Abstract
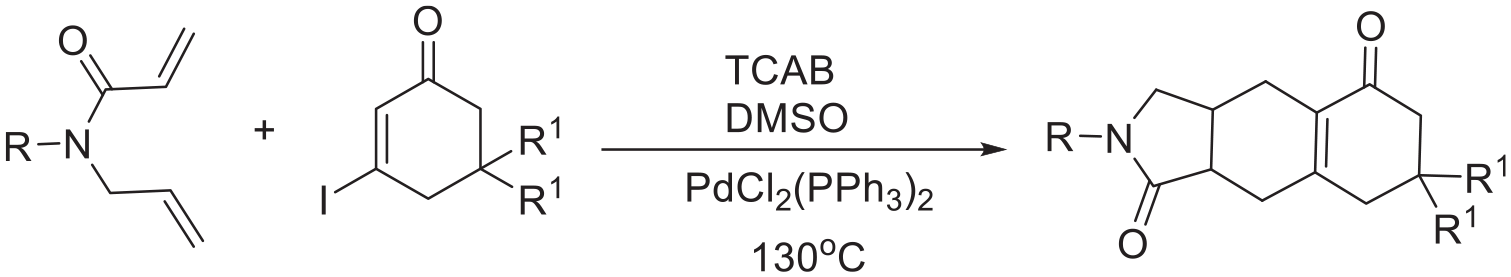
A synthetic method for the preparation of 3,3a,4,6,7,8,9,9a-octahydro-1H-benzo[f]isoindole-1,5(2H)-diones from 1,6-dienes and vinyl iodides is developed using PdCl2(PPh3)2 as the catalyst. The presented approach exhibits a good functional group tolerance and affords moderate yields of the products. A mechanism is also proposed.
Introduction
Due to the benefits of atom economy, saving time, and less waste generation, cascade reactions that allow the generation of molecular complexity in a single operation have attracted significant attention.1–5 Although cascade reactions have been successfully employed for the synthesis of the core skeleton of many important natural products, the design and performance of cascade reactions remains a challenging aspect of organic chemistry.6–10 Recently, the α-arylation of enones and their derivatives has been developed.11–14 For example, Liu reported an efficient synthesis of N-fused polycyclic indoles via a palladium-catalyzed annulation/acyl migration cascade reaction (Scheme 1a). 11 McGlacken reported that 1,3-diketone-derived enol ethers could undergo Heck cyclization, offering tricyclic oxoisochromene derivatives in good yields (Scheme 1b). 12 In these cases, the introduction of a halogen atom into certain molecules was necessary.15,16 Meanwhile, reports on alkylations at the α-position of enones and their derivatives are comparatively scarce.17,18 Starting from electron-deficient vinyl iodides and propargyl ethers, Huang provided an efficient synthesis of structurally complex polycycles with 2,3-dihydrofuran units (Scheme 1c). 18 Given this background, we envisioned that Pd-catalyzed reactions of 1,6-dienes and vinyl iodides would proceed via a cascade reaction to form polycyclic compounds through alkylation of the α-position of the enone (Scheme 1d).
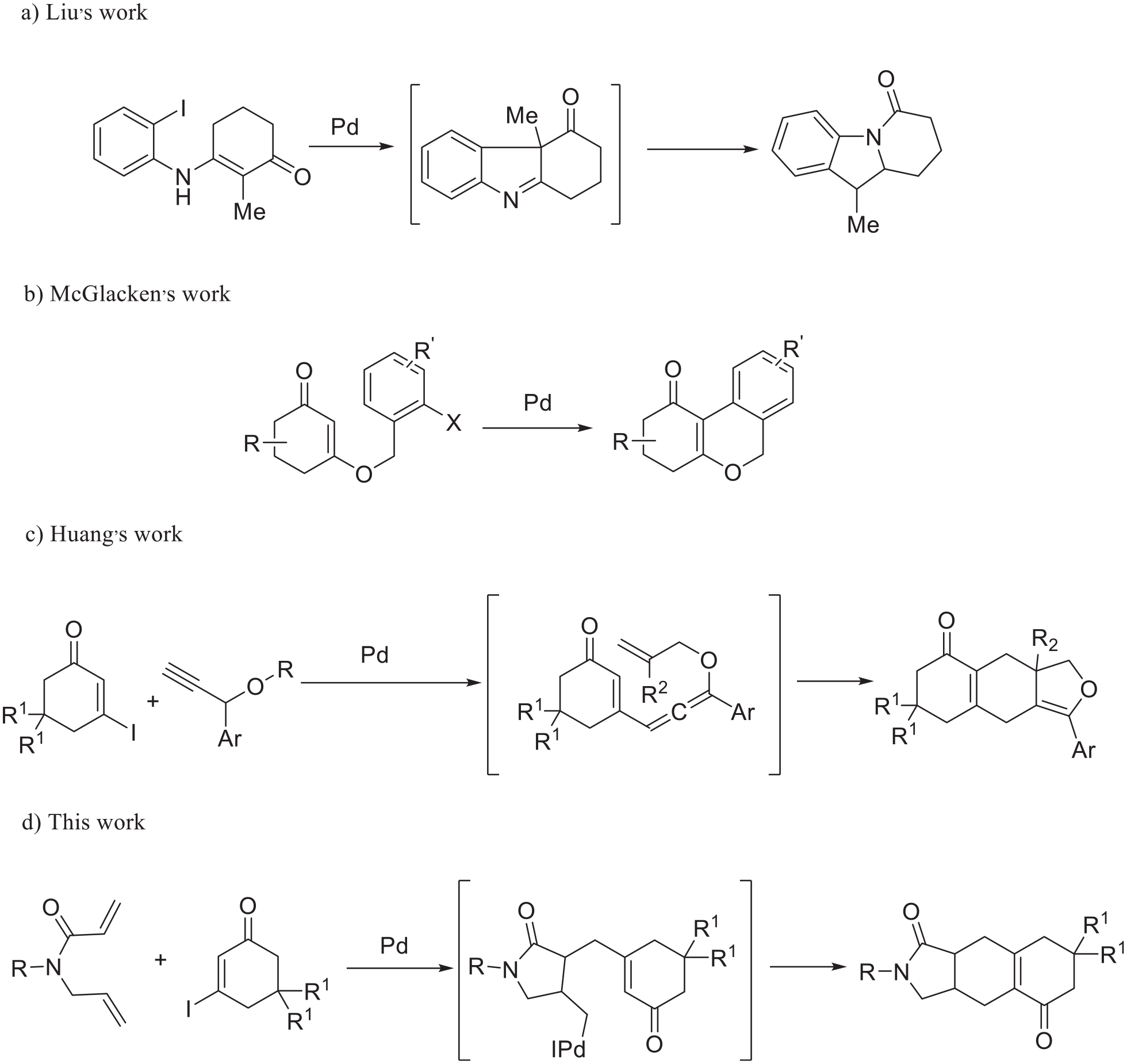
Previous work and our synthetic strategy: (a) Liu’s work, (b) McGlacken’s work, (c) Huang’s work, and (d) this work.
Results and discussion
Our initial study was carried out with the reaction between N-allyl-N-benzylacrylamide (
Optimization of the reaction conditions. a

Reaction conditions:
Yield of isolated product.
With optimized reaction conditions in hand, we next explored the scope and generality of the method. Pleasingly, N-allyl-N-benzylacrylamides substituted with an electron-donating group (Me) or an electron-withdrawing group (Cl) on the aromatic ring were tolerated, leading to the formation of target products
Substrate scope of the reaction. a

Reaction conditions:
Yield of isolated product.

Molecular structure of
Crystal data and structure refinement for 3K. 21

A plausible reaction mechanism has been proposed (Scheme 2). Initial oxidative addition and carbo-palladation gives Intermediate
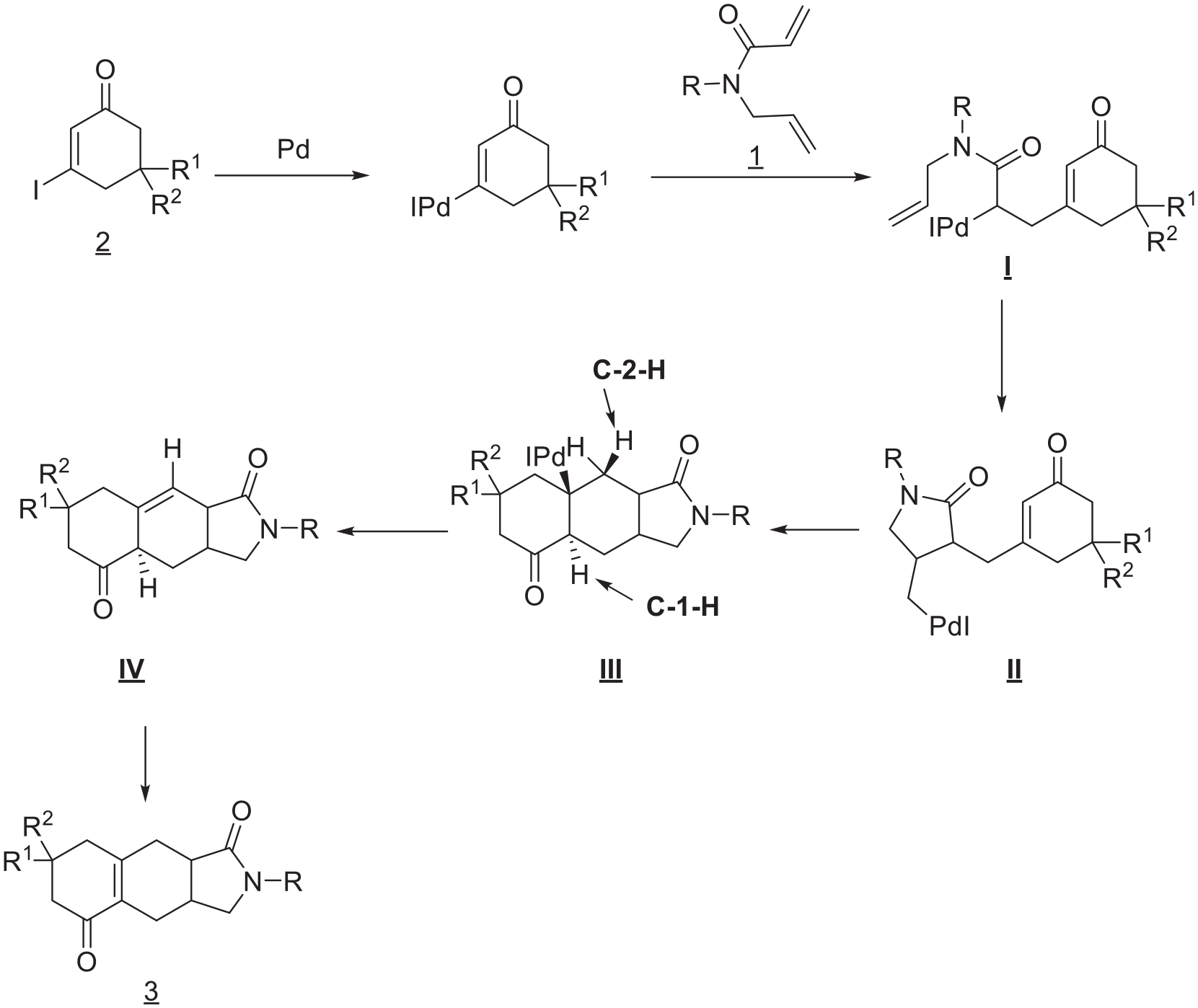
A plausible reaction mechanism.
Conclusion
In summary, we have developed an efficient method for the synthesis of polycyclic oxindole moieties in the presence of a PdCl2(PPh3)2 catalytic system under mild conditions. The ready accessibility of the starting materials, the wide range of compatibility of the substrates, and the generality of this make the reported process advantageous.
Experimental
All starting materials and reagents are commercially available and were used without further purification. The organic solvents used in this study were dried over appropriate drying agents and distilled prior to use. Melting points were determined with an X-4 apparatus and are uncorrected. IR spectra were recorded with a Shimadzu FTIR-8300 spectrophotometer. High-resolution mass spectra (HRMS) were obtained on a waters G2-Xs with an ESI source (Waters, Manchester, UK). The 1H NMR spectra were determined in CDCl3 using TMS as an internal reference with a Bruker 500 FT NMR spectrometer operating at 500 MHz. The 13C NMR spectra were determined in CDCl3 using TMS as an internal reference with a Bruker 500 FT NMR spectrometer operating at 125 MHz.
Polycyclic Oxindoles 3 general procedure
The 1,6-diene
Yield: 35.6 mg (35%); white solid; m.p. 192 °C–198 °C (PE/EtOAc); IR (film) 2962, 2936, 1700, 1668, 1635, 1426, 1389, 810 cm−1; 1H NMR (500 MHz, CDCl3): δ 7.51 (d, J = 9.0 Hz, 2H), 6.92 (d, J = 9.0 Hz, 2H), 3.90–3.87 (m, 1H), 3.82 (s, 3H), 3.64 (t, J = 9.5 Hz, 1H), 2.91–2.88 (m, 1H), 2.64–2.60 (m, 1H), 2.42–2.23 (m, 6H), 2.12–2.09 (m, 2H), 1.09 (s, 3H), 1.03 (s, 3H); 13C NMR (125 MHz, CDCl3): δ 198.8, 173.4, 156.4, 153.9, 132.8, 130.7, 121.3, 114.0, 55.5, 53.3, 51.4, 45.7, 44.9, 36.7, 33.1, 32.3, 29.4, 27.0, 26.2; HRMS (ESI): m/z [M+H]+ calcd for C21H26NO3: 340.1913; found: 340.1912.
Supplemental Material
sj-cif-1-chl-10.1177_17475198211058653 – Supplemental material for A palladium-catalyzed cascade approach for the synthesis of 3,3a,4,6,7,8,9,9a-octahydro-1H-benzo[f]isoindole-1,5(2H)-diones
Supplemental material, sj-cif-1-chl-10.1177_17475198211058653 for A palladium-catalyzed cascade approach for the synthesis of 3,3a,4,6,7,8,9,9a-octahydro-1H-benzo[f]isoindole-1,5(2H)-diones by Dong Cheng, Xiangzhen Meng, Dan Wang, Xuan Zhao, Yusheng Wang and Zhiyu Ji in Journal of Chemical Research
Supplemental Material
sj-docx-1-chl-10.1177_17475198211058653 – Supplemental material for A palladium-catalyzed cascade approach for the synthesis of 3,3a,4,6,7,8,9,9a-octahydro-1H-benzo[f]isoindole-1,5(2H)-diones
Supplemental material, sj-docx-1-chl-10.1177_17475198211058653 for A palladium-catalyzed cascade approach for the synthesis of 3,3a,4,6,7,8,9,9a-octahydro-1H-benzo[f]isoindole-1,5(2H)-diones by Dong Cheng, Xiangzhen Meng, Dan Wang, Xuan Zhao, Yusheng Wang and Zhiyu Ji in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We acknowledge the Key Natural Science Foundation of Anhui Higher Education Institution (KJ2017A446), the Excellent Young Talents Support Program of Anhui Higher Education Institutions (gxyq2018075), and the Innovation and Entrepreneurship Project of College Students in Anhui Province (S202010380031).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


