Abstract
Scoparia dulcis L. is one of the edible widely distributed Scropholariaceae species in Asia, Africa and America. It is used in the treatment of respiratory and inflammatory diseases, diabetes, hypertension, cancer, hepatitis and tuberculosis. A phytochemical investigation on S. dulcis led to the isolation of two new acyclic diterpenes Acetic acid 6-hydroxy-2-(6-hydroxy-4-methyl-hex-4-enylidene)-4,8-dimethyl-undeca-4,8-dienyl ester (
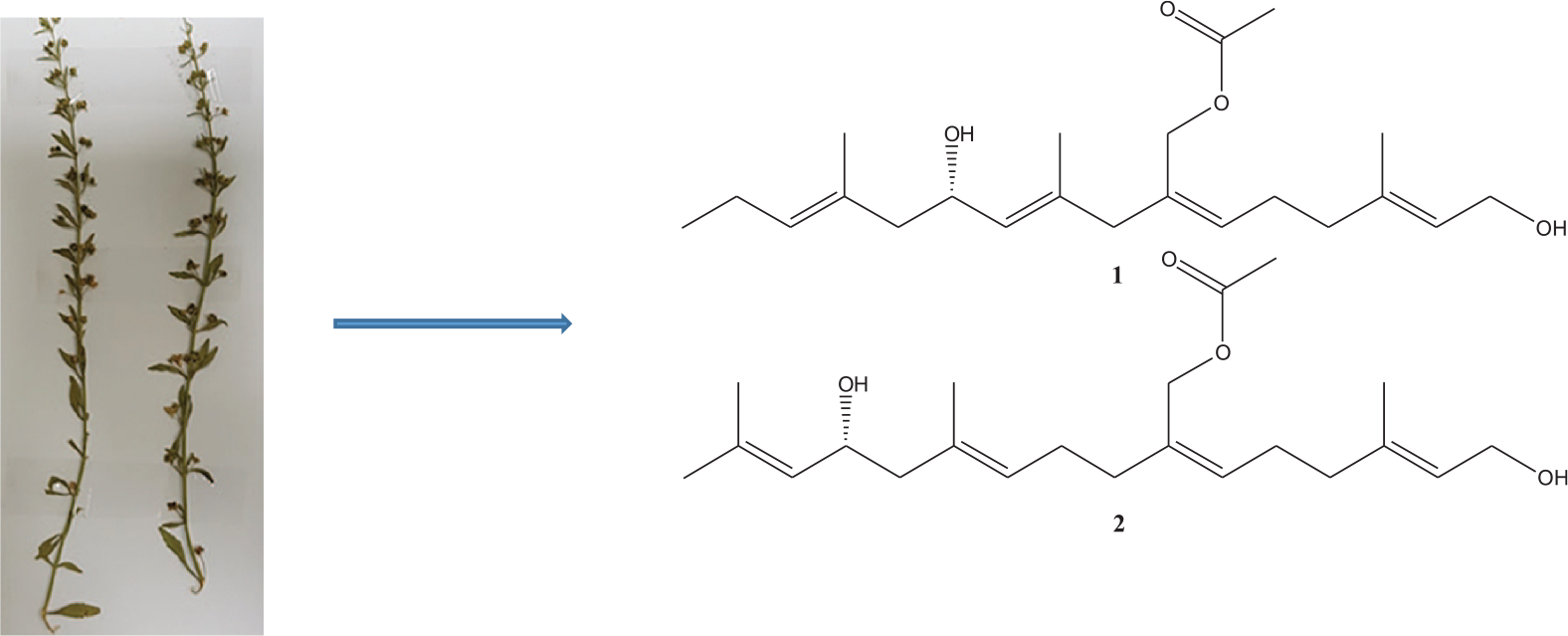
Scrophulariaceae, the figwort family of flowering plants, have about 65 genera and 1700 species which are globally distributed. 1 The plants are annual and perennial herbs, as well as one genus of shrubs. The genus Scoparia of this family comprises 32 species of flowering plants. 2
Scoparia dulcis L., which belongs to genus Scoparia also known as sweet broom weed, is a perennial edible herb with white flowers and serrated leaves, dispersed in tropical and subtropical area of Asia, Africa and America. 3 Folklorically it is used to treat ailments like bronchitis, diabetes, hypertension, cancer, fever, haemorrhoids, stomach ache, ulcers hepatitis and tuberculosis4,5 and also used to treat respiratory and inflammatory diseases in South America. 6
S. dulcis has been extensively studied for its chemical constituents like triterpenes,2,4,7,8 diterpenes,4,7,9–15 benzoxazinoid, flavonoids, iridoid and iridoid glycosides compounds3,16 and other constituents coixol, glutinol, glutinone, friedelin, betulinic acid, and tetratriacontan-1-ol 17 are also reported.
Several compounds isolated from S. dulcis have been found to have different types of biological activities.3,18 Compounds such as scoparic acid D, scoparic acid A and 8-hydroxytricetin-7-glucronide are reported to possess anti-diabetic activity;18–20 scopadulin holds anti-viral; 14 glutinol has analgesic; 21 scoparinol possesses analgesic, diuretic and anti-inflammatory activity; 22 scopadulciol, scopadulcic acid B, and diacetyl scopadol are inhibitor of gastric H+, K(+)-ATPase.23,24 Iso-dulcinol, 4-epi-scopadulcic acid B, dulcidiol, scopadulciol and scopadiol have been examined for cytotoxicity. 10
In this paper, we report isolation and structure elucidation of two new acyclic diterpenes Acetic acid 6-hydroxy-2-(6-hydroxy-4-methyl-hex-4-enylidene)-4,8-dimethyl-undeca-4,8-dienyl ester (
Results and discussion
A phytochemical work on acetonitrile fraction of S. dulcis led to the isolation of two new compounds

Structures of compound
Compound
In COSY spectrum, it was observed that

HMBC and COSY correlations of compound
In a similar manner COSY cross peaks between δH 5.07 m (1H, H-10)/ 4.25 m (1H, H-11)/ 2.11 (1H, Ha-12, dd, J = 7.0,7.0 Hz) and 1.93 (1H, Hb-12, dd, J = 7.0,7.0 Hz) corresponding to δC 126.1 (C-10), 66.4 (C-11) and 48.5 (C-12), and δH 5.05 m (1H, H-14)/ 2.02 m (2H, H-15)/ 1.58 t (3H, H-16) corresponding to δC 130.3 (C-14), 26.8 (C-15) and 18.5 (C-16), respectively, together with HMBC correlations building third and fourth segment of the molecule individually. In 1H NMR, signals at δH 4.35 d (7.0 Hz) and 4.46 t (7.0 Hz) are ascribed to two hydroxyl protons and their attachment to C-1 (δC 58.0) and at C-11 (δC 66.4) were established via HMBC correlations (Figure 2) which was in agreement with the previously reported data for oxymethylene and oxymethine, respectively. 3 Occurrence of carbonyl carbon was defined by a signal appearing at δC 170.8 (C-21).
The oxymethylene signals δH/δC 4.55 br s (2H, H-20) /61.5 (C-20) showing J3 correlation with C-21 and the methyl signals δH/δC 2.0 s (3H, H-22)/21.1(C-22) showing J2correlation with C-21 consequently forming a methylene acetate moiety.
29
The cross peaks in HSQC spectrum δH/δC 1.64 s (3H, H-17)/25.9 (C-17), 1.57 s (3H, H-18) / 16.8 (C-18) and 1.58 s (3H, H-19) / 16.4 (C-19) are attributable for three vinyl methyls and their allocation at δC 131.4 (C-13), 132.5 (C-9) and 135.5 (C-3), respectively, were based on HMBC correlations.
29
The aggregate structure of
The vinyl methyl proton δH 1.58 (H-19) showing J2 correlation with δC 135.5 (C-3) and J3 correlation with 126.1 (C-2) reversely δH 5.27 showing J3 correlation with 16.2 (C-19) suggested C-2 and C-3 are connected with each other through an olefinic bond. Beside this δH 5.27 (H-2) also showing J3 correlation with 39.5 (C-4), also δH 1.97 (H-4) exhibiting J3 correlation with δC 126.1 (C-2) and δC 16.4 (C-19). The protons resonating at δH 2.16 (H-5) presented J3 connection with and δC 134.0 (C-7) and J2 connection with δC 130.3 (C-6) suggesting the olefinic linkage between C-6 and C-7. Furthermore, δH 2.16 also giving J3 connection with δC 135.5 (C-3) and J2 connection δC 39.5 (C-4). The oxymethylene protons at δH 4.55 of the methylene acetate moiety showing J2 correlation with δC 134.0 (C-7) and J3 correlation with δC 35.2 (C-8) and δC 130.3 (C-6) and proton at δH 1.97 (H-8) and δH 5.38 showing J3 correlation with δC 61.4 (C-20) secured the position of methylene acetate moiety at C-7. Another vinyl methyl proton resonation at δH 1.57 (H-18) showing J2 correlation with 132.5 (C-9) and J3 correlation with δC 126.1 (C-10) proposed olefinic bond between C-9 and C-10, J3 correlation is also observed between δH 1.57 (H-18) and δC 35.2 (C-8). The oxymethine proton δH 4.25 (H-11) showing J2 correlation with 48.5 (C-12) and J3 correlation with δC 132.5 (C-9). Signals at δH 5.07 (H-10) showing J2 correlation with δC 66.4 (C-11) and δC 132.5 (C-9) J3 connectivity with δC 35.2 (C-8) and δC 48.5 (C-12). Protons resonating at δH 1.64 (H-17) has already been assigned for vinyl methyl showing J2 and J3 connectivity with δC 131.4 (C-13) and δC 131.4 (C-14), respectively, proposing the olefinic linkage between C-13 and C-14 and also showing J3 connectivity with δC 48.5 (C-12). The H-12 protons δH 2.11 and 1.93 both showing J3 connectivity with δC 130.3 (C-14). The methylene protons resonating at δH 2.02 (H-15) showing J2 correlation with δC 130.3 (C-14) and J3 correlation with δC 131.4 (C-13). The terminal methyl proton δH 1.58 (H-16) showing J3 correlation with δC 130.3 (C-14) and J2 correlation with δC 26.8 (C-15).
On the basis of this spectral evidences, the structure of
Compound
1H and 13C NMR data of the isolated new compounds

The α configuration of OH at C-11 and C-13 of compounds
In the history of mankind, plants were the primary source of medicines. Throughout the world, a great number of plants as whole and their different parts were used as drug like entities to cure all kind of diseases. In the scientific community, the awareness that chemical entities present inside the plant material used folklorically is responsible for potential pharmacological activities either as single or in synergetic manner and these chemical entities can be isolated for their usage as a single agent came up in the 19th century in the context of the emerging natural science-based medicine and pharmacy. 31
The biological process of inflammation is very critical for the management of body homeostasis. It is very crucial for positively fighting with pathogens and to restoration of injured tissues. The process of inflammation is also involved in several severe complaints, such as asthma, chronic inflammatory bowel diseases, rheumatoid arthritis, neurodegenerative diseases, cancer and type-2 diabetes. 32
There are many complex (humoral and cellular) mechanisms of inflammation and these mechanisms involve in the factors of gene regulation such as the nuclear factor-kappa B (NF-κB) and also involved synthesis of signalling substance produced by immune system cells such as cytokines and prostaglandins.33,34 Usually many steroidal and nonsteroidal anti-inflammatory drugs (NSAIDs) and immunosuppressants, used for the treatment of inflammatory diseases, need long period of treatment and often have many serious side effects. The secondary metabolites produced by various medicinal plants have already been shown their potency in the management of inflammation and pain. 33
The extractives of various plants and their isolates have been examined in vitro for cytotoxicity by using a number of human cell lines such as stomach, prostate, colon, liver and so on. as well as animal cells such as kidney cells of monkey. Testing of toxicity through cell is a valuable and cost-effective and have short period testing time. This test is able to provide the information on the dose-dependent connection as well as the dose range for exposure and risks to human’s health. The cytotoxicity of plant extractive and isolates must be assessed before their influence in drug discovery while taken into consideration. 35
Out of the two new compounds, only compound
Compounds
Anti-inflammatory and cytotoxic activities of the isolated compounds from S. dulcis.
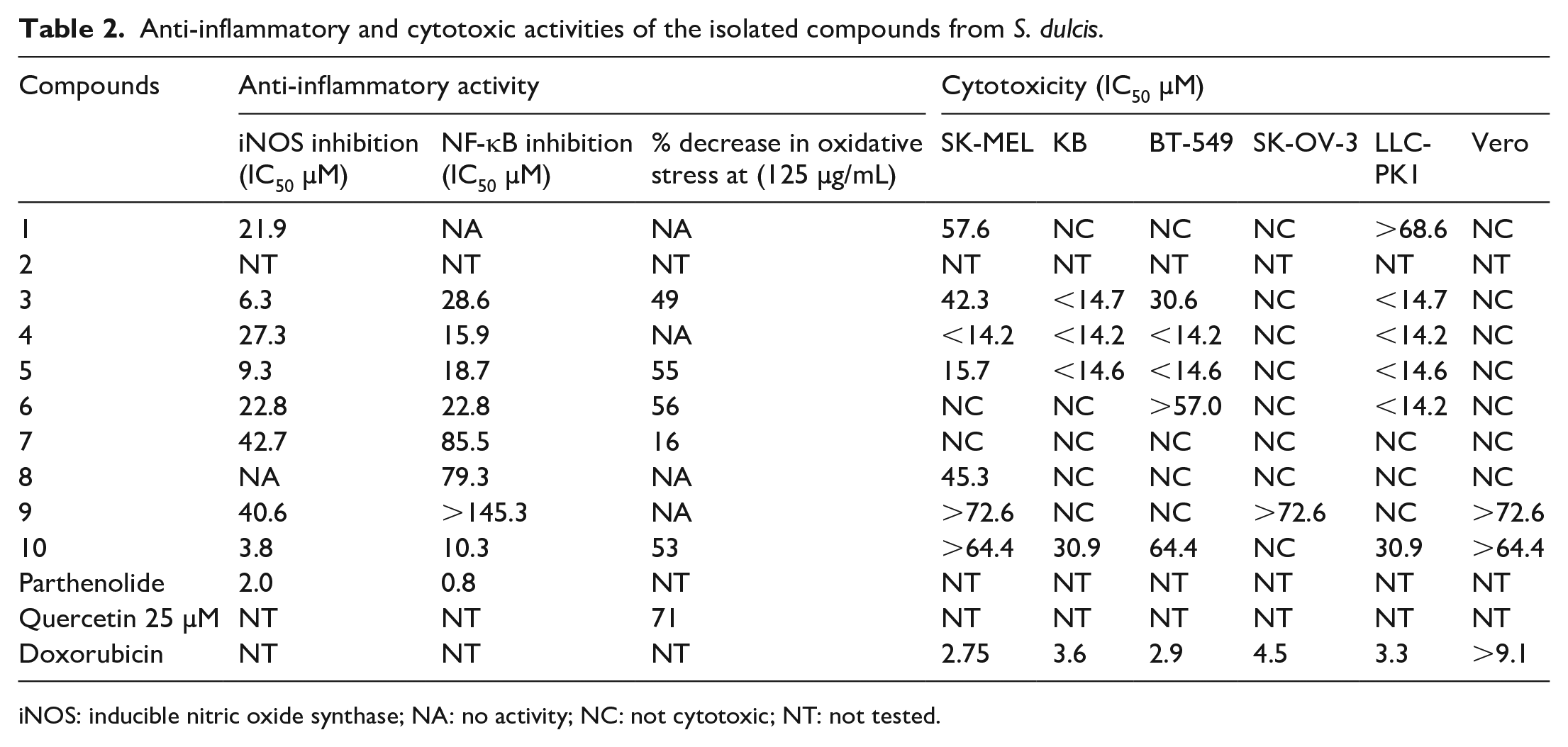
iNOS: inducible nitric oxide synthase; NA: no activity; NC: not cytotoxic; NT: not tested.
Conclusion
In conclusion, 10 different compounds including two new
Experimental
General experimental procedures
The ESI-HRMS are measured on Agilent Technologies 6200 series mass spectrometer. 1H NMR (700 MHz), 13C NMR (175 MHz) and 2D NMR spectra are measured on Bruker Avance NMR spectrometer. UV spectra are measured on UV 1601 PC (Shimadzu). Optical rotations are recorded on Jasco Polarimeter P-2000. Thin-layer chromatograph (TLC) is performed on precoated silica gel plates with UV254 fluorescence indicator (Merck). The chromatograms are visualized under UV light (at 254 and 366 nm) CAMAG UV cabinet and also spraying with anisaldehyde-sulphuric acid reagent followed by heating with heat gun to visualize the spots. Silica gel (230–400 mesh) and LiChroprep RP-18 (40–63 μm; Merck, Darmstadt) are utilized for column chromatography (CC). High-performance liquid chromatography (HPLC) is performed on a Shimadzu system (Kyoto, Japan), consisting of two LC-6AD semi-preparative solvent delivery pumps, bus module CBM-20A, a multi wavelength photodiode array detector (SPD-M20A), columns shim-pack PREP-ODS (H) Kit (A) 250 mm × 4.6 mm I.D. with 5 μm particles (B) 250 mm × 20 mm I.D., 5 μm.
Plant material
The plant material was collected in February 2013 from Haili near Dam, Kingdom of Saudi Arabia (KSA). The collected plant material was identified by the taxonomist Dr. M. Yusuf and a plant voucher # 16026 was deposited at the herbarium of the College of Pharmacy, King Saud University, Riyadh, KSA.
Extraction and isolation
The dried and coarsely ground plant material (2.070 kg) was extracted with dichloromethane (5 L × 3) at room temperature for 72 h (24 h × 3). The extract was filtered through filter paper (Whatman No.1) and solvent was evaporated to dryness at 40 °C under reduced pressure using Buchi Rotavapour (R 215) and yielded green solid mass 193.1 g. The dichloromethane extract was partitioned between hexane and acetonitrile (presaturated), yielded acetonitrile fraction 43.2 g and hexane fraction 145.6 g. Acetonitrile fraction 40.0 g (out of 43.2 g) was undertaken for column chromatography over normal phase silica gel that provided 28 fractions (Fr 1. to Fr. 28). Further extensive chromatographic work on these fractions including reverse phase column and semi-preparative HPLC yielded compounds
Compound 1
Colourless sticky material; [α] D 27 −2.4° (c 0.05, MeOH); UV (MeOH) λmax (log ε) 339 (2.53); 1H and 13C NMR, see Table 1; ESI-HRMS m/z 387.2504 calcd for C22H36O4 Na, 387.2511.
Compound 2
Colouless sticky material; [α]D27 −3.0° (c 0.06, MeOH); UV (MeOH) λmax (log ε) 249 (2.46); 1H and 13C NMR, see Table 1; ESI-HRMS m/z 387.2504 calcd for C22H36O4 Na, 387.2511.
Biological assays
Determination of NF-κB inhibition
Inhibition of NF-κB activity was determined in human chondrosarcoma (SW1353) cells through a reporter gene assay as described earlier. 37 The cells were transfected with NF-κB luciferase plasmid construct, seeded into the wells of 96-well plates (1.25 × 105 cells/well) and incubated for 24 h for confluency. Test compounds at various concentrations were added, and after 30 min, phorbol 12-myristate 13-acetate (PMA, 70 ng/mL) was added. After incubating further for 6−8 h, luciferase activity was detected using a Luciferase Assay Kit (Promega, Madison, WI, USA). The decrease in luciferase activity upon treatment with the test compound was calculated in comparison to untreated cells. Parthenolide served as the positive control. A control transcription factor Sp-1 was also included to determine the general toxicity and specificity of the compound towards NF-κB as described earlier. 38
Determination of iNOS inhibition
The inhibition of iNOS activity was determined in mouse macrophages (RAW264.7) according to a procedure described earlier. 37 In brief, macrophages were plated in 96-well plates (50,000 cells/well) and plates were incubated for confluency up to 24 h. Cells were treated with various concentrations of test compounds and after incubating for 30 min, LPS (5 μg/mL) was added and further incubated for 24 h. The nitrite production in cell culture supernatant was measured by Griess reagent. Percent decrease in nitrite level was calculated in comparison to the vehicle control. Parthenolide was included as a positive control.
Determination of the effect on oxidative stress
The decrease in cellular oxidative stress as a measure of antioxidant effect was determined in HepG2 cells as described earlier. 39 For the assay, the cells were seeded in the wells of a 96-well plate (60,000 cells/well) and incubated for 24 h. The cells were washed with phosphate-buffered saline and treated with the test compounds diluted in serum-free medium containing 25 μM DCFH-DA, for 1 h. The media was removed and the cells were treated with ABAP (600 μM). The plate was immediately read on a SpectraMax plate reader every 5 min for 1 h (37 °C, emission at 538 nm and excitation at 485 nm). The percent decrease in oxidative stress was calculated. Quercetin served as the positive control.
Determination of cytotoxicity
The cytotoxicity of the compounds was tested against a panel of cancer cell lines (SK-MEL, KB, BT-549, SK-OV-3) and kidney cell lines (LLC-PK1 and VERO). The cells were plated at a density of 25,000 cells/well and incubated for 24 h for confluency. After adding the test samples at various concentrations, cells were incubated for 48 hrs. The cell viability was determined by using a tetrazolium dye (WST-8). Percent decrease in cell viability was calculated in comparison to untreated cells. IC50 value was calculated from the concentration response curves. Doxorubicin was included as the positive control for cytotoxicity.
Supplemental Material
Supplementary_Information_of_scoparia_revised_updated – Supplemental material for Isolation and characterization of cytotoxic and anti-inflammatory constituents from Scoparia dulcis L
Supplemental material, Supplementary_Information_of_scoparia_revised_updated for Isolation and characterization of cytotoxic and anti-inflammatory constituents from Scoparia dulcis L by Mohammad Nur-e-Alam, Sarfaraz Ahmed, Muhammad Yousaf, Shabana I Khan, Ramzi A Mothana and Adnan J Al-Rehaily in Journal of Chemical Research
Footnotes
Acknowledgements
The authors extend their appreciation to Researchers Supporting Project number (RSP-2019/119), King Saud University, Riyadh, Saudi Arabia for funding this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
