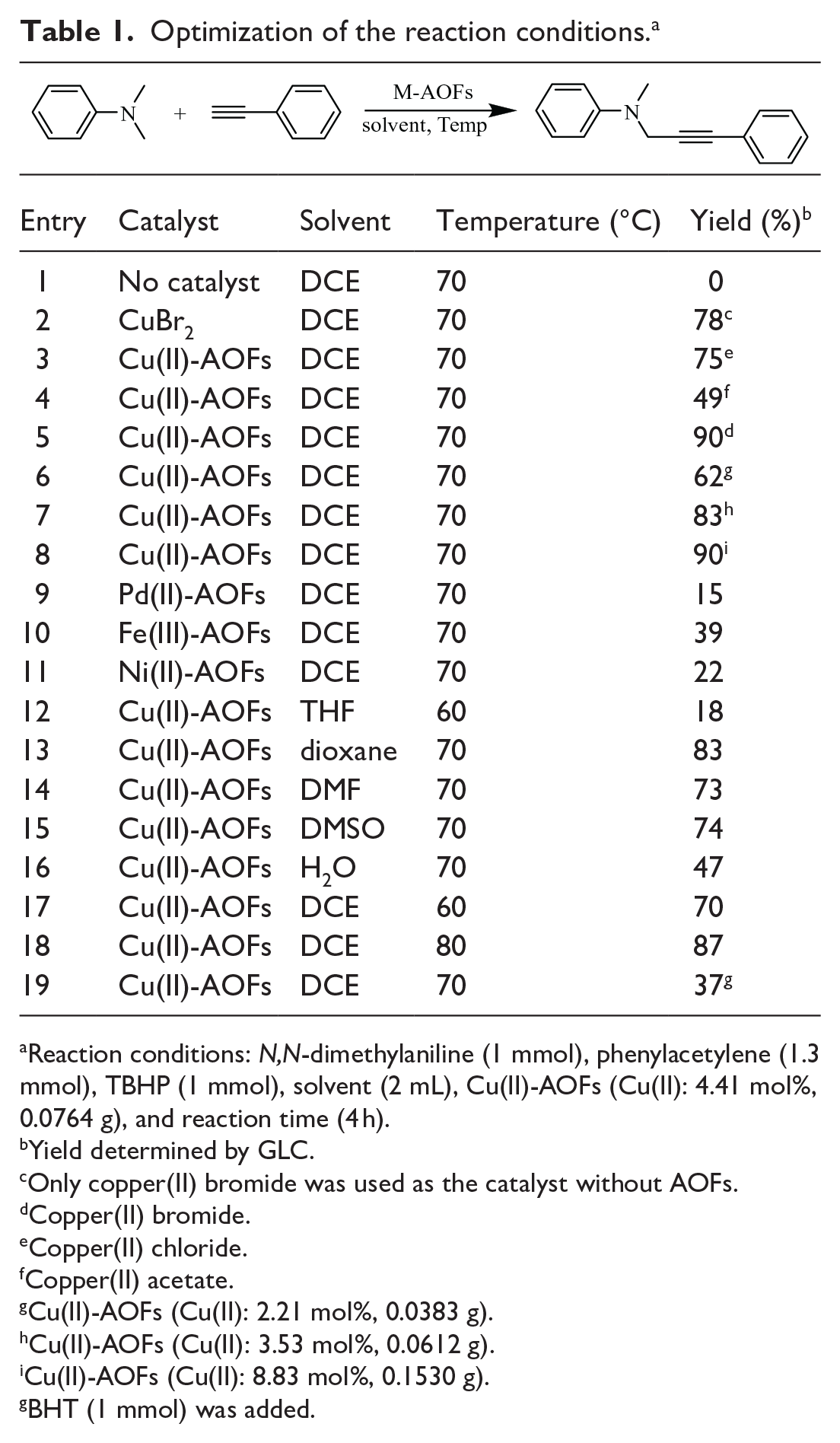
Reaction conditions: N,N-dimethylaniline (1 mmol), phenylacetylene (1.3 mmol), TBHP (1 mmol), solvent (2 mL), Cu(II)-AOFs (Cu(II): 4.41 mol%, 0.0764 g), and reaction time (4 h).
Yield determined by GLC.
Only copper(II) bromide was used as the catalyst without AOFs.
Copper(II) bromide.
Copper(II) chloride.
Copper(II) acetate.
Cu(II)-AOFs (Cu(II): 2.21 mol%, 0.0383 g).
Cu(II)-AOFs (Cu(II): 3.53 mol%, 0.0612 g).
Cu(II)-AOFs (Cu(II): 8.83 mol%, 0.1530 g).
BHT (1 mmol) was added.
The catalytic efficiency of AOFs supported on copper was higher than that of AOFs supported on Pd, Fe, and Ni (entries 5, 9, 10, and 11). Compared with fibers loaded with copper(II) chloride and copper(II) acetate, the fiber loaded with copper(II) bromide gave the product in 90% (entries 3–5). The difference in yields may be related to the Ksp of the Cu(I) substance. Because the Ksp of copper(I) bromide is smaller than that of copper(I) chloride and copper(I) acetate, it is easier to generate Cu(I), thereby promoting reaction efficiency and increasing yield.
A control experiment was conducted with copper(II) bromide as the catalyst without AOFs; however, the yield was only 78% (entry 2). This is because AOFs can be used as ligands to increase the electron density of copper and make it electron rich, thereby improving the stability of intermediate product B and increasing its yield.
Subsequently, the effects of six solvents were also screened with Cu(II)-AOFs as the catalyst (entries 5, and 12–16). THF, dioxane, DMF, DMSO, and H2O gave the product in yields of 18%, 83%, 73%, 74%, and 47%, respectively. DCE prove to be the best solvent (entry 1). The reaction yield increased when the temperature was increased (entries 5, 17, and 18); when the temperature was 70 or 80 °C, the reaction yield was mostly unchanged. In addition, the content of copper supported in Cu(II)-AOFs had a significant influence on the yield. It can be observed that on increasing the content of copper supported in Cu(II)-AOFs, the reaction yield reached the optimum, and when it was higher than 4.41 mol%, the yield did not change (entries 6–8). To sum up, the use of Cu(II)-AOFs as the catalyst and DCE as the solvent gave the highest yield 90% of the coupling product at 70 °C over 4 h. Besides, the yield of this reaction catalyzed by Cu(II)-AOFs was suppressed by the presence of dibutylhydroxytoluene (BHT) as a free radical scavenger, indicating that radical intermediates are involved in the reaction (entry 19).
The effect of substituent groups on the catalytic performance
Under the above reaction conditions, the effect of different substituents on the catalytic performance of Cu(II)-AOFs was also studied. As shown in Table 2, the properties and steric effects of the substituents had no significant influence on the reaction, and good yields of the desired coupling products were obtained. These results indicate that the Cu(II)-AOFs catalyst has good substrate adaptability for the coupling reaction of N,N-dimethylaniline and aromatic alkyne derivatives.
The effect of substituent groups on the catalytic performance. a

Reaction conditions:
Reuse of the Cu(II)-AOFs catalyst
In order to further explore the catalytic performance of the Cu(II)-AOFs in this reaction, the reuse of the catalyst was tested and the results are shown in Figure 3. It can be seen the Cu(II)-AOFs can be reused at least four times without a significant decrease in activity. In order to explore the nature of the catalyst, we conducted a hot filtration experiment. Under the optimized reaction conditions, the Cu(II)-AOFs catalyst was used to catalyze the reaction of phenylacetylene and N,N-dimethylaniline. When the product was present in 30% yield after 0.5 h, the Cu(II)-AOFs were separated by hot filtration, and the filtrate was reacted at 70 °C for 3.5 h. The experimental results showed that the yield of product was not further improved. Combined with the efficiency of reuse, this proved that there is only a small amount of copper dissolved in the reaction solution.

Reuse of the Cu(II)-AOFs catalyst.
Exploration of the experimental mechanism
Based on the above catalytic performance and catalyst characterization analysis, we have proposed a possible mechanism for the Cu(II)-AOFs–catalyzed dehydrogenative coupling of N,N-dimethylaniline and phenylacetylene (Scheme 1). First, the N,N-dimethylanilines react with Cu(II)-AOFs through a redox reaction to form N,N-dimethylanilines radical cation

Possible mechanism of the Cu(II)-AOFs catalyzed CDC reaction of N,N-dimethylanilines and aromatic terminal alkynes.
Conclusion
In summary, Cu(II)-AOFs show excellent catalytic performance for the CDC reaction of N,N-dimethylanilines and aromatic terminal alkynes to form propynylamines, and the desired product can be obtained in 90% yield in air at 70 °C over 4 h. Importantly, by simple filtration and recovery after reaction, the catalyst can be reused at least four times. SEM and XPS characterization proved that the Cu(I)/Cu(II)-AOFs redox behavior of the catalyst is the key to the reaction cycle. This research provides a new pathway for the CDC reaction for the synthesis of propargylamines using a heterogeneous catalyst.
Experimental section
Reagents and characterization
PAN was purchased from Anqing Carbon Co., Ltd., China. Reagents were provided by Sinopharm Chemical Reagent Co., Ltd., China, and were not purified further. The morphology and elemental analysis of the catalyst Cu(II)-AOFs were established using a Hitachi S-4800, Japan field-emission SEM and EDX. The element valence states on the surface of Cu-AOFs were characterized using a Thermo Fisher Scientific ESCALAB 250 XPS, USA. Infrared spectra (IR) were recorded on a Perten DA 7200, Sweden spectrometer with KBr films. High-resolution mass spectra (HRMS) were obtained on an Agilent, USA mass spectrometer. The Cu content in the Cu(II)-AOFs catalyst was measured using an Inductively Coupled Plasma Optical Emission Spectrometer (ICP-OES), Varian 710-ES, USA. A Bruker ARX-400, Germany nuclear magnetic resonance spectrometer was used to obtain the 1H NMR, 13C NMR, and 19F NMR for fluorine-containing products spectra of the reaction products.
Preparation of Cu(II)-AOFs
The Cu(II)-AOFs catalyst is prepared as follows. 20 PAN fibers (5 g) were added to a pre-prepared hydroxylamine aqueous solution (1 mol L−1, 250 mL), and the reaction was stirred in a water bath at 65 °C. After 1.5 h, the fibers were filtered, washed with distilled water to neutrality, and dried naturally to afford the AOFs (5 g). Finally, the AOFs were added copper(II) bromide solution (0.1 mol L−1, 10 mL) at 45 °C for 2 h to give a fiber-supported Cu(II) catalyst (Cu(II)-AOFs). The Cu(II)-AOFs were filtered, washed with distilled water and ethyl acetate, and dried at 45 °C to obtain the green Cu(II)-AOFs. The Cu content in Cu(II)-AOFs was 3.67 wt% according to inductively coupled plasma optical emission spectrometer (ICP-OES) analysis. Scheme 2 shows the synthesis of the Cu(II)-AOFs catalyst.

The synthetic route of the Cu(II)-AOFs.
General procedure for the CDC reaction
At room temperature, Cu(II)-AOFs (Cu(II): 4.41 mol%, 0.0764 g) was added to a solution of terminal alkynes
N-(3-(4-ethylphenyl)prop-2-ynyl)-N-methylaniline (3f )
Yellow oil; IR (film): 3057, 3037, 3026, 2951, 2919, 2810, 1600, 1578, 1504, 1457, 1362, 1335, 1238, 1201, 1111, 1032, 993, 921, 810, 750, 689, 600, 518 cm−1; 1H NMR (400 MHz, CDCl3) δ 7.31-7.21 (m, 4H), 7.08 (d, J = 8.3 Hz, 2H), 6.90 (dd, J = 8.8, 0.9 Hz, 2H), 6.80 (t, J = 7.3 Hz, 1H), 4.24 (s, 2H), 3.02 (s, 3H), 2.60 (q, J = 7.6 Hz, 2H), 1.19 (t, J = 7.6 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ 149.4, 144.5, 131.7 (2C), 129.1 (2C), 127.7 (2C), 120.2, 118.1, 114.4 (2C), 84.3, 84.2, 43.3, 38.7, 28.8, 15.4. ESI-HRMS (m/z): calcd for C17H20N [M + H]+: 250.1590; found: 250.1591.
N-(3-(2-chlorophenyl)prop-2-ynyl)-N-methylaniline (3n )
Brown oil; IR (film): 3065, 3035, 2958, 2923, 1601, 1560, 1507, 1473, 1364, 1339, 1239, 1112, 1062, 1033, 995, 922, 750, 690, 670 cm−1; 1H NMR (400 MHz, CDCl3) δ 7.36 (ddd, J = 15.5, 7.6, 1.4 Hz, 2H), 7.31-7.23 (m, 2H), 7.17 (dtd, J = 20.0, 7.5, 1.5 Hz, 2H), 6.93 (d, J = 8.0 Hz, 2H), 6.81 (t, J = 7.3 Hz, 1H), 4.31 (s, 2H), 3.05 (s, 3H); 13C NMR (100 MHz, CDCl3) δ 149.3, 136.0, 133.5, 129.2, 129.1, 129.1 (2C), 126.3, 123.0, 118.4, 114.7 (2C), 90.6, 81.0, 43.5, 38.8. ESI-HRMS (m/z): calcd for C16H15NCl [M + H]+: 256.0888; found: 256.0889.
Supplemental Material
sj-pdf-1-chl-10.1177_1747519821997978 – Supplemental material for Amidoxime fibers-copper–catalyzed cross-dehydrogenative coupling of N,N-dimethylanilines and aromatic terminal alkynes
Supplemental material, sj-pdf-1-chl-10.1177_1747519821997978 for Amidoxime fibers-copper–catalyzed cross-dehydrogenative coupling of N,N-dimethylanilines and aromatic terminal alkynes by Wei Gao, Tingxian Tao, Siyu Lu, Siyi Xu and Zhichuan Wu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Nature Science Foundation of China (grant no. 21571003).
Supporting information
The characterization of other coupling products and the CuL3M45M45 Auger spectroscopy of Cu-AOFs are shown in Supporting Information.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


