Abstract
Panax bipinnatifidus belongs to the ginseng genus and it is used in traditional Vietnamese and Chinese medicine. Phytochemical studies of the roots of this plant led to the isolation of two new oleanane triterpenoid saponins, panabipinoside A and panabipinoside B, and three known compounds, ginsennoside Ro, 3-O-β-D-glucopyranosyl-(1→3)-β-glucuronopyranosyl oleanolic acid, and spinasaponin A 28-O-glucoside. Their structures are established by extensive spectroscopic analysis (IR, high-resolution electrospray ionization mass spectrometry, and nuclear magnetic resonance) and by comparison of the spectral data with those reported in the literature. The anti-inflammatory activity of the isolated compounds is evaluated by their inhibition of nitric oxide production in lipopolysaccharide stimulated RAW 264.7 cells. Compounds
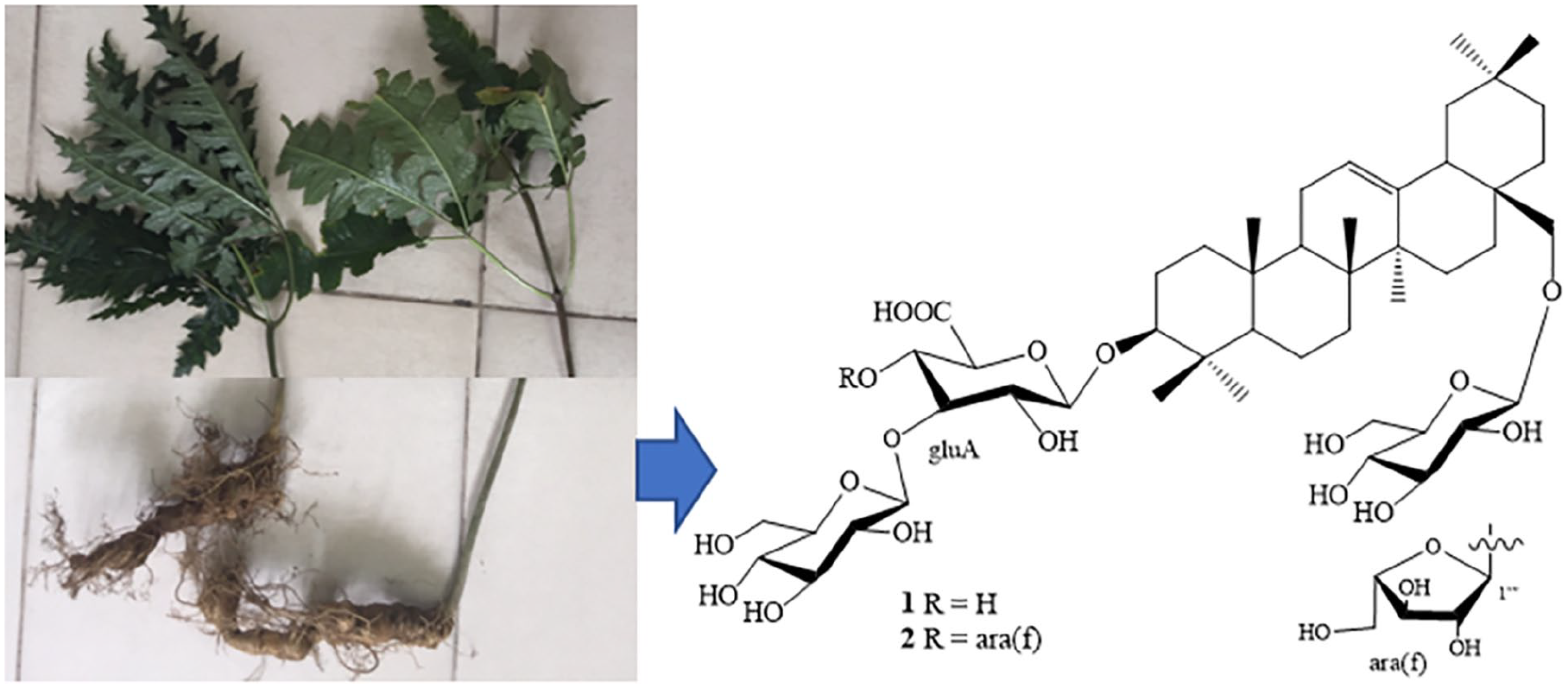
Introduction
Nitric oxide (NO) is a very short half-life molecule. It is known to be a ubiquitous signaling molecule involved in numerous important biological processes in the body such as neurotransmission, in the vascular system and in immune defenses. NO also plays a key role in the pathogenesis of inflammatory diseases. 1 In mammalian cells, the formation of NO is catalyzed by inducible nitric oxide synthase (iNOS) enzymes. In the presence of NADPH and oxygen, iNOSs oxidize L-arginine into L-citrulline and NO. The NO produced aids host defenses by killing invaders such as microorganisms and cancer cells. However, an excess production of NO can cause host and tissue damage which lead to acute and chronic inflammation.2,3 Thus, the level of NO produced by iNOS can be reflected in the degree of inflammation and is considered as an indicator for monitoring inflammatory processes. In addition, NO has been shown as a potential promoter of several types of cancer such as breast, cervical, lung, and gastrointestinal cancers. 4 An understanding of NO inhibitors can be helpful in reducing the risk of cancer.
Panax, also known as the ginseng genus, which is a member of the Araliaceae family, consists of 18 species.
5
Most species of the genus Panax are distributed in the Himalayas, Nepal, and China. Previously, the southern area of Yunnan province (China) and the northern provinces of Vietnam near the China–Vietnam border, which are located at latitude 23°N, were considered to be the distribution limit of the Asia Panax genus. Only 2 of the 18 species of this genus are native to North America. Panax is very popular in Asia and has been used as an edible food and as a tonic and has history of use as medicine over four millennia. The chemical composition of the Panax genus can be classified into four main groups: saponins, polyacetylenes, polysaccharides, and flavonoids.6,7 Besides, there are eight other sub-groups, including phenolics, sterols, carbohydrates, proteins, amino acids, minerals, lipids, and fatty acids.7,8 Several plants of the Panax species have long been used as medicinal herbs in oriental countries, for example, P. ginseng C.A. Meyer (Korean ginseng), P. quinquefolius L. (American ginseng), and P. notoginseng F.H. Chen (Notoginseng). Panax bipinnatifidus Seem. belongs to the ginseng genus, and is distributed in China, Nepal, and Vietnam. In Vietnam, it was found in the Hoang Lien Son mountains at an altitude of 1800–2400 m and grows wild under the canopy of a tropical forest.
9
According to traditional medicine, P. bipinnatifidus has been used to stop bleeding, to cure hemorrhages and nosebleeds, to improve memory, and to reduce cancer risk and blood sugar levels in diabetes.9–11 Recently, more than 10 saponins isolated from P. bipinnatifidus roots have been reported, all of which have the oleanane-type structure.12,13 In this study, with the aim of continuing to research on bioactive compounds in P. bipinnatifidus from Vietnam, five saponins were successfully isolated from the roots of this species including two new oleanane triterpenoid saponins, and three known compounds. The inhibition of NO production in lipopolysaccharide (LPS) stimulated RAW 264.7 cells by compounds
Results and discussion
Compound
The NMR data of compounds

NMR: nuclear magnetic resonance; nd: not detected.
Measured at 125 MHz.
Measured at 500 MHz.
Measured in CD3OD.
Measured in dimethyl sulfoxide d6 (DMSO-d6).
indicates overlapped signals.
Signals were detected from heteronuclear multiple bond correlation (HMBC) spectra.

Chemical structures of compounds
Compound
Compounds
Saponins have been reported to be major components responsible for numerous pharmacological properties of the Panax species. Most saponins have anti-cancer and anti-inflammatory properties. Inhibition of NO productions is not only part of the explanation for the anti-inflammatory activity but also the use of P. bipinnatifidus in traditional medicine to reduce the risk of cancer.
Saponins
Effects of compounds
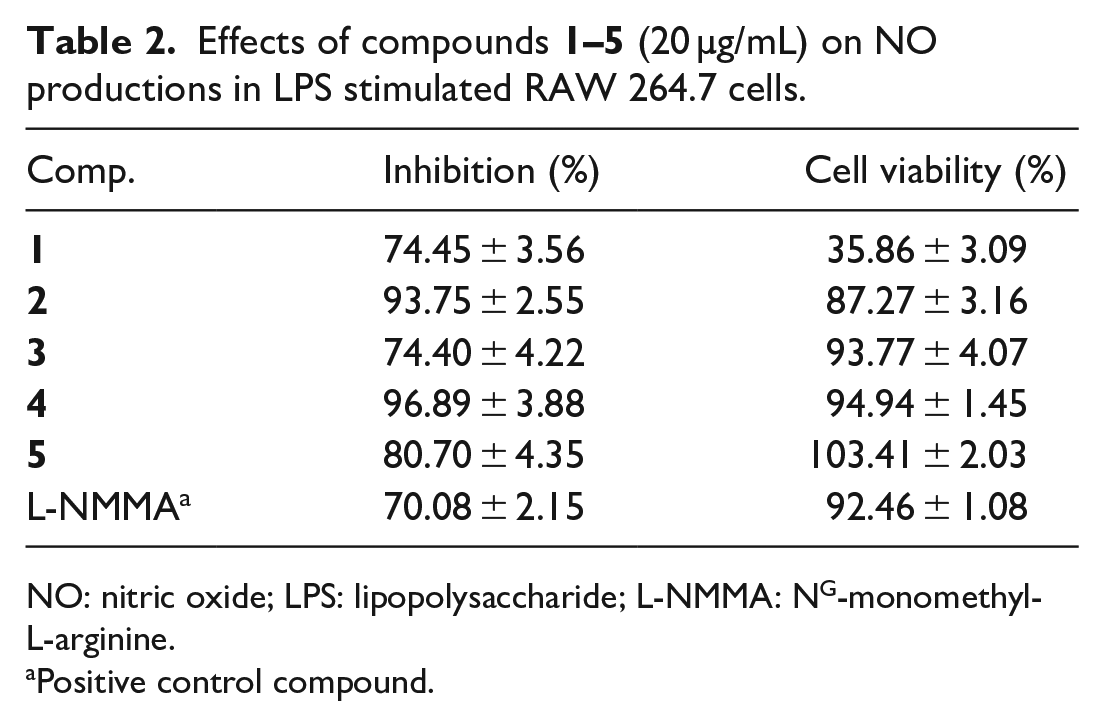
NO: nitric oxide; LPS: lipopolysaccharide; L-NMMA: NG-monomethyl-L-arginine.
Positive control compound.
Effects of compounds

NO: nitric oxide; LPS: lipopolysaccharide; L-NMMA: NG-monomethyl-L-arginine.
Positive control compound.
Conclusion
Two new oleanane triterpenoid saponins, named panabipinoside A (
Material and methods
General
Optical rotations were measured on a Jasco P2000 polarimeter. IR spectra were recorded on a Spectrum Two Fourier transform infrared (FTIR) spectrometer. HR-ESI-MS were measured on an Agilent 6530 Accurate Mass Q-TOF LC/MS or a Thermo Scientific Q Exactive:tm: Focus Hybrid Quadrupole-Orbitrap. NMR spectra were recorded on a Bruker 500 MHz spectrometer. Preparative high-performance liquid chromatography (HPLC) were run on an Agilent 1100 system including a quaternary pump, an autosampler, a DAD detector, and a preparative HPLC column YMC J’sphere ODS-H80 (4 µm, 20 × 250 mm). Anisocratic mobile phase with a flow rate of 3 mL/min was used for pre-HPLC. Compounds were monitored at wavelengths of 205, 230, 254, and 280 nm. Flash column chromatography was performed using silica gel, reversed phase C-18, and Dianion HP-20 resins as the stationary phase. TLC was carried out on pre-coated silica gel 60 F254 and RP-18 F254S plates. The spots were detected by spraying with an aqueous solution of 5% H2SO4 followed by heating with a heat gun.
Plant material
The roots of Panax bipinnatifidus Seem. were collected at Sapa, Lao Cai, Vietnam, in May 2019, and identified by Dr Do Ngoc Dai, Department of Forestry, Nghe An University of Economics. A voucher specimen (coded: SVD-BK2019) was deposited at the School of Chemical Engineering, Hanoi University of Science and Technology.
Extraction and isolation
The air-dried roots of P. bipinnatifidus (1.0 kg) were ultrasonically extracted in C2H5OH (3 L × 3, 80 °C). After filtration, the extracts were concentrated in vacuo to dryness. The obtained C2H5OH residue (350 g) was suspended in H2O (1 L) and then partitioned with n-hexane and ethyl acetate (each 2.5 L × 3), successively, to give n-hexane and ethyl acetate soluble fractions in the weights of 82.5 and 35.72 g, and the water layer. The water layer was subjected to a Dianion HP-20 column and eluted stepwise using a mixture of H2O and C2H5OH (100:0→4:96; v/v) to give four fractions (fr. 1.1–fr. 1.5). After checking these fractions by TLC, the sub-fraction 1.3 (3 g) was first separated on a reverse phase C-18 resin column chromatography, eluting with acetone/water (1/1.2, v/v) to give seven smaller fractions, 1.3A–G. Fraction 1.3B was further purified by HPLC using a J’sphere ODS H-80, 250 mm × 20 mm column, MeCN in H2O (20%, v/v), and a flow rate of 3 mL/min to yield compound
Panabipinoside A (
Colorless amorphous powder, [∝] D 25: +12.2 (c 0.1, MeOH);IR (KBr): νmax 3401 (broad), 2944, 1732, 1612, 1076, 1028 cm−1; HR-ESI-MS: m/z 941.51031 [M–H]− (calcd for [C48H77O18], 941.51099).
1H NMR (CD3OD, 500 MHz) and 13C NMR (CD3OD, 125 MHz) data: see Table 1.
Panabipinoside B (
Colorless amorphous powder, [∝]
D
25: +16.7 (c 0.1, MeOH); IR (KBr): νmax 3406 (broad), 2945, 1732, 1613, 1077, 1029 cm−1; HR-ESI-MS: m/z 1073.54980 [M–H]− (calcd for [C53H85O22], 1073.55325).
1H NMR (CD3OD, 500 MHz) and 13C NMR (CD3OD, 125 MHz) data: see Table 1.
Nitric oxide assay
Refer to Supplemental Material
Acid hydrolysis and confirmation of the monosaccharides
Refer to Supplemental Material
Supplemental Material
sj-pdf-1-chl-10.1177_17475198211018988 – Supplemental material for Panabipinoside A and panabipinoside B, two new oleanane triterpenoid saponins from the roots of Panax bipinnatifidus with nitric oxide inhibitory activity
Supplemental material, sj-pdf-1-chl-10.1177_17475198211018988 for Panabipinoside A and panabipinoside B, two new oleanane triterpenoid saponins from the roots of Panax bipinnatifidus with nitric oxide inhibitory activity by Phan Van Kiem, Vu DinhHoang, Nguyen Thi Hoang Anh, Dinh Thi Phuong Anh, Do Thi Trang and Bui Huu Tai in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by the Ministry of Education and Training of Vietnam under Grant B2019_BKA.02 (2019–2020).
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
There are no human subjects in this article and informed consent is not applicable.
Human and animal rights
This article does not contain any studies with human or animal subjects.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
