Abstract
Ticagrelor is an oral antiplatelet agent that has been approved for preventing de novo and recurrent acute coronary syndrome. To date, only a few studies have attempted to clarify population differences in the pharmacokinetics and pharmacodynamics of ticagrelor between Caucasians and Asians. Our aim was to develop a simple quantification method for ticagrelor and its pharmacologically active metabolite M8 (AR-C124910XX) in human plasma and urine. First, we concisely synthesized M8 from ticagrelor via a five-step sequence: the hydroxyethyl group of ticagrelor was removed by bromination and subsequent zinc-mediated chemoselective reduction. Then, we developed a simple liquid chromatography–mass spectrometry method using ticlopidine as an internal standard. Ticagrelor, M8, and internal standard were separated with a reverse-phase C18 column, and ticagrelor and M8 were detected at m/z [+] of 523.25 and 479.25, respectively, with good sensitivity and precision in the concentration ranges of 10 to 100 µM and 5 to 50 µM, respectively. This novel liquid chromatography–mass spectrometry system may be attractive to investigators in private laboratories because it is less costly than liquid chromatography/mass spectrometry/mass spectrometry systems. Our method is expected to accelerate further clinical studies on the disposition and pharmacodynamics of ticagrelor.

Introduction
Ticagrelor is a recently approved oral antiplatelet agent for preventing restenosis after coronary artery revascularization in patients with coronary stenosis (e.g. acute coronary syndrome (ACS)). Ticagrelor suppresses platelet aggregation by directly inhibiting P2Y12 receptors in the same way as clopidogrel sulfate and ticlopidine hydrochloride. The efficacy of ticagrelor for preventing restenosis in ACS patients treated with coronary intervention was compared with that of clopidogrel in a large, randomized, double-blind clinical trial (PLATO study, N = 18,624) in mostly Caucasian patients (91.7%). Results revealed that the occurrence of cardiovascular events by 12 months was significantly less in the ticagrelor group than in the clopidogrel group (hazard ratio: 0.84; 95% confidence interval: 0.77 to 0.92). The risk of major bleeding events was similar between the groups. 1 Subsequently, a randomized study was performed to compare ticagrelor and clopidogrel in Asian patients (PHILO study, N = 801). In contrast to the PLATO study, no statistically significant differences were observed in efficacy and risk of major bleeding events between ticagrelor and clopidogrel in the PHILO study. 2 While the discrepant results may be attributable to the smaller sample size in the PHILO study than in the PLATO study, differences in the pharmacokinetics (PK) and/or pharmacodynamics (PD) of ticagrelor between Caucasians and Asians may also play a role. In this context, further studies are needed on the PK and PD of ticagrelor, particularly in Asians.
The lack of a simple assay method for ticagrelor and its active metabolite AR-C124910XX (M8) 3 appears to be an obstacle to clinical studies. While several liquid chromatography/mass spectrometry/mass spectrometry (LC/MS/MS) methods have been developed mainly for large clinical trials supported by pharmaceutical companies,4–8 to our knowledge, no assay method is available for LC/MS systems. In general laboratories at hospitals or academic institutions, an LC/MS system would be a versatile alternative to LC/MS/MS systems. Another obstacle is that no pure M8 standard is commercially available. Since M8 may be at least partly associated with clinical effects and adverse reactions, simultaneous determination of ticagrelor and M8 is necessary for interpreting clinical outcomes following administration of ticagrelor. In this context, we decided to develop a novel synthetic method for M8. Using commercially available ticagrelor and synthetized M8, we developed a simple LC/MS method that would be suitable for general laboratories in hospitals and academic institution.
The total synthesis of ticagrelor derivatives, including M8 (

Structures of ticagrelor (
Results and discussion
Preparation of M8
Initially, the three hydroxyl groups in ticagrelor (
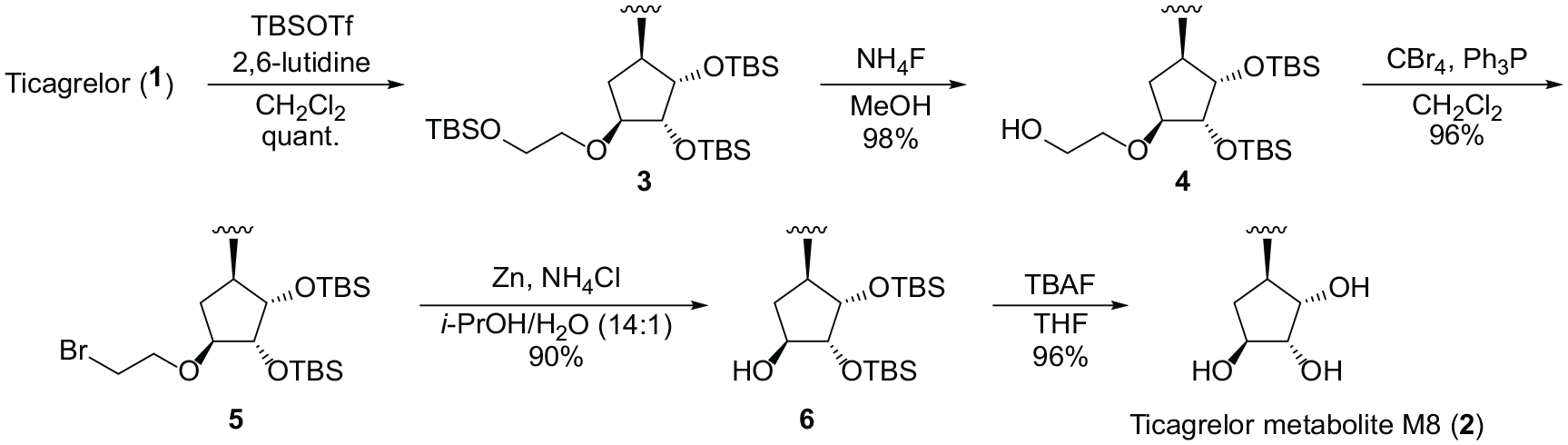
Synthesis of ticagrelor metabolite M8 (
Identification of ticagrelor and M8 by LC/MS
The LC/MS traces obtained in selected ion monitoring mode for a standard solution containing a mixture of ticagrelor, M8, and ticlopidine (the internal standard) at higher range are shown in Figure 2. Ticlopidine is a readily available antiplatelet agent for research and is never used concomitantly with ticagrelor in clinical settings; therefore, we considered that ticlopidine would be a good internal standard in this assay. Clear separation of the peaks corresponding to ticagrelor, M8, and ticlopidine was obtained without substantial ion suppression between ticagrelor and M8 using the LC/MS conditions described in the method section. The peaks were detected at approximately 4.2, 4.2, and 8.0 min, respectively.
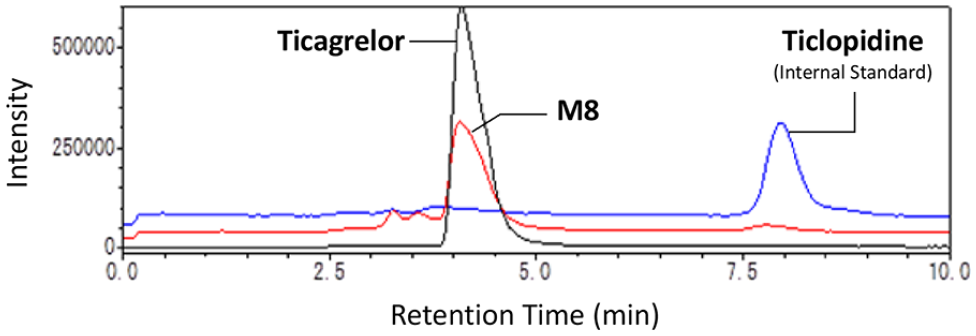
Typical LC/MS profile of a mixture of ticagrelor, M8, and ticlopidine (internal standard).
Figure 3 shows the calibration curves of ticagrelor and M8 in human plasma and under conditions for a metabolism assay containing human liver microsomes (approximately 0.28 mg/mL) deactivated by phosphoric acid, with an ethylenediaminetetraacetic acid (EDTA) and nicotinamide adenine dinucleotide phosphate (NADPH)-generating system in potassium phosphate buffer. Linear calibration curves for ticagrelor and M8 were obtained up to 100 and 50 µM, respectively. The intra- and inter-day coefficients of variation were within 10% over all concentration ranges for both ticagrelor and M8. The percentage of recovery of ticagrelor and M8 from human plasma in this protocol was 94.1% ± 11.7% and 103.5% ± 16.1%, respectively. The lowest plasma concentration of ticagrelor in our assay was 0.56 µM, whereas the average plasma concentration range of ticagrelor after 60–90 mg twice daily would be from 0.2 to 2.5 µM according to its package insert. 16 Therefore, there is a limitation in detecting the lower plasma concentration range of ticagrelor in clinical samples. Further drug–drug interaction studies would be possible within these concentration ranges using our LC/MS assay.

Calibration curves of (a) ticagrelor and its (b) active metabolite M8 in (c) human plasma and those in (d) human liver microsome.
Conclusion
We accomplished a concise synthesis of the major metabolite M8 (
Experimental
Materials and methods
1H NMR spectra were recorded at 300 MHz (JNM-AL300, JEOL, Tokyo, Japan) or 400 MHz (JNM-ECS400, JEOL). The chemical shifts are expressed in ppm relative to tetramethylsilane (δ = 0) as an internal standard (CDCl3 or CD3OD solution). Splitting patterns are indicated as follows: s, singlet; d, doublet; t, triplet; q, quartet; quint, quintet; sext, sextet; sep, septet; m, multiplet; br, broad peak. Infrared (IR) spectra were measured on an IR spectrometer (VALOR-III, JASCO, Tokyo, Japan) and are reported in wavenumbers (cm−1). Low-resolution MS and high-resolution mass spectrometry (HRMS) were performed on a mass spectrometer (JMS 700, JEOL; FAB mode) with a direct inlet system. Optical rotations were measured on a polarimeter (P-2200, JASCO) using a cell with an optical path length of 100 mm. Column chromatography was performed on silica gel (40–100 mesh). Analytical thin-layer chromatography was conducted using 0.25 mm silica gel 60 F plates.
A chromatographic system comprising two pumps (LC-20AD, Shimadzu, Kyoto, Japan), an automatic sample-injector (SIL-20A, Shimadzu), and a single-quadrupole mass spectrometer (LCMS-2010, Shimadzu) was used for our quantitative analysis. The mass spectrometer was operated in positive ion mode using electrospray ionization. Chromatographic separation was performed using a C18 column (Capcell Pak C18 MG II, Shiseido Co., Ltd., Tokyo, Japan; 3 µm, 50 mm × 4.6 mm ID) with a 10-mm guard column maintained at 40 °C by a temperature-controlled column oven (CTO-20A, Shimadzu). The mobile phase was a 10/90 (v/v) mixture of 10 mM ammonium acetate aqueous solution (10%; solvent A) and 100% acetonitrile (90%; solvent B). The flow rate of the mobile phase was maintained at 0.2 mL/min, and the injection sample volume was 2 µL. The analytes separated on the column were detected by MS with the following settings: collision energy of 20 V, capillary temperature of 220 °C, ionization voltage of 4.5 kV in positive mode, sheath gas at 60 psi, and nitrogen auxiliary gas at 10 psi. Single-ion mode was used with m/z values of 523.25, 479.25, and 264.05 for ticagrelor, M8, and ticlopidine (internal standard), respectively. Ticagrelor and ticlopidine were purchased from Sigma-Aldrich Japan Inc. (Tokyo, Japan). Data acquisition and analysis were performed using the LC/MS Solution software (version 3, Shimadzu). To obtain the calibration curves in human plasma, 100 µL of standard solutions containing ticagrelor (2.25 to 225 µM), M8 (1.27 to 63.2 µM), and ticlopidine (100 ng/mL) were added to 400 µL of human plasma. Then 5 mL of diethyl ether were added to each standard sample and 4 mL of supernatants were collected after shaking and centrifugation followed by redissolution with 100 µL of mobile phase. To obtain the calibration curves under condition for a metabolic assay, ticagrelor, M8, and ticlopidine (10 µL each) were added to a mixture of EDTA and NADPH generating system, potassium phosphate buffer, and human liver microsomes (H0610, Sekisui Medical Co., Ltd., Tokyo, Japan) deactivated by phosphoric acid (final volume of 470 µL) prior to add substrates to give the final concentrations (ticagrelor-M8-ticlopidine: 10-5-50 µM, 50-25-50 µM, and 100-50-50 µM). After 20 min centrifugation (18,000g, 4 °C), ticagrelor, M8, and ticlopidine were extracted from 200 µL of the supernatant using 5 mL of diethyl ether followed by redissolution with 30 µL of the mobile phase, and then injected into the LC/MS system.
Tris-TBS ether 3
TBSOTf (0.20 mL, 0.87 mmol) was added to a stirred solution of
1H NMR (500 MHz, CDCl3) δ: –0.51 (3H, s), –0.15 (3H, s), 0.09 (3H, s), 0.10 (3H, s), 0.14 (6H, s), 0.68 (9H, s), 0.91 (9H, s), 0.95 (9H, s), 1.01 (3H, t, J = 7.5 Hz), 1.30-1.41 (2H, m), 1.73 (2H, sext, J = 7.5 Hz), 2.02 (1H, m), 2.15 (1H, ddd, J = 14.0, 7.5, 1.0 Hz), 2.57 (1H, ddd, J = 17.0, 10.0, 7.5 Hz), 3.00–3.19 (3H, m), 3.55 (1H, dt, J = 10.0, 5.5 Hz), 3.60 (1H, dt, J = 10.0, 5.0 Hz), 3.72–3.85 (3H, m), 3.93 (1H, d, J = 3.0 Hz), 4.61 (1H, dd, J = 8.5, 3.5 Hz), 5.14 (1H, q, J = 9.0 Hz), 6.20 (1H, br s), 6.99–7.27 (3H, m). IR (CHCl3) 3268, 2929, 1613, 1521, 1327, 1098, 836 cm-1. [α] –66.4 (c = 0.09, CHCl3). HRMS (FAB+) m/z: [M + H]+ Calcd for C41H71F2N6O4SSi3 865.4533; Found 865.4537.
Primary alcohol 4
NH4F (65.6 mg, 1.77 mmol) was added to a stirred solution of
1H NMR (500 MHz, CDCl3) δ: –0.53 (3H, s), –0.16 (3H, s), 0.135 (3H, s), 0.143 (3H, s), 0.70 (9H, s), 0.94 (9H, s), 0.99 (3H, t, J = 7.0 Hz), 1.31–1.42 (2H, m), 1.65–1.80 (2H, m), 2.17 (1H, td, J = 8.0, 3.0 Hz), 2.21 (1H, dd, J = 15.0, 6.5 Hz), 2.76 (1H, ddd, J = 17.0, 10.5, 6.0 Hz), 2.82 (1H, br s), 3.00–3.18 (3H, m), 3.59 (1H, ddd, J = 10.0, 7.5, 3.0 Hz), 3.66 (1H, ddd, J = 10.0, 5.0, 2.5 Hz), 3.72 (1H, m), 3.76–3.86 (2H, m), 4.09 (1H, m), 4.83 (1H, dd, J = 7.5, 3.5 Hz), 5.27 (1H, ddd, J = 10.5, 8.0, 7.0 Hz), 6.41 (1H, br s), 7.00–7.28 (3H, m). IR (CHCl3) 3425, 3269, 2929, 1612, 1522, 1323, 1104, 838 cm-1. [α] –78.5 (c = 0.23, CHCl3). HRMS (FAB+) m/z: [M + H]+ Calcd for C35H57F2N6O4SSi2 751.3669; Found 751.3674.
Bromide 5
Ph3P (57.9 mg, 0.221 mmol) and CBr4 (95.7 mg, 0.289 mmol) were added to a stirred solution of
1H NMR (500 MHz, CDCl3) δ: –0.48 (3H, s), –0.14 (3H, s), 0.15 (6H, s), 0.69 (9H, s), 0.95 (9H, s), 1.01 (3H, t, J = 7.0 Hz), 1.31–1.42 (2H, m), 1.74 (2H, sext, J = 7.0 Hz), 2.17 (1H, td, J = 8.0, 3.0 Hz), 2.30 (1H, ddd, J = 14.5, 8.0, 2.5 Hz), 2.74 (1H, ddd, J = 14.5, 10.5, 7.0 Hz), 3.03–3.18 (3H, m), 3.50 (2H, t, J = 6.0 Hz), 3.75 (1H, dt, J = 10.5, 6.5 Hz), 3.80 (1H, dt, J = 7.0, 2.0 Hz), 3.89 (1H, dt, J = 11.0, 6.0 Hz), 4.11 (1H, m), 4.75 (1H, dd, J = 8.5, 4.0 Hz), 5.27 (1H, dt, J = 9.5, 8.0 Hz), 6.41 (1H, br s), 7.02–7.28 (3H, m). IR (CHCl3) 3268, 2929, 1613, 1520, 1327, 1117, 837, 775 cm-1. [α] –57.4 (c = 0.20, CHCl3). MS (FAB+) m/z: 813 [MH+], 815 [MH+ + 2]. HRMS (FAB+) m/z: [M + H]+ Calcd for C35H56BrF2N6O3SSi2 813.2825; Found 813.2826.
Secondary alcohol 6
Zn (646 mg, 9.88 mmol) and NH4Cl (26.4 mg, 0.494 mmol) were added to a stirred solution of
1H NMR (500 MHz, CDCl3) δ: –0.53 (3H, s), –0.21 (3H, s), 0.117 (3H, s), 0.124 (3H, s), 0.77 (9H, s), 0.90–0.97 (3H, m), 0.93 (9H, s), 1.30–1.45 (2H, m), 1.57–1.73 (2H, m), 1.99 (1H, dd, J = 15.0, 4.0 Hz), 2.20 (1H, m), 2.91 (1H, m), 3.01 (1H, ddd, J = 15.5, 12.0, 6.0 Hz), 3.07 (1H, m), 3.15 (1H, m), 3.97–4.08 (2H, m), 4.90 (1H, dd, J = 7.0, 3.0 Hz), 4.97–5.07 (1H, m), 5.27 (1H, ddd, J = 11.5, 7.0, 4.0 Hz), 6.66 (1H, br s), 6.96–7.15 (3H, m). IR (CHCl3) 3274, 2930, 1610, 1521, 1328, 1124, 838 cm-1. [α] –3.4 (c = 0.32, CHCl3). HRMS (FAB+) m/z: [M + H]+ Calcd for C33H53F2N6O3SSi2 707.3406; Found 707.3406.
M8 (2 )
Tetra-n-butylammonium fluoride (TBAF; 1 M solution in tetrahydrofuran (THF), 0.31 mL, 0.31 mmol) was added to a stirred solution of
1H NMR (500 MHz, CD3OD) δ: 0.91 (3H, t, J = 7.5 Hz), 1.39 (1H, m), 1.49 (1H, m), 1.55–1.68 (2H, m), 2.08–2.18 (2H, m), 2.79 (1H, ddd, J = 14.0, 9.5, 7.0 Hz), 2.91 (1H, m), 3.07 (1H, m), 3.15 (1H, m), 4.02 (1H, dd, J = 4.5, 2.5 Hz), 4.13 (1H, ddd, J = 7.5, 5.0, 2.5 Hz), 4.85 (1H, m), 5.12 (1H, q, J = 8.0 Hz), 7.05 (1H, m), 7.11–7.25 (2H, m). 13C NMR (125 MHz, CD3OD) δ: 13.6, 15.9, 23.9, 25.6, 34.1, 34.8, 36.8, 63.0, 75.5, 76.5, 78.5, 116.4, (JF-C = 18.0 Hz), 123.9 (JF-C = 3.5, 7.1 Hz), 124.8, 140.2, 150.1 (JF-C = 13.1, 245.3 Hz), 150.7, 151.5 (JF-C = 13.1, 246.5 Hz), 155.8, 172.1. IR (CHCl3) 3286, 1615, 1522, 1326 cm−1. [α] –48.5 (c = 1.36, CHCl3). HRMS (FAB+) m/z: [M + H]+ Calcd for C21H25F2N6O3S 479.1677; Found 479.1680.
Footnotes
Declaration of conflict of interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by a grant from the Dementia Drug Resource Development Center, Project S1511016, the Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan.
