Abstract
Improving production efficiency and developing green catalytic systems are key issues for applying Heck coupling reactions in the chemical industry. In this study, an amidoxime-fiber-supported Pd catalyst is prepared from polyacrylonitrile fiber, hydroxylamine hydrochloride, and PdCl2. The morphology and structure of amidoxime-fiber-supported Pd are characterized by scanning electron microscopy, energy-dispersive spectroscopy, X-ray photoelectron spectrometer, X-ray diffraction, and inductively coupled plasma-optical emission spectrometer. A packed-bed flow reactor is designed and employed for catalysis of the Heck reaction of iodobenzene and styrene based on the amidoxime-fiber-supported Pd catalyst. The effects of reaction temperature, flow rate, cycle time, and reactant concentration on the conversion and yield are investigated. The results show that the optimal conditions for the continuous-flow single reactor are 70 °C, iodobenzene concentration of 30 mmol L−1 in a solution of N,N-dimethylformamide (100 mL) under continuous flow at a flow rate of 2 mL min−1. The catalyst showed good activity and stability after a continuous operation time of 50 h, with catalytic activity 2.21 times higher than that obtained with traditional batch reaction. Furthermore, we have established a new flow reaction system based on the numbering-up of reactors with a parallel connection. The catalytic efficiency of four parallel reactors is four times higher than that of a single reactor. Kinetic studies reveal that the reaction has low activation energy (8.8771 kJ mol−1), indicating that amidoxime-fiber-supported Pd has good catalytic activity. This research provides an alternative pathway for highly efficient Heck coupling in continuous flow.
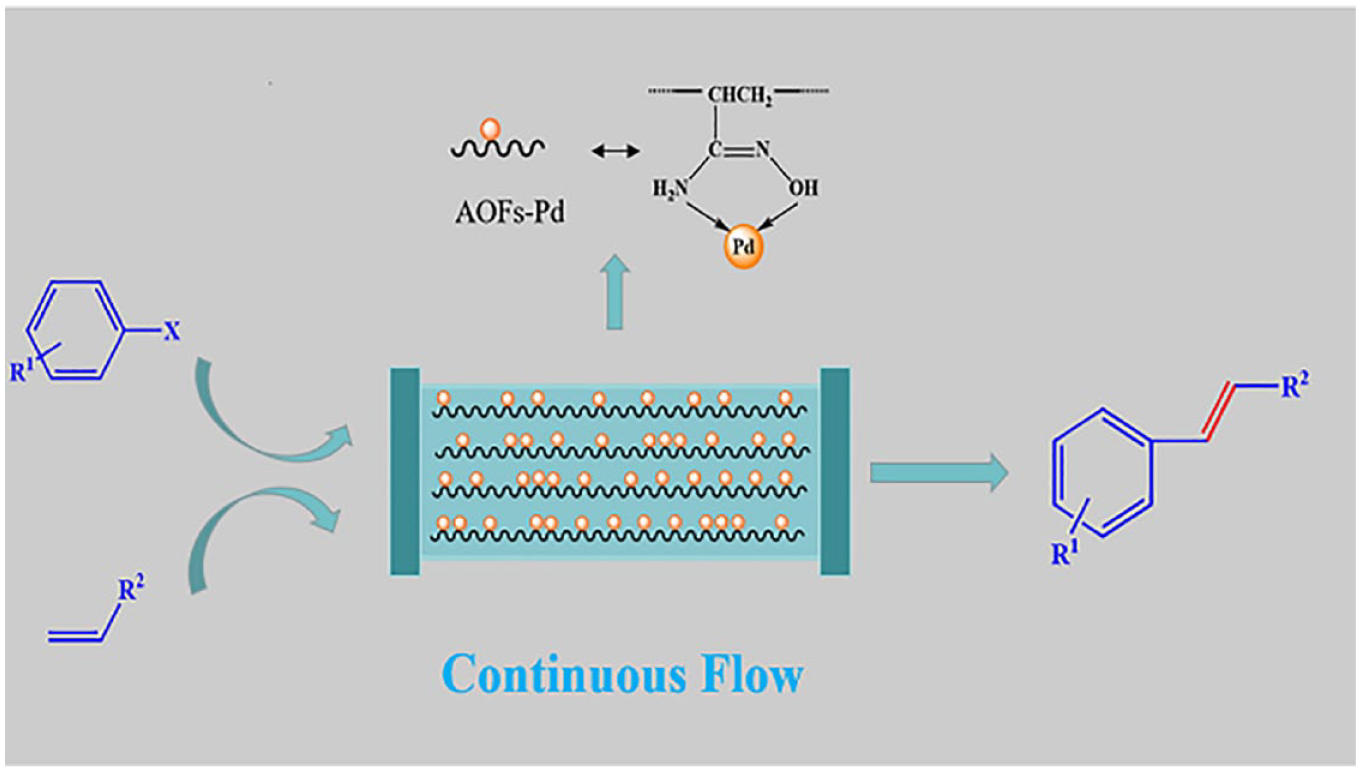
Introduction
The Heck reaction of halogenated aromatics with olefins is among the most direct effective methods for forming C–C bonds and has been extensively applied for the synthesis of drugs and intermediates, natural products, and functional materials.1–4 Since the discovery of the Heck reaction, reaction selectivity, catalyst types, and catalytic mechanisms have been intensively studied.5–7 Among the investigated catalysts, traditional homogeneous palladium catalysts show excellent catalytic activity but also have some limitations such as air-sensitivity, poor recyclability and reusability, and cumbersome post-treatment. Fortunately, supported catalysts can solve the above problems. For example, Yu et al. used polyaniline loaded with a low content of nanoparticles (Pd@PANI) to catalyze the coupling reaction of iodobenzene with styrene. The catalyst had uniform dispersion of palladium nanoparticles and retained good activity after eight reuses, indicating that Pd@PANI is an efficient and stable catalyst. 8 In addition, Houdayer et al. 9 developed polyaniline-loaded nickel nanoparticles (Ni(0)/PANI) with an average diameter of 6.0 ± 2.3 nm and a uniform distribution of Ni particles, which has good catalytic activity in Heck coupling reactions, and more importantly, this catalyst has good stability. However, research on supported catalysts is mainly based on batch reactions at present, which is not beneficial to industrial production due to the low automation degree and production efficiency. Continuous-flow technology allows precise control of process parameters such as temperature, residence time, and concentration. 10 In this technology, raw materials are continuously pumped into the reactor, which has a high specific surface area, and reaction solutions are rapidly diffused and are in full contact with the catalyst. It not only uses raw materials effectively and reduces the generation of side reactions but also shortens reaction times and reduces energy consumption, which is advantageous for industrial applications.11,12
Recently, supported palladium catalysts have been used in continuous-flow chemistry. For example, Bourne et al. developed a method for the rapid and safe preparation of functionalized styrenes using palladium acetate catalyzed coupling of aryl iodides and ethylene, with the amount of ethylene gas precisely controlled by on-line infrared analysis for quantitative monitoring. The continuous-flow technique was combined with the reactive gas as a means of merging the gas build-up into the molecule, and the resulting styrene reacted with the iodoaryl substrate at 120 °C for 20 min to give about 70% of the stilbene product. 13 Viviano et al. 14 have investigated the conditions for the palladium acetate catalyzed reaction of 4-iodophenyl ether with methyl vinyl ketone under intermittent microwave treatment and continuous flow, and the product yield was 67% in the flow state with a residence time of 10 min at 180 °C, while the flow technique optimized the reaction conditions to give high product yields and selectivity in very short reaction times. Laudadio et al. 15 investigated the Heck coupling reaction catalyzed by several Pd catalysts in a continuous-flow-filled bed, and the experimental results showed that the Pd(PPh3)4-PS catalyst gave 93% regioselectivity, an 81% separation yield, and less than 1% leaching of Pd after one reaction, and could be washed and dried for reuse. However, the above catalysts have their own drawbacks, such as complex preparation and separation processes. Furthermore, the catalyst ligands are toxic, cannot be reused, and easily generate palladium black. In addition, the type of catalyst carrier and immobilization methods with fluid bed reactors also affect the flow properties and energy transfer properties of the material fluid in the reaction system, which affects the reaction efficiency and stability. Therefore, it is particularly important to develop green and economic heterogeneous catalysts, which are suitable for continuous-flow reactors.
In this work, cheap and readily available polyacrylonitrile (PAN) fiber was modified with hydroxylamine hydrochloride, and then the −C≡N functional group on the surface of PAN was transformed into an amidoxime group with strong coordination ability. The amidoxime-fiber-supported Pd (AOFs-Pd) catalyst was successfully prepared by the chemical coordination of Pd2+ and amidoxime fiber (AOF). The coordination loading of AOFs with active metals improves the catalytic activity of the metals on one hand, and the strong coordination bonding enhances the stability of the catalyst on the other hand. Because of its unique physical form and good fluid flow properties, fiber is an excellent carrier for continuous-flow packed-bed reactors. The process for the preparation of the AOFs-Pd catalyst is simple, and it can be produced on a large scale. The reaction conditions are mild, and the integral washing and drying can be realized with a packed-bed reactor, allowing the catalyst to be reused and reducing manual operations such as catalyst refilling. The catalytic performance of AOFs-Pd for Heck reactions was investigated in continuous flow, and the effects of temperature, flow rate, concentration, and substrate suitability on the coupling yields were explored.
Results and discussion
Catalyst characterization
High-resolution field emission scanning electron microscopy (SEM) was used to characterize the catalysts before and after the reaction. The results showed that the surface of AOFs-Pd before the reaction is very smooth (Figure 1(a)) and after the reaction maintains the original morphology of the fiber and a lot of particles appear on the surface (Figure 1(b)). The AOFs-Pd particles after the reaction were metallic palladium according to energy-dispersive spectroscopy (EDS) energy spectral analysis (Figure 1(c)).

(a and b) SEM images of AOFs-Pd, (c) EDS image of AOFs-Pd, (d) N1s XPS spectra of AOFs-Pd (unreacted), (e) O1s XPS spectra of AOFs-Pd (unreacted), (f) Pd3d XPS spectra of AOFs-Pd (unreacted), (g) XPS survey spectra of AOFs-Pd (unreacted), (h) Pd3d XPS spectra of AOFs-Pd (reacted), and (i) XRD patterns of AOFs-Pd (unreacted) and AOFs-Pd (reacted).
The AOFs-Pd catalyst was characterized by Kα-type X-ray photoelectron spectroscopy, and the results showed that it consisted of five elements: C, N, O, Pd, and Cl. As shown in Figure 1(d), the two binding energies of N1s shifted from 399.30 and 400.50 eV (AOF) to 399.72 and 400.99 eV (AOFs-Pd), 16 respectively. This was ascribable to coordination between −C=N and Pd2+ ions, which decreased the electron cloud density of N and increased the binding energy of N1s. Figure 1(e) shows that the binding energy of O1s in the AOFs-Pd (533.32 eV) was 0.92 eV higher than that of the AOF (532.4 eV), and this can be attributed to O providing a lone pair electrons to Pd when participating in coordination, thus decreasing the density of the electron cloud around it. As shown in Figure 1(f), the binding energy of Pd 3d5/2 (337.76 eV) in AOFs-Pd was 0.24 eV less than that of PdCl2 (338.00 eV), indicating that Pd2+ accepted lone pairs of electrons provided by the O and N of −OH and −NH2, leading to an increase in the electron cloud density and a decrease in the binding energy. The above results demonstrated that Pd successfully coordinated with the −NH2 and −OH groups in the AOF to form a stable chelate. Figure 1(h) shows that two valence states of Pd (Pd0 (3d5/2, 355.96 eV; 3d3/2, 341.20 eV) and PdII (3d5/2, 337.72 eV; 3d3/2, 342.20 eV)) were present in AOFs-Pd after the reaction. Pd0 and PdII were present on the fiber surface at the same time, indicating that Pd0 was generated after the reaction. According to the X-ray diffraction (XRD) patterns in Figure 1(i), the catalyst before and after reaction contained characteristic diffraction peaks of fibers, and the catalyst after reaction produced new diffraction peaks. The diffraction peaks almost completely matched the Powder Diffraction File (PDF) card of standard Pd (JCPDS PDF#46-1043), in which the (111) and (200) crystal plane peaks appear at 40.1° and 46.7°, respectively. The XRD spectrum showed that Pd2+ on AOFs-Pd was reduced to Pd0, which also confirmed that the real catalytically active center was Pd(0).
Optimization of the reaction conditions
AOFs-Pd was used as the catalyst for the Heck reaction of iodobenzene with styrene in a continuous-flow reactor. In order to obtain the optimum reaction conditions, the effects of the reaction temperature, flow rate, and substrate concentration on promoting the reaction were screened and the results are summarized in Table 1.
Optimization of the continuous-flow Heck coupling reaction process.

Entries 1–5 in Table 1 show that the reaction yield increases on raising the temperature from 40 °C to 80 °C. The yield reached 95% at 70 °C, and further increasing the temperature did not lead to significant change in the yield. Therefore, the optimum reaction temperature for this continuous-flow reaction was 70 °C from an energy consumption point of view.
At 70 °C and an iodobenzene concentration of 20 mmol L−1, we explored the effect of different flow rates (2, 3, 4, 5, and 6 mL min−1) on the reaction yields (see the Supporting Information, section S1 for more details). According to Table 1, entries 4 and 6–9, the residence time of the reaction solution in the packed-bed reactor decreased with an increase in flow rate, resulting in insufficient contact between the reactants and the catalyst and a decrease in the reaction yield. At low flow rates, the yield decreased as the cycle reaction times increased, most likely because the reaction time was too long, resulting in by-product generation. The yield decreased with the number of cycles at high flow rates. However, when we analyzed the hourly molar amount of catalytic products, we discovered that the highest yield (2.28 mmol h−1) was obtained at a flow rate of 2 mL min−1 for one continuous-flow reaction, and the process conditions were the simplest, so the optimum flow rate to complete the reaction was selected as 2 mL min−1.
From entries 4 and 10–13 in Table 1, it was found that the yields were above 95% for a substrate concentration below 30 mmol L−1, and the hourly catalytic production increased with increasing substrate concentrations. When the substrate concentration was higher than 30 mmol L−1, the hourly catalytic production also increased, but the yields gradually decreased. Considering both the reaction yield and the hourly catalytic production, 30 mmol L−1 was found to be the optimum substrate concentration.
Continuous operation stability of the catalyst
Under the above optimal continuous-flow synthesis process conditions, the continuous operation stability of the AOFs-Pd catalyst was investigated. As shown in entries 14–18 of Table 1, the reaction yield was above 90% for the first 30 h of continuous operation of the catalyst, and it began to decline slightly as time progressed, dropping to 82% after 50 h of continuous operation. It was found that the total palladium catalyst content was 0.00468 g, the total catalytic production was 28.85 g, the hourly production was 3.20 mmol h−1, and the Pd catalytic activity was 683.8 mmol g−1 h−1 under continuous-flow conditions. In addition, this catalyst was reused eight times and reacted for 48 h under intermittent conditions. The total palladium content of the catalyst was 0.00488 g, the production was 13.04 g, the hourly production was 1.51 mmol h−1, and the Pd catalytic activity was 309.4 mmol g−1 h−1. 16 By comparison, the Pd catalytic activity of the continuous-flow reaction was 2.21 times higher than that of the batch reaction. The solution was tested by inductively coupled plasma (ICP) spectroscopy after 50 h, and the palladium leakage was measured to be 0.055 mg L−1, with a palladium loss rate of approximately 7%. This demonstrated that the catalyst has high catalytic activity and good stability, implying that the AOFs-Pd-catalyzed Heck reaction under continuous flow is a more green and cost-effective process.
Continuous-flow process applicability
Using the optimum synthesis process under continuous-flow conditions, substrate scope studies were undertaken to evaluate the applicability of this catalytic system. Aryl iodide and alkene substrates with different functional groups were examined next, and the results are summarized in Table 2.
Effects of different substituted groups on the Heck coupling reactions.

Yields determined by column chromatography purification.
The results show that some of the aryl halides could be converted successfully into the desired products with high yields under continuous flow with AOFs-Pd as the catalyst. The presence of electron-withdrawing or electron-donating substituents on the aryl halides had little effect on the catalytic performance, and the yields were all around 90% and the selectivities were above 96%. For iodinated aromatics such as iodobenzene and styrene, the reaction temperature was reduced by 20 °C compared with the intermittent type, and the hourly production was increased 2.16 times. With brominated aromatics, for example, the reaction of bromobenzene with styrene, a batch reaction temperature of 160 °C and time of 24 h, gave a 83% yield. 16 However, the yield under continuous-flow conditions increased to 91%, the temperature was reduced by 20 °C, the reaction time was reduced to 2.5 h, and the hourly production increased to 3.15 times. Also, for chlorinated aromatics, in the reaction of chlorobenzene with styrene, for example, the yield increased by 18% over the intermittent type and the hourly production was improved to 3.41 times. In summary, AOFs-Pd exhibited higher catalytic activity and selectivity in a continuous-flow reactor than in batch reactions.
Factors that affect the continuous-flow numbering-up reactors
To further improve the production capacity, we investigated the effect of the number of reactors on the catalytic yield. From Figure 2(a), it can be seen that the hourly catalytic production in continuous-flow numbering-up reactors was related to that of the single reactor. The system has a uniform flow distribution. A large total flow in parallel reactors and a low pressure drop inside the columns of the flow reactors allowed for more effective contact between the substrate and the catalyst, saving time and achieving the desired catalytic efficiency. The continuous-flow quad parallel reactors had a hourly catalytic production (13.80 mmol h−1), which was 8.51 times higher than the batch reaction (1.62 mmol h−1). So, the continuous-flow numbering-up reactors greatly improved the catalytic yield and effectively reduces energy losses.

(a) Exploration of the numbering-up reactors, (b)a determination of the reaction series, and (c)b activation energies of AOFs-Pd.
High catalytic activity of AOFs-Pd
To study the catalytic activity of AOFs-Pd, the rate constants of the catalysts under intermittent conditions were determined through catalytic kinetics. To exclude the interference of product concentration on the reaction rate, the initial concentration method was used to determine the initial reaction rate at different concentrations, and then the number of reaction steps was determined by the differential method to calculate the change in reactant concentration with time at different temperatures (see the Supporting Information, section S2 for more details). The results showed that the number of reaction stages n = 1.0885≈1, the reaction activation energy (Ea) was about 8.8771 kJ mol−1, and the rate equation under optimum conditions is C = C0e−0.0677t (Figure 2(b) and (c)). Thus, the AOFs-Pd catalyst has high catalytic activity.
Conclusion
In this experiment, AOFs with coordination ability were prepared by modification of PAN fibers with hydroxylamine, and stable polymer catalysts were obtained by coordination of Pd with the OH and the NH2 groups of AOFs. This polymer catalyst is suitable for filling the reactor as the carrier and can be reused after the reaction by simply washing with ethanol and water. The AOFs-Pd-catalyzed continuous-flow Heck reaction achieved a 95% yield at a temperature of 70 °C, a flow rate of 2 mL min−1, and a substrate concentration of 30 mmol L−1. Compared with the batch reaction, the catalytic efficiency of the flow technique was improved by 2.21 times. The numbering-up system not only maintained the characteristics of a single reactor but also increased output. The production capacity of a quad parallel reactor reached four times that of a single reactor at the same time, while the energy consumption of the equipment was greatly reduced. In conclusion, the combination of an AOF palladium catalyst and continuous-flow chemistry results in excellent catalytic activity and stability, and the production efficiency is greatly improved, which is in line with the requirements of green chemical production and has wide application prospects.
Experimental
Reagents
PAN fiber was purchased from Sinopec Anqing Branch; hydroxylamine hydrochloride, anhydrous sodium carbonate, hydrochloric acid, and palladium chloride were purchased from China Shanghai Sinopharm Chemical Reagent Co., Ltd., and the purity was of analytical grade (AR). Iodobenzene, styrene, 4-methoxyiodobenzene, 4-nitroiodobenzene, 4-methyliodobenzene, bromobenzene, 4-nitrobromobenzene, 4-methylbromobenzene, 4-methoxybromobenzene, acrylic acid, chlorobenzene, acrylamide, triethylamine, N,N-dimethylformamide, ethyl acetate, petroleum ether, and ethanol were purchased from Shanghai Aladdin Co., Ltd. All reagents were used without additional purification.
Instruments
A D8-Focus X-ray diffractometer (XRD), Bruker, Germany; an S-4800 field emission scanning electron microscope (SEM, EDS), Hitachi, Japan; a Bruker AV 500M nuclear magnetic resonance (NMR) spectrometer, Bruker, Germany; a 5110 ICP-OES Ltd., USA; a high-performance liquid chromatography LC-16 (HPLC), Shimadzu, Japan; a Kα-type XPS, Thermo Fisher Scientific, USA; and a BT100LC/YZ15MaT peristaltic pump (Baoding Chuangrui Precision Pump Co.) were used.
Preparation of the AOFs-Pd catalyst
AOFs were prepared according to the literature 17 : PAN (10 g) was immersed in hydroxylamine solution (0.8 mol L−1, 500 mL) and then heated at 65 °C with stirring for 1.5 h. The obtained fibers were filtered, washed with distilled water, and dried naturally to afford the AOFs. Finally, the AOFs (10 g) were added to PdCl2 solution (0.02 mol L−1, 44.4 mL) and stirred. After 30 min, the treated fibers were washed and dried to give yellowish AOFs-Pd. The palladium content in AOFs-Pd was 0.088 mmol g−1 according to ICP-OES analysis.
Characterization of the catalyst
The chemical composition and elemental valence state of the catalyst were characterized by X-ray photoelectron spectroscopy (XPS, excitation source: Al Kα X-rays, operating voltage: 15 kV, filament current: 10 mA). The wide-angle XRD measurement was recorded on a D8 Focus X-ray diffractometer (40 kV, 40 mA, 2° min−1, 10–80°). The morphology images of the catalysts were obtained by SEM and by EDS on a Hitachi S-4800 field emission high-resolution SEM. The content of palladium metal in the catalyst was determined by ICP-OES. The 1H NMR spectra of the reaction products were recorded on a Bruker Avance III spectrometer (500 MHz).
Heck coupling reaction of halogenated aromatic hydrocarbons with vinyl compounds
Iodobenzene (2 mmol), styrene (3 mmol), and triethylamine (6 mmol) were added to N,N-dimethylformamide (100 mL). The mixture was then stirred at reflux (70 °C) in an oil bath. The AOFs-Pd catalyst (0.5 g) was packed in the catalytic column, and a constant temperature was maintained using a circulating heating device. After the reaction temperature had been reached in the preheating and reaction zones, the reaction mixture was flowed into the catalytic column using a peristaltic pump at a set flow rate. Finally, the reacted solution was collected from the outlet of the catalytic column. The samples were cooled to room temperature and analyzed by liquid chromatography (LC) with the following conditions: C18-WR chromatographic column (5 μm, 4.6 × 250 mm), H2O:CH3OH (1:9), a detection wavelength of 254 nm, a column temperature of 25 °C, a flow rate of 1.0 mL min−1, and an injection volume of 20 μL. The sample was extracted with ethyl acetate and concentrated to give the crude product. The pure product was obtained by column chromatography and was characterized by NMR (Scheme 1).
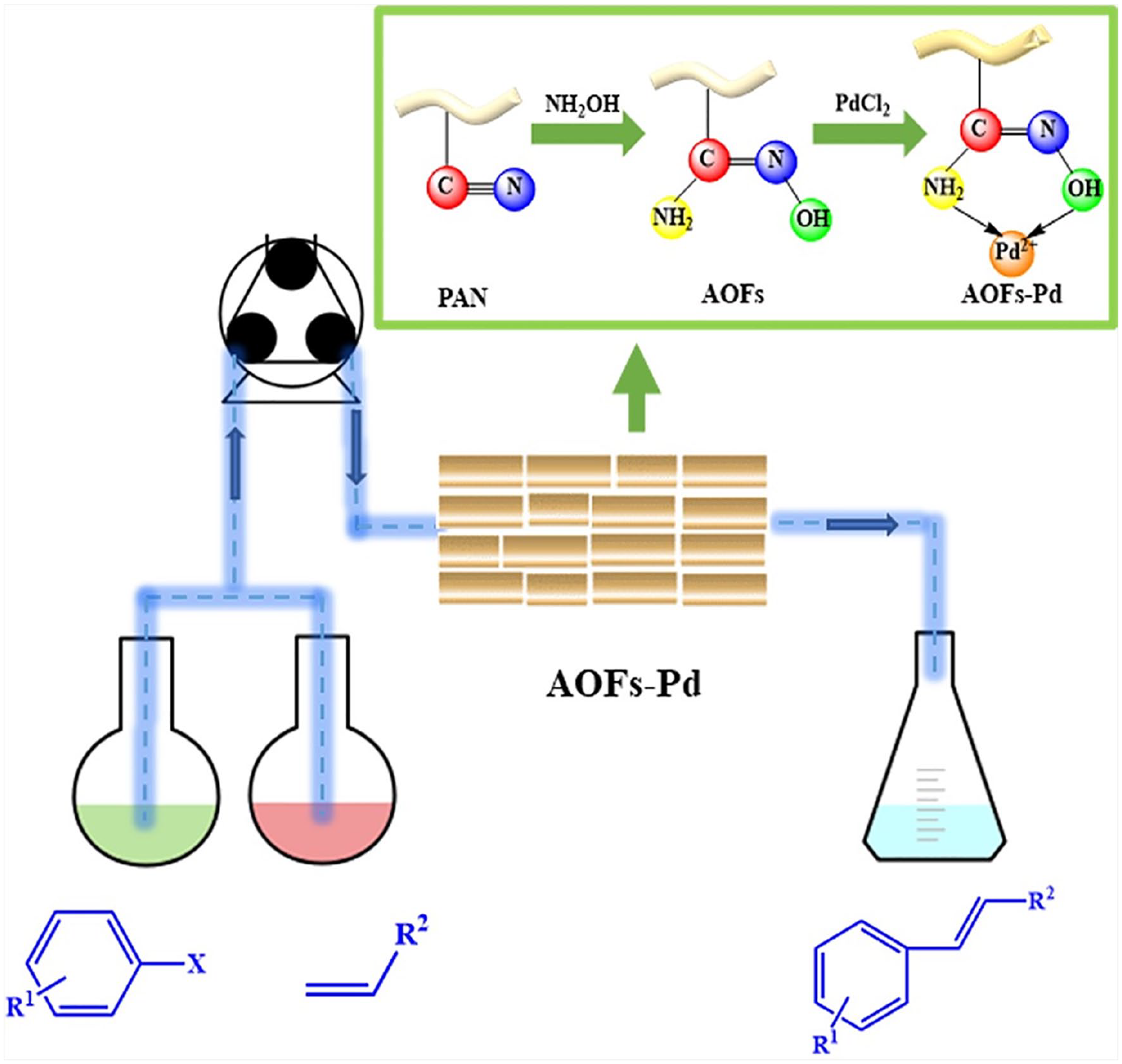
Schematic diagram of the continuous-flow Heck coupling reaction process.
Stilbene
Crystalline white solid; Yield 95%. 1H NMR (500 MHz, CDCl3) δ = 7.51 (d, J = 8 Hz, 4H), 7.35 (t, J = 7.5 Hz, 4H), 7.27–7.22 (m, 2H), 7.11 (s, 2H); 13C NMR (125 MHz, CDCl3) δ = 137.39, 128.77, 127.71, 126.60.
Cinnamic acid
White solid; Yield 95%. 1H NMR (500 MHz, d6-DMSO) δ = 12.43 (s, 1H), 7.69–7.68 (m, 2H), 7.61 (d, J = 16.0 Hz, 1H), 7.42–7.41 (m, 3H), 6.55 (d, J = 16.0 Hz, 1H); 13C NMR (125 MHz, d6-DMSO) δ = 168.07, 144.40, 134.69, 130.68, 129.36, 128.67, 119.69.
Supplemental Material
sj-doc-1-chl-10.1177_17475198231157010 – Supplemental material for Continuous-flow Heck coupling reactions catalyzed by Pd complexes of amidoxime fibers
Supplemental material, sj-doc-1-chl-10.1177_17475198231157010 for Continuous-flow Heck coupling reactions catalyzed by Pd complexes of amidoxime fibers by Jiaxin Xie, Siyu Lu, Yilin Zhang, Xiaohan Lu, Zhen Ding, Tingxian Tao, Maodong Xu and Zhichuan Wu in Journal of Chemical Research
Footnotes
Author contributions
Z.W. and T.T. conceptualized and coordinated the research project. J.X. and S.L. built the packed-bed reactor. J.X., Y.Z., X.L., Z.D., and M.X. conducted the research. J.X., Y.Z., X.L., Z.D., and M.X. prepared the manuscript. All authors reviewed the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is financially supported by the National Natural Science Foundation of China (grant nos. 21571003 and 22072001).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

