Abstract
A copper-catalyzed multicomponent cascade cyclization using readily available substrates, including 2-formylbenzonitriles, phenylacetylenes, and diaryliodonium salts, is achieved. A broad reaction scope is presented with good functional group compatibility, giving rise to a range of 3-(2-oxopropyl)-2-arylisoindolinones in moderate to good yields.
Keywords
Introduction
Isoindolin-1-ones, including 3-(2-oxopropyl)-2-substituted isoindolinones that can be derived from isoindolinone derivatives, are a class of nitrogen-containing heterocyclic compounds that are usually employed as key structural scaffolds for organic synthesis and are very common structural motifs in natural products and pharmaceuticals exhibiting important biological activities (Scheme 1).1–6 For example, pagoclone7–9 and pazinaclone10,11 are clinically utilized anxiolytic and sedative drugs, respectively. With the expanding role of 3-(2-oxopropyl)-2-substituted isoindolinones in medicinal chemistry, numerous methods have been developed for their synthesis over the past few decades.12–27 A common method involves C–H functionalization, which employs N-substituted benzamides as reactants bearing either activating or directing groups.28–33 Liu and Lu’s group developed an efficient rhodium-catalyzed coupling of α-allenols and N-methoxybenzamide derivatives to afford 3,3-disubstituted isoindolinones. 34 Liu and Li’s groups have reported the coupling of propargyl alcohol derivatives and N-ethoxy or N-methoxybenzamides by employing an Rh(III) catalyst under argon, leading to a broad variety of substituted isoindolinones.35,36 Jeganmohan has described an oxidative C–H coupling/cyclization cascade of allylic alcohols and N-alkyl/arylbenzamides to provide a broad range of 3-subtituted isoindolinones. 37 In 2019, Li’s group published a cobalt-catalyzed oxidative C–H functionalization cyclization with α-diazoketones. 38 In the methodologies mentioned above, the use of precious metal catalysts and highly functionalized starting materials limit expansion for the synthesis of compound libraries. Thus, the development of novel and efficient strategies to access 3-(2-oxopropyl)-2-substituted isoindolinone scaffolds remains highly desirable.

Selected active 3-(2-oxopropyl)-2-arylisoindolinones and synthetic strategies.
Multicomponent coupling reactions have emerged as a powerful strategy for the generation of molecular complexity in an atom-economical fashion.39–44 The groups of Singh and Cai independently utilized o-formyl methylbenzoates, trimethylsilyl enol ethers, and aniline derivatives as the starting materials to synthesize various isoindolinones with zinc(II) or scandium(III) triflate catalysts in one pot (Scheme 1).45,46 Recently, we reported a Cu-catalyzed three-component cascade cyclization using readily available substrates including 2-formylbenzonitriles, alkyl aryl ketones and diaryliodonium salts.47 In continuation of these studies, we herein disclose a three-component cascade cyclization of diaryliodonium salts with 2-formylbenzonitriles and phenylacetylenes for the construction of various 3-(2-oxopropyl)-2-substituted isoindolinones.
Results and discussion
We began our optimization studies using 2-formylbenzonitrile (
Optimization of the reaction conditions. a

Reaction conditions:
Isolated yield.
After the optimization of the reaction conditions, we proceeded to explore the robustness of the protocol with respect to the substrates. The scope of phenylacetylenes
Reaction conditions:
Isolated yield.
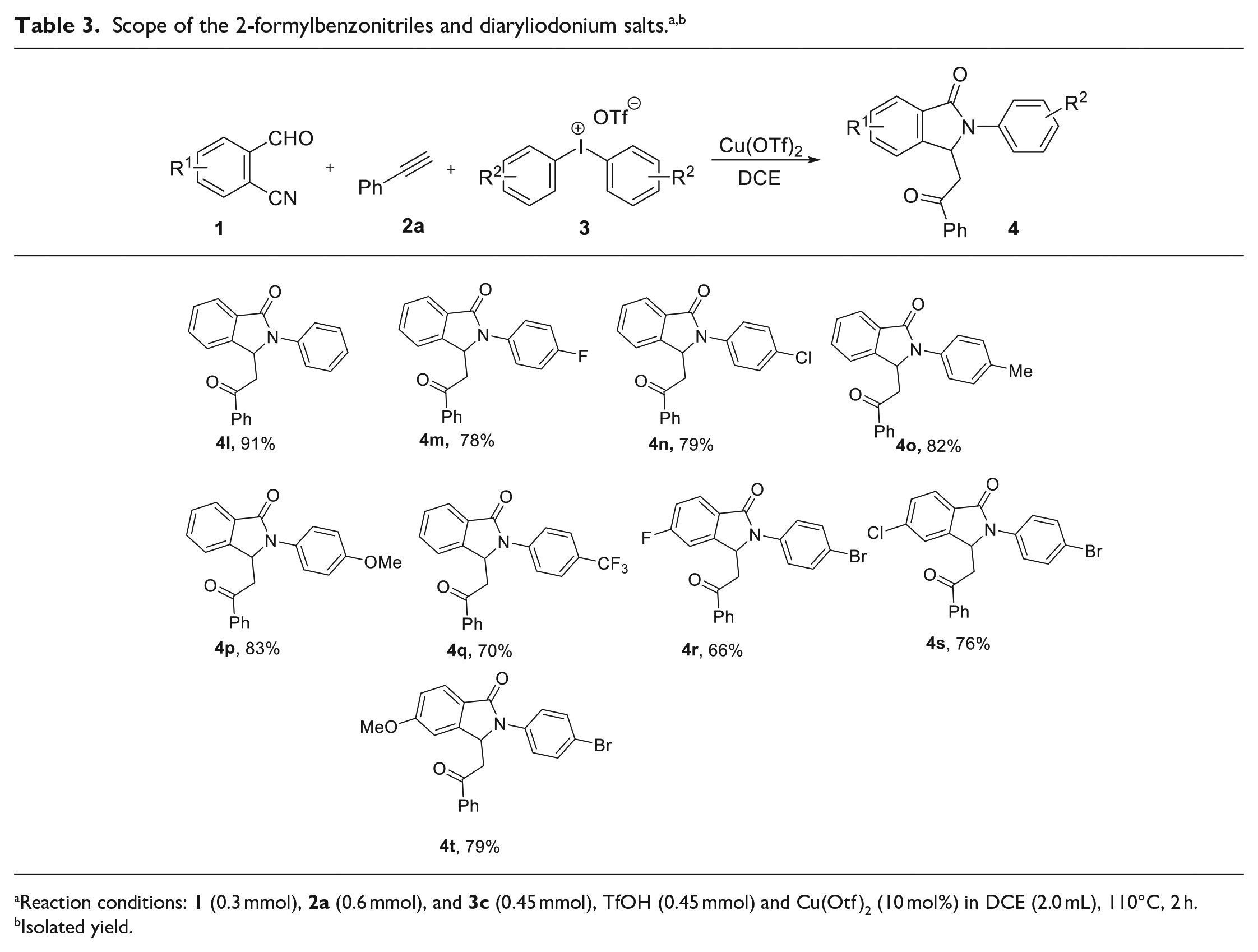
Next, the three-component cascade cyclization was carried out with different substituted 2-formylbenzonitriles
Reaction conditions:
Isolated yield.
To further understand the mechanism of this reaction, control experiments were investigated. Initially, we conducted a control reaction using phenylacetylene (
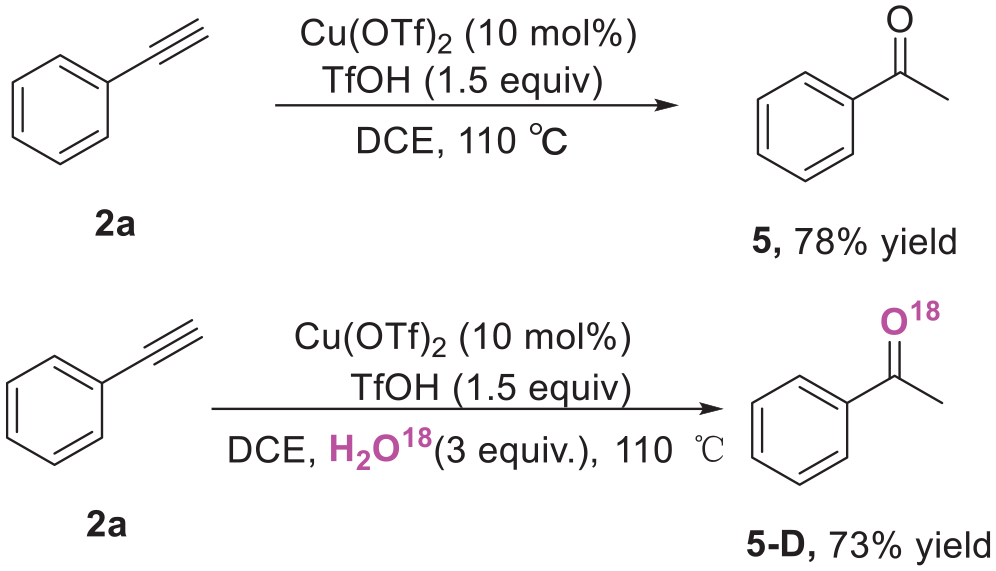
Control experiments.
On the basis of the control experiments and relevant literature, we tentatively propose a mechanism for this Cu-catalyzed transformation involving an N-arylnitrilium cation (Scheme 3). First, the reaction of 2-formylbenzonitrile (
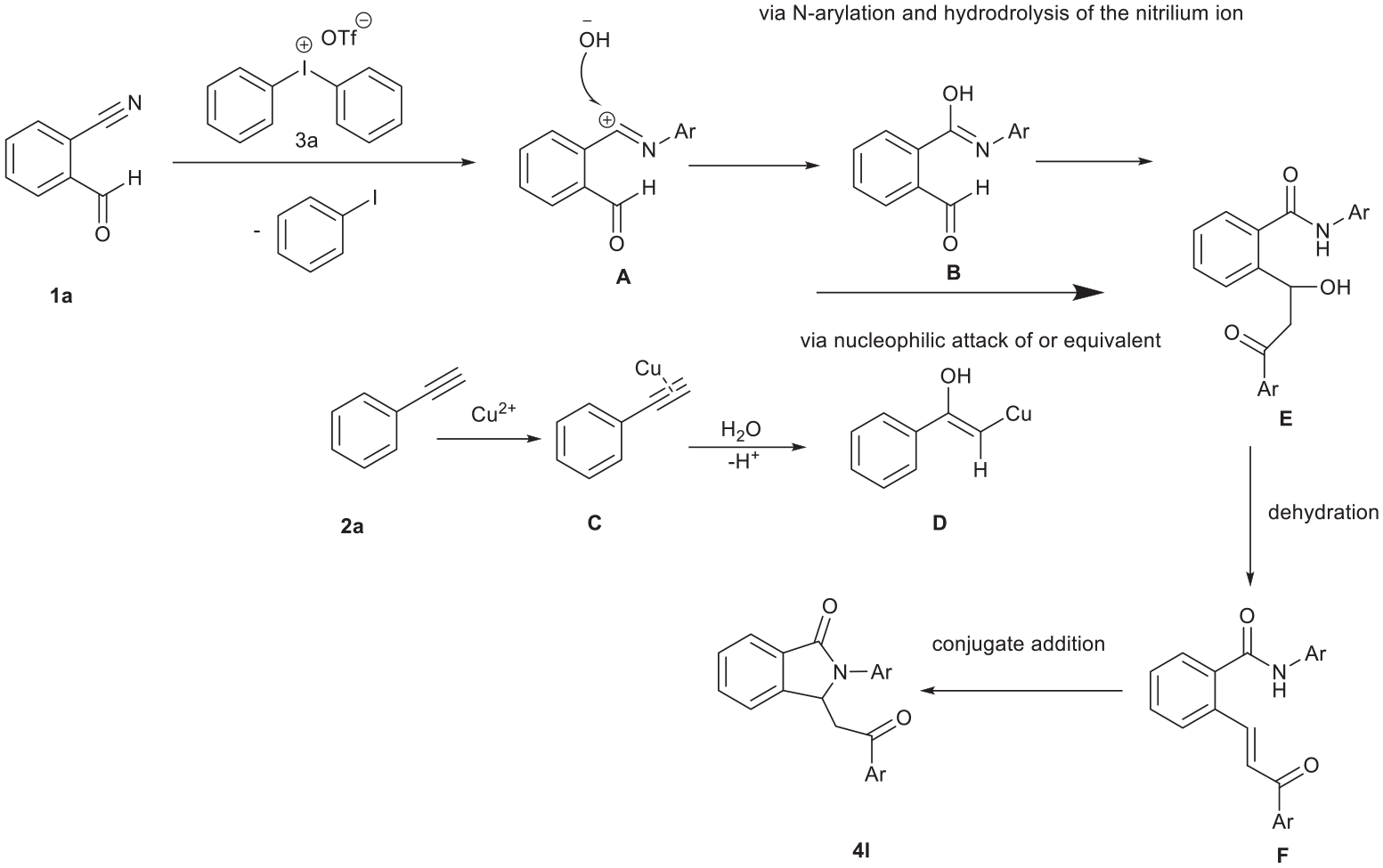
Proposed reaction mechanism.
Conclusion
In summary, we have developed a Cu-catalyzed, three-component cascade cyclization using readily available substrates including 2-formylbenzonitriles, phenylacetylenes and diaryliodonium salts. Our reaction operates under simple conditions with broad functional group compatibility, and is applicable to various phenylacetylenes and diaryliodonium salts. A wide range of 3-(2-oxopropyl)-2-arylisoindolinone derivatives is obtained in good yields under simple reaction conditions. Further applications of this methodology are being pursued in our laboratory.
Experimental section
General information
All reactions were carried out under an air atmosphere. Reagents were purchased from Aldrich, Acros, or Alfa. The diaryliodonium salts
Preparation of Compounds 4; general procedure
A solution of 2-formylbenzonitrile
All the products are known and their structures were confirmed by NMR spectral comparison with literature data.14,47,55
2-(4-Bromophenyl)-3-(2-oxo-2-phenylethyl)isoindolin-1-one (
2-(4-Bromophenyl)-3-[2-(4-fluorophenyl)-2-oxoethyl]isoin dolin-1-one (
2-(4-Bromophenyl)-3-[2-(4-chlorophenyl)-2-oxoethyl]iso indolin-1-one (
2-(4-Bromophenyl)-3-[2-(4-bromophenyl)-2-oxoethyl]iso indolin-1-one (
2-(4-Bromophenyl)-3-{2-oxo-2-[4-(trifluoromethyl)phenyl] ethyl}isoindolin-1-one (
2-(4-Bromophenyl)-3-[2-(4-nitrophenyl)-2-oxoethyl]isoin dolin-1-one (
2-(4-Bromophenyl)-3-(2-oxo-2-p-tolylethyl)isoindolin-1-one (
2-(4-Bromophenyl)-3-(2-oxo-2-o-tolylethyl)isoindolin-1-one (
2-(4-Bromophenyl)-3-[2-(naphthalen-1-yl)-2-oxoethyl] isoindolin-1-one (
2-(4-Bromophenyl)-3-(2-oxo-2-(2,3,4,5-tetrafluorophenyl) ethyl)isoindolin-1-one (
2-(4-Bromophenyl)-3-[2-oxo-2-(thiophen-2-yl)ethyl]isoindo lin-1-one (
3-(2-Oxo-2-phenylethyl)-2-phenylisoindolin-1-one (
2-(4-Fluorophenyl)-3-(2-oxo-2-phenylethyl)isoindolin-1-one (
2-(4-Chlorophenyl)-3-(2-oxo-2-phenylethyl)isoindolin-1-one (
3-(2-Oxo-2-phenylethyl)-2-p-tolylisoindolin-1-one (
2-(4-Methoxyphenyl)-3-(2-oxo-2 phenylethyl)isoindolin-1-one (
3-(2-Oxo-2-phenylethyl)-2-(4-trifluoromethylphenyl)iso indolin-1-one (
2-(4-Bromophenyl)-5-fluoro-3-(2-oxo-2-phenylethyl)iso indolin-1-one (
2-(4-Bromophenyl)-5-chloro-3-(2-oxo-2-phenylethyl)iso indolin-1-one (
2-(4-Bromophenyl)-5-methoxy-3-(2-oxo-2-phnylethyl)iso indolin-1-one (
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: They are grateful to the Science and Technology Foundation of Jiangsu Food and Pharmaceutical Science College (Nos. JSFP2019008 and JSFP2019002), and the Natural Science Foundation of the Jiangsu Higher Education Institutions of China (No. 20KJB360014).