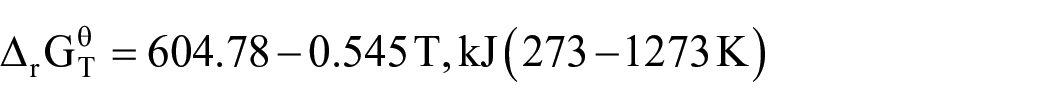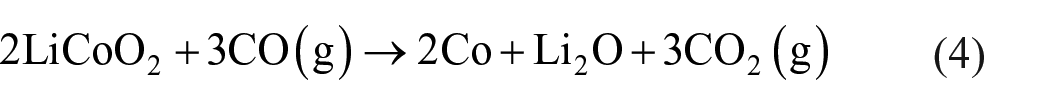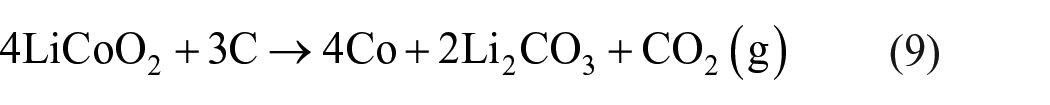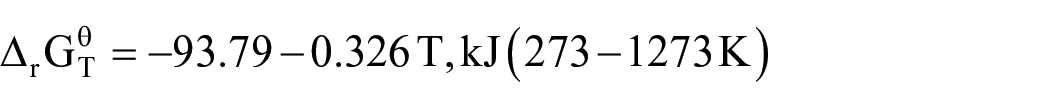Abstract
Carbothermal reduction to recover lithium-ion batteries is an environmentally friendly recycling method. This work provides a theoretical analysis of the thermodynamics and an experimental verification process for the carbon thermal reduction recovery of lithium cobaltate (LiCoO2), as well as an explanation of the microstructural changes. The reaction conditions are controlled to obtain the ideal recovery product. A thermodynamic graph of the possible reaction between LiCoO2 and graphite (C) and the reduction of cobalt oxide (CoO, Co2O3, Co3O4) by carbon monoxide (CO) is obtained by thermodynamic analysis. The feasibility of the carbothermic reduction reaction of LiCoO2 at high temperature is studied under standard atmospheric pressure. By controlling the reaction temperature (800 °C) and the ratio of the reactants (LiCoO2/C = 4:5), the reduction products cobalt monoxide (CoO) and lithium carbonate (Li2CO3) are obtained, and the recovery rates are 89% and 84%, respectively. From the perspective of the crystal structure, the reduction process of LiCoO2 is analyzed. The Li-O bond in the LiCoO2 crystal structure is destroyed by CO, which promotes destruction of the Li-O octahedral structure and the formation of a Co-O octahedron. The Li-O octahedron is transformed into a tetrahedral structure in Li2O, and Li2O then reacts with CO2 to form Li2CO3; the Co-O octahedrons combine to form a CoO crystal structure. When the temperature continues to rise, CoO is reduced to Co. The carbothermic reduction recovery method does not require any additional hazardous chemicals and can avoid secondary pollution during the recovery process. This research provides theoretical support for the industrial recycling of lithium-ion batteries.
Introduction
In recent years, with the steady increase in the demand for lithium-ion batteries from 3C products (computers, communications, and consumer electronics), as well as the gradual expansion of the market scale of new energy vehicles and the expansion of demand for energy storage batteries, the scale of production of lithium-ion batteries in China has expanded year-on-year. 1 The output of lithium-ion batteries was 15.722 billion, a year-on-year increase of 12.4% in China in 2019, 2 and the life span of a lithium-ion battery is generally 3–5 years. 3 The crystal form of the cathode material will also change during repeated charging and discharging, and the crystal structure of lithium cobaltate (LiCoO2) is transformed from a hexagonal crystal system into a monoclinic crystal system, as shown in Figure 1. Due to the monoclinic crystal system, the capacity decay is a serious problem, and the change of crystal type will prevent lithium-ions from freely being inserted and extracted in and out of the crystal structure, resulting in serious capacity decay and failure.4–6 When reaching the end of their service life, a large number of waste lithium-ion batteries will be produced. Generally, waste lithium-ion batteries contain 5%–15%, 2%–7%, and 0.5%–2% of cobalt, lithium, and nickel, respectively, as well as metal elements such as Cu and Al.7,8 The content of cobalt in natural cobalt ore is relatively low. Most cobalt forms symbiotic ores with other metals. The content of cobalt in the ore is only 0.01%–0.20%, while the cost of smelting is high. Moreover, China is a cobalt-poor country and requires a large amount of cobalt; the import volume of cobalt into China in 2019 was 124,000 tons. 9 There were about 500,000 tons of lithium-ion battery waste in 2019, and 90% of the cobalt in the waste can be recycled and reused. The recycling of lithium-ion batteries can avoid the waste of resources and has huge economic benefits. 10 The recycling of lithium-ion batteries is an obvious choice for energy conservation and environmental protection. From an environmental point of view, cobalt is a heavy metal element, nickel has obvious carcinogenicity, while burning or thermal decomposition of lithium carbonate will produce toxic lithium cobalt oxide, diaphragms, and other non-degradable materials, and electrolytes and the binder polyvinylidene fluoride (PVDF) will cause organic pollution and fluorine. 11

Schematic diagram of the changes in the crystal structure of LiCoO2.
The main active material in lithium-ion batteries is LiCoO2, and the key to recycling lithium-ion batteries is to separate and recover LiCoO2. LiCoO2 is converted into metals and their oxides or soluble metal salts that can be further utilized by the fire method, the wet method, and so on, so as to realize the recovery of waste LiCoO2 batteries.12–14 Previous studies on the reduction and recovery of lithium batteries mainly focused on methods such as wet acid leaching,15–17 chemical precipitation,18–20 bioleaching,21–23 electrochemical treatment,24,25 and so on. There are relatively few studies on the fire recovery of lithium-ion batteries. Tang and others studied the recovery and regeneration of LiCoO2-based waste lithium-ion batteries under vacuum conditions by carbothermal reduction pyrolysis. 26 The electroactive material is separated by a vacuum pyrolysis method, and then the obtained cathode material is mixed with graphite from the anode, and LiCoO2 is selectively converted into cobalt monoxide (CoO) and lithium carbonate (Li2CO3) by carbothermal reduction under vacuum conditions. The reaction mechanism relies on the fact that the attraction of graphite to oxygen is stronger than that of lithium and cobalt, which makes the internal structure of LiCoO2 unstable. The oxygen therein oxidizes the graphite and forms a reducing atmosphere to promote the pyrolysis and reduction of LiCoO2. 27 Thermodynamically, decreasing the amount of the reducing agent and lowering the operating temperature can suppress the generation of Co and leads to CoO. 28 However, considering the cost of vacuum conditions in industrial applications, the benefits of this method are limited. Li et al. 29 have used anaerobic roasting and wet magnetic separation to recover cobalt, lithium carbonate, and graphite from mixed electrode materials. The ferromagnetism of cobalt and the solubility of lithium carbonate enable the pyrolysis reduction products to be fully separated without additional chemical solutions, which saves on the costs of treating secondary pollution. 30 At present, the corresponding recycling and reuse of waste lithium-ion batteries is in the initial stage. Most of the recycling work only leads to the metal components of aluminum, copper, and so on, from the waste lithium-ion batteries, while other metals and electrolytes are directly discarded as waste that causes more direct harm to the environment. Although the elements cobalt and lithium can be further recovered from the batteries through metallurgical and chemical methods, the recovery costs are high, which is not conducive to industry. Therefore, finding an effective and large-scale method for the recycling of used lithium-ion batteries is a very important direction for our future development.31,32
On the basis of previous studies, our research attempts have been to recover the LiCoO2 cathode material through the carbon thermal reduction reaction under high temperature in the presence of an air atmosphere. On the basis of thermodynamic analysis of the carbon thermal reduction reaction of LiCoO2 batteries we now analyze and explain the reduction reaction process at the crystal structure level.
The high-temperature carbothermic reduction of LiCoO2 is carried out under an air atmosphere and the cathode material LiCoO2 of the waste lithium-ion battery is reduced and converted into the metal oxide CoO with high added value. Furthermore, LiCoO2 can be regenerated by the high-temperature solid-phase reaction of the obtained CoO and Li2CO3. 26 LiCoO2 can be selectively converted into Li2CO3 and CoO by changing the reduction temperature and the ratio of LiCoO2 to graphite; the optimum CoO reduction conditions are found. The Li2CO3 and CoO are separated by the wet aqueous solution separation method and the product recovery rate can be calculated. Pyrotechnical recycling can avoid complicated processes, high energy consumption, and secondary pollution of the common wet recycling. Compared with vacuum and oxygen-free atmosphere conditions, obtaining the required products by controlling the temperature and ratio of raw materials in an air atmosphere can simplify the reaction environment, thereby reducing the recycling cost, which is beneficial to industrial applications.
Results and discussion
Thermodynamic analysis
The positive electrode material LiCoO2 decomposes at above 900 °C (1173 K); the decomposition equation is as follows
Co3O4 is unstable above 900 °C (1173 K) and will continue to decompose to form CoO and O2. The reaction is as follows
On combining equations (1) and (2), the decomposition reaction equation of LiCoO2 is obtained as
During the high-temperature pyrolysis of the mixture of LiCoO2 and graphite in air, the possible reactions include chemical equations (3)–(8). The calculations and thermodynamic data for the Gibbs free energy of each reaction are derived from HSC Chemistry version 9.3. According to the thermodynamic data of each substance, the standard reaction Gibbs free energy in the temperature range of 273–1273 K is calculated
Overall, the chemical reaction equations (3)–(6) can be simplified as
According to equation (9), it can be seen that the ratio of LiCoO2 to graphite when it is fully reduced in an ideal state is 4:3, and the products obtained are Co, Li2CO3, and CO2.
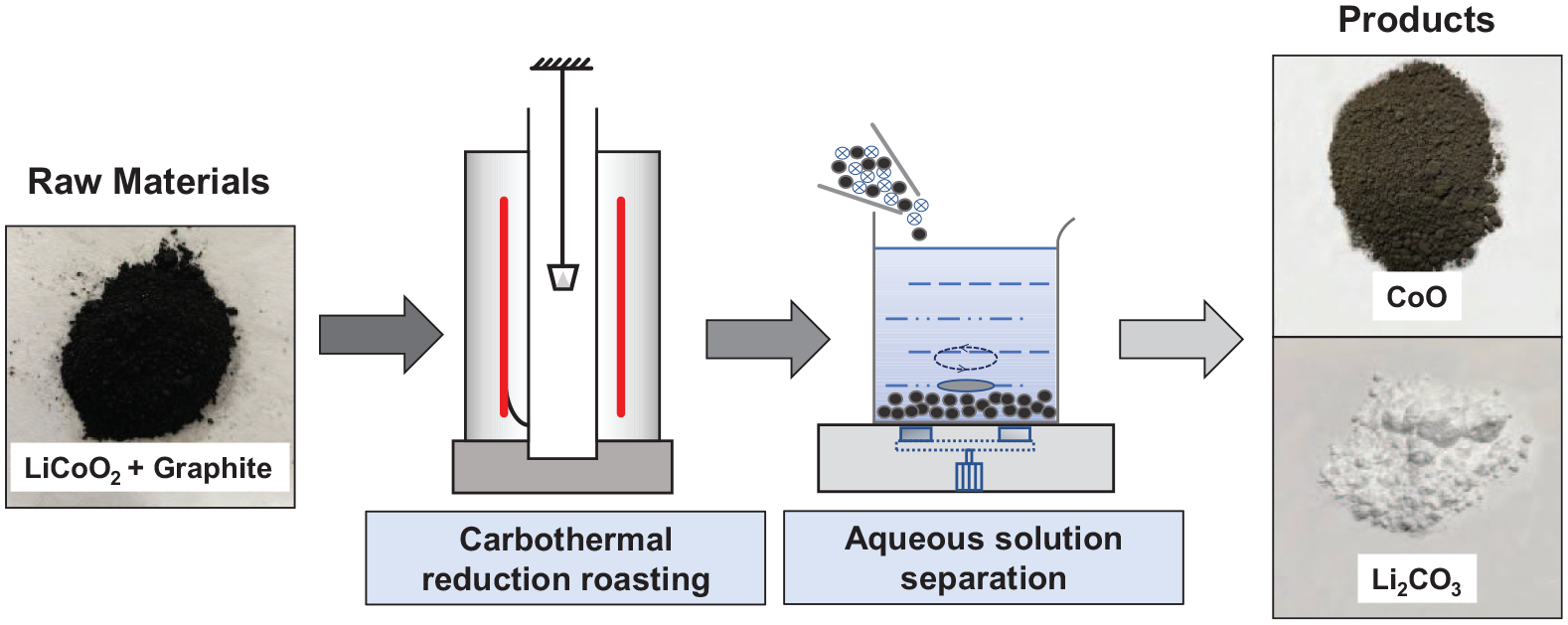
It can be seen from Figure 2(a) that the ΔG values in equations (4), (6), and (7) decrease with increasing temperature, which means that as the temperature increases, the reaction is more likely to occur. Also, the △G values in equations (4), (5), (7), and (8) are less than zero in the range of 273–1273 K, so the reaction can proceed spontaneously in a high temperature environment. As shown in equations (4) and (8), CoO will be reduced by CO to Co and CO2, and CO2 will react with lithium oxide (Li2O) to form Li2CO3 at high temperatures. Based on the above conclusions, it can be further judged that the carbothermal reduction reaction of LiCoO2 under air in a high temperature environment can occur, and the reaction products are mainly Co, Li2CO3, and CO2. According to the reactants and products, the chemical reaction equation of LiCoO2 and carbon can be inferred as equation (9).
(a) The relationship between the Gibbs free energy (△G) and temperature (T) under normal pressure. (b) Reduction curve of CoO and CO2 by CO. (c) The relationship between the logarithm of the CO equilibrium percentage of reactions Ⅰ, Ⅱ, Ⅲ, and temperature.
Figure 2(b) and (c) shows the equilibrium curves of cobalt oxide and CO2 reduction by CO and the relationship between the logarithm of the equilibrium percentage of CO and temperature. From a thermodynamic point of view, the oxide of cobalt is reduced step-by-step and will not be directly reduced to Co. Therefore, the reaction product composition can be controlled by controlling the reaction conditions (temperature, reactant ratio). As shown in Figure 2(b), the CO content curve of CO2 reduction by graphite is higher than that of reactions II and III above 773 K. According to thermodynamic analysis, CoO will be reduced to Co by CO in an air atmosphere. However, in addition to thermodynamic (temperature) considerations, it is possible to control the amount of the reducing agent so that the reduction product exists in the form of CoO to achieve the purpose of controlling the composition of the reaction product by controlling the reaction conditions.
Thermogravimetric analysis results
The thermogravimetric analysis/differential scanning calorimetry (TGA/DSC) results of the three sample materials under air atmosphere are shown in Figure 3(a) and (b). When the temperature range was 0–100 °C, sample 1 demonstrated no mass loss and the heat flow was stable; when the temperature reached 1140 °C, sample 1 began to show a slight mass loss and a weak exothermic peak; when the temperature reached 1200 °C, the final mass loss of sample 1 was 2.2 wt%. Sample 1, after being heated to 1200 °C, was cooled, removed from the furnace, and subjected to X-ray diffraction (XRD) analysis. The results obtained are shown in Figure 3(c). Three substances were detected in the product: Li2CO3, Co, and non-decomposable LiCoO2. It can be seen that pure LiCoO2 has good thermal stability. When the temperature reached 1140 °C, the decomposition of equation (3) begins in air. The temperature conditions required to recover cobalt by calcining pure LiCoO2 in air are relatively high.

(a) Thermogravimetric analysis and (b) heat flow analysis of three sample materials (#1, #2, and #3). (c) X-ray diffraction spectrum of the decomposition products of sample 1 at 1200 °C. (d) X-ray diffraction spectra of the reaction products of samples 2 and 3 at 800 °C.
The main mass loss in samples 2 and 3 is concentrated in the temperature range of 550–800 °C. The obvious endothermic peaks of samples 2 and 3 indicate that an endothermic reaction has occurred at this stage. According to thermodynamic calculations, in this temperature range, it can be seen that reaction 6 is an endothermic process. In order to determine the specific chemical reaction that occurs within this temperature range, samples 2 and 3 that had been heated to 800 °C were cooled and then subjected to XRD analysis. The results are shown in Figure 3(d). It can be seen that the products detected in sample 2 at 800 °C were LiCoO2, Li2CO3, CoO, and Co. The substances detected in sample 3 at 800 °C were C, Li2CO3, and CoO; XRD results show that in the temperature range of 550–800 °C, graphite reacts with carbon dioxide to generate carbon monoxide, and then carbon monoxide reduces LiCoO2. The reduction process is divided into two steps: the first reduction products are Li2O and CoO, and Li2O reacts with the CO2 generated by the reduction to form Li2CO3. In the second step, the reduction product CoO continues to be reduced by CO to form Co and CO2, which further promotes the production of Li2CO3. When the temperature is increased to 800 °C, the mass loss of samples 2 and 3 are 42.6 wt% and 8.9 wt%, which is greater than the initial mass fraction of graphite in the sample (Table 1), which proves that graphite and LiCoO2 undergo a reduction reaction during the heating process, and the generated gas escapes the reaction system. When the temperature exceeds 800 °C, the mass loss and heat flow trends of the three samples are basically the same, indicating that the graphite is consumed in the subsequent heating process, and the self-decomposition reaction of LiCoO2 can only be realized when the temperature condition (i.e. 1140 °C) is reached.
Sample material content.

The results obtained verify the conclusion of the thermodynamic analysis, and that the method of carbothermal reduction and recovery of LiCoO2 is feasible. Under an air atmosphere, changing the ratio of graphite and LiCoO2 will affect the composition of the reaction product. Therefore, follow-up studies can be conducted by controlling the ratio of graphite and LiCoO2 to obtain the ideal reaction conditions for the product composition.
Comparative analysis of the carbothermic reduction results under air conditions
Based on the TG/DSC analysis, the temperature conditions of the carbothermal reduction reaction of LiCoO2 were determined to be 700 °C, 800 °C, 900 °C, and 1000 °C, respectively. Sample 2 (the ratio of C and LiCoO2 is 18:4) was kept under an air atmosphere for 45 min, and after cooling to room temperature, the composition of the reaction product was analyzed. The XRD analysis of the obtained product is shown in Figure 4(a). The product obtained at 700 °C contains C, LiCoO2, CoO, and Li2CO3, which indicates that graphite starts to reduce LiCoO2 to CoO under these conditions, but the reaction cannot proceed fully due to the influence of kinetics, and residual LiCoO2 is present among the products. When the temperature is 800 °C, the reaction products are C, Co, and CoO. Under this temperature regime, no obvious LiCoO2 peak was evident in the XRD pattern of the product. The peak height of CoO increased significantly and a Co peak appeared, indicating that LiCoO2 was completely reduced. When the reaction temperature was 900 °C, the Co peak height in the XRD of the product is significantly increased and the CoO peak height is significantly reduced, which indicates that graphite can further reduce CoO to Co at this temperature. The peak height of Co increases significantly at 1000℃ and the peak due to CoO disappears. The remaining products are found to be C, Li2CO3, and Co.

(a) X-ray diffraction detection spectra of the pyrolysis reduction products of sample 2 at different temperatures (700 °C, 800 °C, 900 °C, 1000 °C). (b) X-ray diffraction spectra of the pyrolysis reduction products of sample 3 at different temperatures (700 °C, 800 °C, 900 °C, 1000 °C). (c) X-ray diffraction spectra of different C/LiCoO2 molar ratios (3:4, 5:4) at 800 °C.
CoO is a raw material for LiCoO2 regeneration, and Co is an undesirable product. In addition to thermodynamic considerations, the products of carbothermal reduction are also related to the reaction kinetics, and the reaction kinetics are related to the reduction temperature and the concentration of the reactants. Therefore, the ratio of the reactants is adjusted, and the products obtained by the reaction of sample 3 at different temperatures were subjected to XRD analysis using the same experimental method. The results are shown in Figure 4(b). The product obtained at 700 °C contains LiCoO2, CoO, and Li2CO3, which indicates that graphite starts to reduce some of the LiCoO2 to CoO under these conditions. The products obtained at 800 °C are the same as those obtained at 700 °C. Under these temperature conditions, the peak height of LiCoO2 decreased significantly, while that of CoO increased appreciably, indicating that the reaction proceeded more completely. When the reaction temperature was 900 °C, the residual LiCoO2 content decreased and Co appears in the product, which indicates that graphite can further reduce CoO to Co at this temperature. The peak height of Co increases significantly at 1000 °C, and the final remaining products are LiCoO2, CoO, Li2CO3, and Co. Based on the above results, it can be proved that the reduction of LiCoO2 by C is a stepwise reduction reaction process, so the ability to control the reaction products can be achieved by controlling the reaction conditions. When the temperature was 800 °C and the ratio of C and LiCoO2 was 3:4, LiCoO2 can be reduced to CoO and Li2CO3.
However, there is still unreduced LiCoO2 in the product, which may be because some of the graphite is oxidized by O2 in the air to escape as a gas. The optimum reduction conditions can be found by increasing the proportion of graphite in the reactants. As shown in Figure 4(c), by changing the molar ratio of the reactants to 5:4 at 800 °C, LiCoO2 is fully reduced, and the product only contains Li2CO3 and CoO, which meets the compositional requirements.
Study on the crystal structure changes during the reduction process and SEM analysis
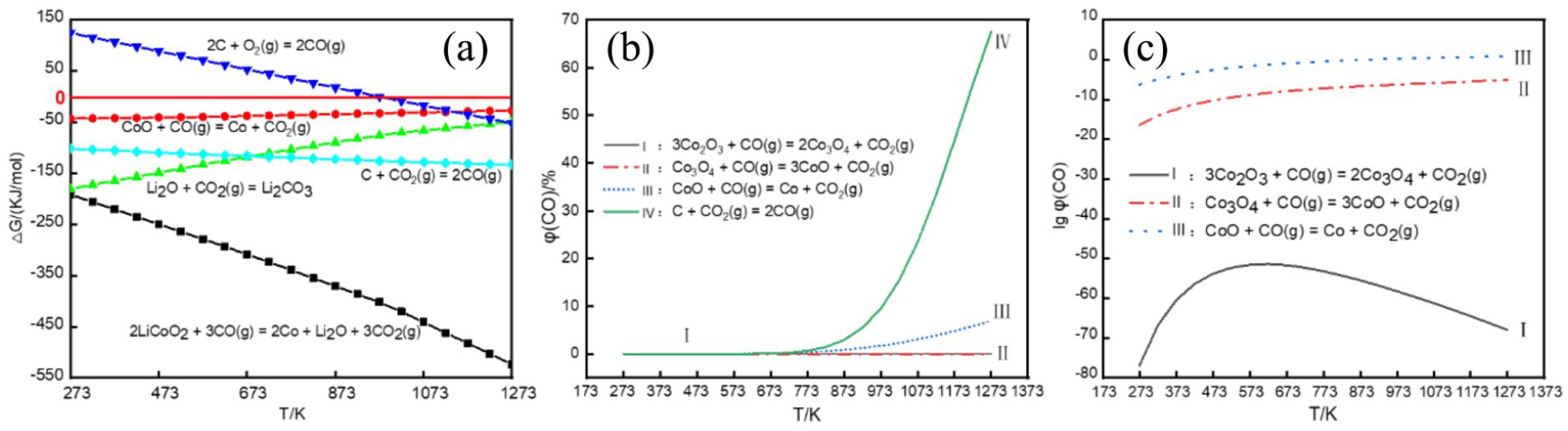
As shown in Figure 5, scanning electron microscopy (SEM) analysis is used to analyze the initial morphology of sample 4 and the morphological changes in the reaction products at different temperatures, while energy-dispersive X-ray spectroscopy (EDS) analysis is used to infer the approximate composition of the scanned product. The initial morphology of the sample is shown in Figure 5(a). In the initial state, the sample has large particles and irregular shapes. It can be seen from the figure that as the reaction temperature gradually increases, the product particles gradually become smaller. Also, the cluster structure of the original organization is destroyed, and the morphology of the product is irregular spherical and stacked sequins. 33 Irregular clustered areas accumulate so that the original topographic structure of the product disappears. Figure 5(f) and (g) shows the EDS scans of the sample material and the reaction product at 800 °C, respectively. According to the element ratio, it can be inferred that the reduction products are CoO and Li2CO3 at 800 °C.
SEM analysis (a) raw material; (b) 700 °C; (c) 800 °C; (d) 900 °C; and (e) 1000 °C. EDS analysis (f) raw material; (g) 800 °C.
It can be seen from Figure 6(a) that the unit cell of LiCoO2 has a hexahedral structure, in which the Co layer and the Li layer are, respectively, arranged between two adjacent O layers.34,35 Therefore, the unit cell structure of LiCoO2 can be further decomposed into a layered arrangement of oxygen octahedrons with Li or Co as the central atom. It is known that the electronegativity of lithium is 0.98, oxygen is 3.44, cobalt is 1.84, and that the difference in electronegativity between lithium and oxygen is 2.46, and that of cobalt and oxygen is 1.6. Elements with electronegativity differences of greater than 1.7 form ionic bonds, and those with electronegativity differences of less than 1.7 form covalent bonds. Although both are oxygen octahedrons, Co and O are connected by covalent bonds, and Li and O are connected by ionic bonds. As the reaction temperature increases, the ionic bond between Li and O is more likely to break, and the octahedral structure of Li and O will be destroyed.

(a) The crystal structure and basic unit structure of LiCoO2. (b) The crystal structure change process during reduction of LiCoO2 by CO.
Figure 6(b) shows a possible route for the reduction of LiCoO2 by CO to Li2O and CoO. As shown in the figure, the Co-O bond energy in the octahedron of Co and O is higher, so CO preferentially removes the O atom in the octahedron of Li and O. Under high-temperature conditions, the +2 valence C in CO has a stronger attraction for O, causing the Li-O bond in the octahedron of Li and O to break. Each octahedron of Li and O breaks two Li-O bonds and loses two oxygen atoms. The octahedron also changes into an oxygen tetrahedron with Li as the center. The tetrahedron is a polyhedral unit in the unit cell structure of Li2O, CO molecules interact with O and turn into CO2, and then CO2 reacts with Li2O to afford Li2CO3. At a temperature of 800 °C, the octahedral structure of Li and O is stable, which is a polyhedral unit in the unit cell structure of CoO. When the temperature continues to rise, CoO continues to be reduced by CO to Co and CO2.
Separation and recovery of CoO and Li2CO3
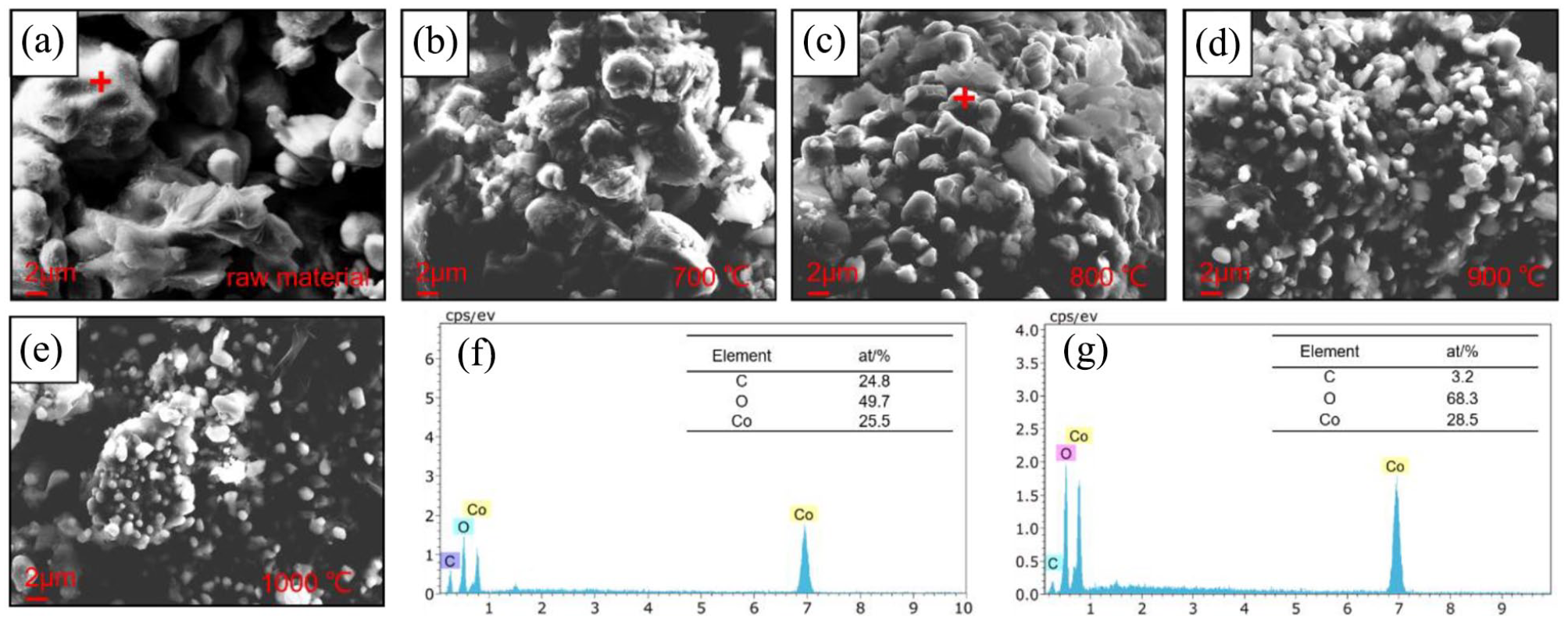
As shown in Figure 7, at a temperature of 800 °C, a molar ratio of C to LiCoO2 of 5:4, and a reduction holding time of 45 min, the reaction products are obtained and then separated and recovered from the aqueous phase. The solution separates thus: lithium carbonate is soluble in water, while cobalt oxide is insoluble in water. Lithium carbonate is separated from the mixture by selective dissolution and drying. The XRD analysis results are shown in Figure 8(a). The water-insoluble solid product cobalt oxide is separated by filtration, and the XRD is shown in Figure 8(b). Figure 8(c) shows the effect of different solid–liquid ratios on the lithium recovery rate. It can be seen that when the solid–liquid ratio is less than 30 g/L, a relatively high recovery rate can be obtained, so this solid–liquid ratio (30 g/L) is selected for recovery. Through X-ray fluorescence spectrometry (XRF) detection of the recovered products, it can be calculated that the recovery rate of lithium carbonate is 84%, and the recovery rate of cobalt oxide is 89%.

The carbothermal reduction and recovery process of LiCoO2.
(a) X-ray diffraction of recovered Li2CO3. (b) X-ray diffraction of recovered CoO. (c) Relation curve of Li recovery rate and the solid–liquid ratio.
Conclusion
Based on thermodynamic analysis, we have studied the carbothermal reduction reaction of LiCoO2 (the active electrode material of spent lithium batteries) and the crystal structure change process during the reaction. Two conclusions can be drawn:
(1) The equations for the reduction process of LiCoO2 and the thermodynamic reduction curve of cobalt oxide have been obtained through thermodynamic analysis, which provides a theoretical basis for the recovery of lithium-ion batteries and cobalt elements.
(2) The reduction of LiCoO2 by graphite is a stepwise reaction, so it can be regulated by controlling the temperature at 800 °C, the ratio of reactants (C/LiCoO2 = 5:4), and a holding time of 45 min.
(3) From the perspective of structural changes during the reaction, CO can absorb O in Li-O octahedrons and fold the octahedrons into tetrahedrons. Li-O tetrahedrons are combined into Li2O crystals and the dissociated Co-O. The octahedrons are combined into CoO crystals, and LiCoO2 is pyrolyzed and reduced through this structural change process of dissociation and collapse.
Carbothermic reduction and recycling of lithium batteries is a self-sufficient process that does not use any external chemicals from the perspective of waste management. This research helps to better understand the effect of carbon heat treatment on the complex chemical system in the actual disposal of waste lithium batteries from a microscopic point of view. It can be used to optimize industrial processing technology, improve the recycling efficiency of lithium batteries, reduce processing energy consumption, and is beneficial to industrial applications.
Experimental materials and methods
TGA/DSC analyses
The LiCoO2 and graphite used in the experiment were obtained from Macleans Biochemical Technology Co., Ltd. Four different molar ratios of LiCoO2 and graphite were measured, as shown in Table 1. (The choice of three samples was as follows: sample 1 is pure LiCoO2; the molar ratio of graphite to LiCoO2 in sample 2 is 18:4, which is close to the ratio of positive and negative active materials in actual lithium batteries; the molar ratio of graphite to LiCoO2 is 3:4 in sample 3, which is the ratio of LiCoO2 that completely reacts with graphite in an ideal state; the molar ratio of graphite to LiCoO2 in sample 4 is 5:4, which is the best reduction ratio of CoO obtained in the experiment.) For the experiments on the first three sets of samples, the reactants were mixed for 2 h using a ball mill. TGA (SDT-Q600) was used to evaluate the thermal response of the three samples under an air atmosphere and the samples were heated from 20 °C to 1200 °C at a temperature increase rate of 10 °C/min. According to the TG test results, the samples were heated from room temperature to 800 °C or 1200 °C, and the products obtained after cooling were subjected to XRD to obtain the composition of the reducing substance.
Carbothermic reduction experiment
The carbothermic reduction experiment was carried out under an air atmosphere in a tube furnace (STG-16) (as shown in Figure 9(a)); the furnace is a vertical single temperature zone tubular electric furnace with a maximum working temperature of 1600 °C. A corundum tube with an inner diameter of 120 mm is inserted into the furnace and the air atmosphere in the corundum tube is maintained for the carbon thermal reduction experiments. The working temperature of the experiment is 973–1273 K and the holding time is 45 min. Mixtures of LiCoO2 and C in different proportions were thoroughly mixed in the ball mill, then 1 g of the mixture was put into the corundum crucible and placed in the stable area of the tube furnace under different temperature conditions to accurately record the reduction temperature. After pyrolysis and reduction, the reaction product was characterized using an X-ray diffractometer (XRD, STOE/2, Stofdarmstadt Co.) and a scanning electron microscope (SEM, SU-1510, Hitachi Co.).
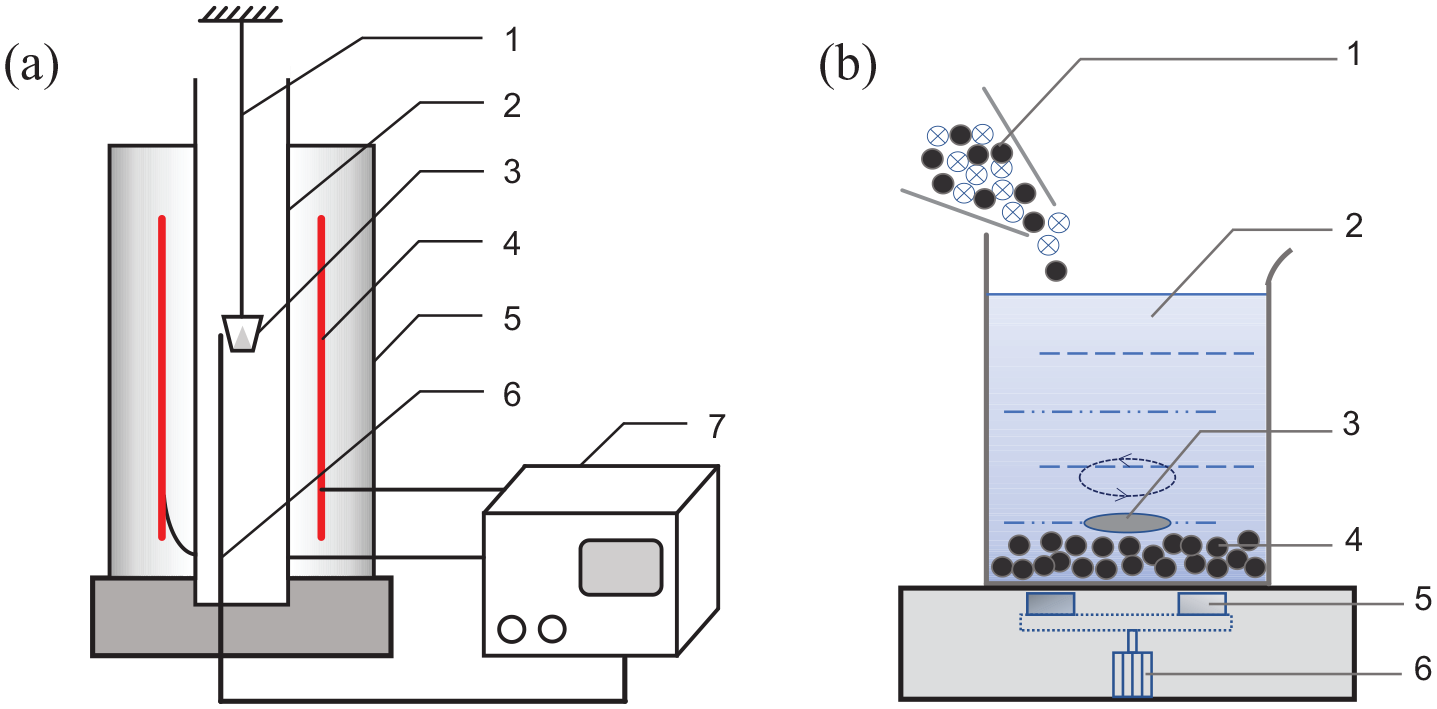
(a) Schematic diagram of the vertical tubular electric furnace: (1) iron wire, (2) corundum tube, (3) corundum crucible, (4) heating element, (5) furnace, (6) thermocouple, (7) temperature controller. (b) Schematic diagram of the magnetic stirrer: (1) mixture, (2) solution, (3) magnetic rotor, (4) precipitate, (5) magnet, (6) motor.
Separation of the products by aqueous solution separation
After the mixed powder had been pyrolyzed and reduced, it was immersed in deionized water at room temperature, and the product was separated from the pyrolyzed mixture using a horizontal magnetic stirrer (MS5, Joanlab Co.) (as shown in Figure 9(b)). The solution temperature was 25 °C, the solid–liquid ratio was 20–50 g/L, the stirring speed was 1500 r/min, and the stirring time was 24 h in the separation process. Soluble substances dissolve in the solution, while insoluble substances precipitate at the bottom of the beaker during continuous magnetic stirring. The resulting solution was further filtered to give a clear lithium carbonate solution, which was slowly evaporated in a drying oven at 150 °C to give an Li2CO3 as white crystalline powder. The insoluble precipitates were collected and washed to give a CoO as black powder. XRD and XRF (EDX8000) were used to characterize and quantitatively analyze the recovered materials. The recovery rate (Ra) of the product is calculated as shown in equation (10)

In the formula,
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (No. 51874029).