Abstract
3-Chloro-4-indolylmaleimides and two different alkynes are used as the starting materials in a novel and highly effective Pd-catalyzed addition/C–H activation/cyclization sequence for the synthesis of pyrrolo[3,4-c]carbazole-1,3(2H,6H)-diones. The desired products are obtained in moderate to excellent yields. Such compounds show a wide range of biological activities.

Introduction
Pyrrolo[3,4-c]carbazole-1,3(2H,6H)-diones have attracted significant attention in pharmaceutical chemistry because of their wide range of biological activities, for example, inhibition of PARP-1, 1 Chk 1,2,3 and D1-CDK4, 4 JNK, 5 GSK-3.6,7 Relatively, few synthetic methods for the synthesis of pyrrolo[3,4-c]carbazole-1,3(2H,6H)-diones have been reported. The main previously reported methods are as follows: (1) A 2-vinylindole derivative is synthesized via a Wittig reaction between an indolaldehyde and phosphorus ylides. The 2-vinylindole derivative then undergoes a Diels–Alder reaction with maleimide, and the product is oxidized by MnO2 or DDQ (2,3-Dichloro-5,6-dicyano-1,4-benzoquinone) to form the target compound (Scheme 1(a)). 2 (2) 3-Bromo-4-indolylmaleimide is reacted with o-bromophenylboronic acid via a Suzuki reaction to obtain a diarylmaleimide derivative, which is then cyclized via a Heck reaction to form the product (Scheme 1(b)). 8 (3) Condensation of indole-3-acetamide with methyl 2-(1H-indol-3-yl)-2-oxoacetate in the presence of potassium t-butoxide gives a diarylmaleimide, which is then converted into the target product using DDQ, I2, 9 Pd(OAc)2, 10 or PdCl2 (Scheme 1(c)). 11 In the current study, a novel highly effective Pd-catalyzed addition/C–H activation/cyclization of 3-chloro-4-indolylmaleimides and alkynes is used to synthesize pyrrolo[3,4-c]carbazole-1,3(2H,6H)-diones. Such compounds have a wide spectrum of biological activities. The procedure is high-yielding and also easy to operate (Scheme 1(d)).

Synthesis of pyrrolo[3,4-c]carbazole-1,3(2H,6H)-dione: (a)–(c) Current synthetic strategies and (d) this work.
Results and discussion
Synthesis
Initially, 3-chloro-4-indolylmaleimide and diphenylacetylene were used as model substrates to investigate the effects of factors such as the base, ligand, solvent, and reaction time (Table 1). Pyrrolo[3,4-c]carbazole-1,3(2H,6H)-dione was obtained in 50% yield via the reaction between
Optimization of the reaction conditions a .

DIPEA: N,N-Diisopropylethylamine; DMF: dimethylformamide; DBU: 1,8-diazabicyclo[5.4.0]undec-7-ene; DIPA: diisopropylamine; NMP: N-Methyl-2-pyrrolidone; DMSO: dimethyl sulfoxide; DCE: 1,2-dichloroethane; DPPE: 1,2-Bis(diphenylphosphino)ethane.
Reaction conditions:
Isolated yields.
Pd(OAc)2 (20 mol%) and PCy3·HBF4 (40 mol%) were used for 20 h.
The optimized conditions for the model reaction were therefore 3-chloro-4-indolylmaleimide (
Further investigations were performed under the optimized reaction conditions to determine the substrate scope of the reaction. First, the effect of the substituent on the indole ring in the 3-chloro-4-indolylmaleimide was investigated. The electronic properties of the R1 group greatly affected the reaction yield (Table 2, products
Scope of substituted indoles a .

DIPA: diisopropylamine; DMSO: dimethyl sulfoxide.
Reaction conditions:

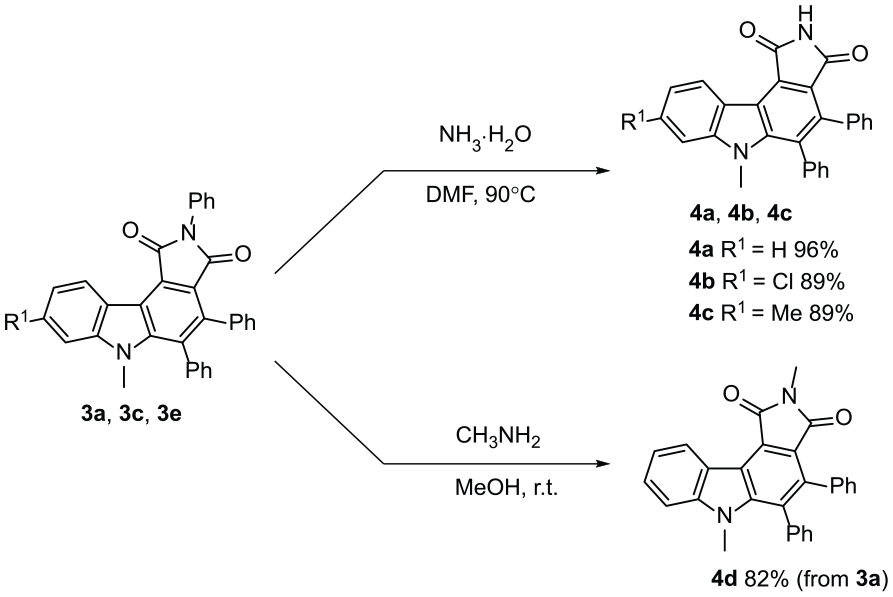
Transformations of the carbazole.

The proposed reaction pathway.
The use of this reaction to obtain structurally diverse pyrrolo[3,4-c]carbazole-1,3(2H,6H)-diones was explored by investigating transformations of products
Compounds
On the basis of relevant literature reports12–15 and our experimental results, the following possible reaction mechanism is proposed (Scheme 4). In the first step, Pd(0) catalyzes an oxidative-addition reaction of the C–Cl bond in substrate
Conclusion
In the current study, a novel and highly effective Pd-catalyzed addition/C–H activation/cyclization strategy, with 3-chloro-4-indolylmaleimides and alkynes as starting materials, has been used for the synthesis of pyrrolo[3,4-c]carbazole-1,3(2H,6H)-diones. These compounds have been previously shown to demonstrate a wide range of biological activities.1–7 The method gives high yields and tolerates a wide range of functional groups. The obtained target compounds have high conversion potential.
Experimental section
Experimental
All reagents used in the synthesis were obtained commercially and used without further purification, unless otherwise specified. Melting points were determined with a BÜCHI Melting Point B-450 apparatus (BüchiLabortechnik, Flawil, Switzerland) and are uncorrected. The 1H NMR (500 MHz) and 13C NMR (125 MHz) spectra were recorded on a Bruker ANANCE III spectrometer using TMS as the internal standard, and the coupling constants are reported in hertz. CDCl3 or DMSO (dimethyl sulfoxide) was used as the solvent, and chemical shifts (δ) are reported in parts per million (ppm). The reactions were followed by thin-layer chromatography (TLC) on GF-254 glass-packed precoated silica gel plates and visualized in an iodine chamber or with a ultraviolet (UV) lamp. Column chromatography purifications were performed on silica gel (200–300 mesh). High-resolution mass spectra (HRMS) were recorded on a Thermo Scientific LTQ Orbitrap XL mass instrument (ESI). Anhydrous THF and toluene were distilled from sodium-benzophenone. Anhydrous CH2Cl2, DMF, and DMSO were distilled from calcium hydride.
General procedure for the synthesis of pyrrolo[3,4-c]carbazole-1,3(2H,6H)-diones 3a–i
To a dried Schlenk tube were added 3-chloro-4-indolylmaleimide
6-Methyl-2,4,5-triphenylpyrrolo[3,4-c]carbazole-1,3(2H,6H)-dione (3a )
Yellow solid, 94% yield, m.p.: >250°C. 1H NMR (500 MHz, CDCl3): δ 9.31 (d, J = 7.9 Hz, 1H), 7.67–7.59 (m, 1H), 7.57–7.45 (m, 4H), 7.42–7.46 (m, 3H), 7.33–7.27 (m, 3H), 7.26–7.19 (m, 5H), 7.15 (dd, J = 6.6, 2.9 Hz, 2H), 3.25 (s, 3H). 13C NMR (125 MHz, CDCl3): δ 168.0, 167.6, 144.2, 142.8, 137.8, 136.3, 136.1, 132.2, 131.3 (2C), 130.2, 130.1 (2C), 128.8 (2C), 128.5, 127.8, 127.7 (2C), 127.6, 127.2 (2C), 127.0, 126.8 (2C), 126.3, 125.2, 121.2, 120.5, 120.3, 120.1, 109.0, 32.6; HRMS (ESI): m/z calcd for C33H23N2O2 [M + H]+: 479.1754, found: 479.1767.
8-Fluoro-6-methyl-2,4,5-triphenylpyrrolo[3,4-c]carbazole-1,3(2H,6H)-dione (3b )
Yellow solid, 68% yield, m.p.: >250°C. 1H NMR (500 MHz, CDCl3): δ 9.24 (dd, J = 8.8, 5.7 Hz, 1H), 7.53–7.44 (m, 4H), 7.41–7.33 (m, 1H), 7.33–7.26 (m, 3H), 7.25–7.16 (m, 5H), 7.14–7.09 (m, 3H), 7.03 (dd, J = 9.7, 2.3 Hz, 1H), 3.19 (s, 3H). 13C NMR (125 MHz, CDCl3): δ 167.9, 167.4, 164.5 (d, J = 246.5 Hz), 145.3 (d, J = 12.1 Hz), 143.3, 137.6, 136.0, 135.8, 132.1, 131.2 (2C), 130.3, 130.0 (2C), 128.8 (2C), 127.9, 127.8 (d, J = 10.4 Hz), 127.7 (2C), 127.6, 127.2 (2C), 127.1, 126.8 (2C), 124.7, 120.5, 120.0, 116.9, 109.4 (d, J = 23.7 Hz), 96.1 (d, J = 27.2 Hz), 32.8; HRMS (ESI): m/z calcd for C33H22FN2O2 [M + H]+: 497.1655, found: 497.1661.
8-Chloro-6-methyl-2,4,5-triphenylpyrrolo[3,4-c]carbazole-1,3(2H,6H)-dione (3c )
Yellow solid, 66% yield, m.p.: >250°C. 1H NMR (500 MHz, CDCl3): δ 9.20 (d, J = 8.4 Hz, 1H), 7.53–7.44 (m, 4H), 7.42–7.33 (m, 3H), 7.33–7.26 (m, 3H), 7.26–7.17 (m, 5H), 7.17–7.09 (m, 2H), 3.21 (s, 3H). 13C NMR (125 MHz, CDCl3): δ 167.9, 167.4, 144.8, 143.1, 138.1, 136.0, 135.8, 134.5, 132.0, 131.2 (2C), 130.5, 130.0 (2C), 128.8 (2C), 128.0, 127.8 (2C), 127.7, 127.3 (2C), 127.2, 127.1, 126.8 (2C), 125.2, 121.7, 120.6, 119.7, 119.1, 109.3, 32.8l; HRMS (ESI): m/z calcd for C33H22ClN2O2 [M + H]+: 513.1370, found: 513.1363.
9-Chloro-6-methyl-2,4,5-triphenylpyrrolo[3,4-c]carbazole-1,3(2H,6H)-dione (3d )
Yellow solid, 58% yield, m.p.: >250°C. 1H NMR (500 MHz, CDCl3): δ 9.29 (d, J = 2.1 Hz, 1H), 7.55 (dd, J = 8.7, 2.2 Hz, 1H), 7.53–7.45 (m, 4H), 7.41–7.34 (m, 1H), 7.32–7.26 (m, 5H), 7.26–7.17 (m, 5H), 7.17–7.09 (m, 2H), 3.23 (s, 3H). 13 C NMR (125 MHz, CDCl3): δ 167.8, 167.4, 143.0, 142.4, 138.3, 136.0, 135.8, 132.0, 131.3 (2C), 130.6, 130.0 (2C), 128.9 (2C), 128.6, 127.9, 127.75 (2C), 127.7, 127.3, 127.2, 126.8 (2C), 126.6 (2C), 125.6, 125.5, 121.4, 120.5, 119.2, 110.1, 32.8. HRMS (ESI): m/z calcd for C33H22ClN2O2 [M + H]+: 513.1370, found: 513.1377.
6,8-Dimethyl-2,4,5-triphenylpyrrolo[3,4-c]carbazole-1,3(2H,6H)-dione (3e )
Yellow solid, 96% yield, m.p.: >250°C. 1H NMR (500 MHz, CDCl3): δ 9.16 (d, J = 8.1 Hz, 1H), 7.54–7.43 (m, 4H), 7.40–7.32 (m, 1H), 7.32–7.26 (m, 2H), 7.28–7.13 (m, 8H), 7.18–7.09 (m, 2H), 3.22 (s, 3H), 2.61 (s, 3H). 13C NMR (125 MHz, CDCl3): δ 168.1, 167.7, 144.7, 142.9, 139.2, 137.3, 136.5, 136.2, 132.3, 131.3 (2C), 130.1 (2C), 130.0, 128.8 (2C), 127.7, 127.6 (2C), 127.5, 127.2 (2C), 127.0, 126.9 (2C), 125.9, 124.6, 122.8, 120.5, 119.9, 118.3, 109.2, 32.6, 22.5; HRMS (ESI): m/z calcd for C34H25N2O2 [M + H]+: 493.1916, found: 493.1922.
9-Methoxy-6-methyl-2,4,5-triphenylpyrrolo[3,4-c]carbazole-1,3(2H,6H)-dione (3f )
Orange solid, 88% yield, m.p.: >250°C. 1H NMR (500 MHz, CDCl3): δ 8.88 (d, J = 2.4 Hz, 1H), 7.54–7.45 (m, 4H), 7.41–7.33 (m, 1H), 7.30–7.27 (m, 5H), 724–7.18 (m, 5H), 7.16–7.11 (m, 2H), 4.02 (s, 3H), 3.22 (s, 3H). 13C NMR (125 MHz, CDCl3): δ 168.2, 167.6, 154.9, 143.0, 139.1, 137.5, 136.3, 136.1, 132.2, 131.3, 130.2, 130.1 (2C), 128.9 (2C), 127.8, 127.7, 127.6 (2C), 127.2 (2C), 127.1 (2C), 127.0, 125.3, 120.8, 120.2, 119.7, 118.8, 109.9, 107.3, 56.0, 32.7; HRMS (ESI): m/z calcd for C34H25N2O3 [M + H]+: 509.1865, found: 509.1859.
9-(Benzyloxy)-6-methyl-2,4,5-triphenylpyrrolo[3,4-c]carbazole-1,3(2H,6H)-dione (3g )
Yellow solid, 82% yield, m.p.: >250°C. 1H NMR (500 MHz, CDCl3): δ 9.02 (d, J = 2.4 Hz, 1H), 7.60–7.54 (m, 2H), 7.55–7.45 (m, 4H), 7.46–7.32 (m, 5H), 7.34–7.24 (m, 4H), 7.25–7.17 (m, 5H), 7.16–7.12(m, 2H), 5.29 (s, 2H), 3.23 (s, 3H). 13C NMR (125 MHz, CDCl3): δ 168.2, 167.6, 154.0, 143.0, 139.2, 137.5, 137.2, 136.3, 136.1, 132.2 (2C), 131.3 (2C), 130.2, 130.1, 128.9 (2C), 128.5 (2C), 128.0 (2C), 127.9, 127.8, 127.7, 127.6 (2C), 127.2 (2C), 127.1, 127.0 (2C), 125.3, 120.8, 120.2, 119.7, 119.3, 110.0, 108.7, 70.8, 32.7; HRMS (ESI): m/z calcd for C40H29N2O3 [M + H]+: 585.2178, found: 585.2176.
6-Benzyl-2,4,5-triphenylpyrrolo[3,4-c]carbazole-1,3(2H,6H)-dione (3h )
Yellow solid, 95% yield, m.p.: >250°C. 1 H NMR (500 MHz, CDCl3): δ 9.40 (d, J = 7.9 Hz, 1H), 7.60–7.41 (m, 6H), 7.41–7.34 (m, 1H), 7.29 (d, J = 10.6 Hz, 1H), 7.18–7.07 (m, 9H), 7.04–6.97 (m, 2H), 6.95–6.89 (m, 2H), 6.56–6.49 (m, 2H), 5.04 (s, 2H). 13C NMR (125 MHz, CDCl3): δ 168.0, 167.5, 143.9, 142.2, 138.1, 136.8, 136.0, 135.5, 132.2, 130.8 (2C), 130.6, 129.9 (2C), 128.8 (2C), 128.7 (2C), 128.3 (2C), 127.6, 127.5, 127.4 (2C), 127.1 (2C), 127.0, 126.9, 126.8 (2C), 126.4, 125.3, 125.2, 121.6, 120.8, 120.7, 120.5, 109.8, 47.8; HRMS (ESI): m/z calcd for C39H27N2O2 [M + H]+: 555.2073, found: 555.2064.
6-Methyl-2-phenyl-4,5-dipropylpyrrolo[3,4-c]carbazole-1,3(2H,6H)-dione (3i )
Yellow solid, 95% yield, m.p.: >250°C. 1H NMR (500 MHz, CDCl3): δ 9.22 (d, J = 7.9 Hz, 1H), 7.63–7.56 (m, 1H), 7.55 (d, J = 4.3 Hz, 4H), 7.48–7.38 (m, 2H), 7.34 (d, J = 7.3 Hz, 1H), 4.15 (s, 3H), 3.35–3.30 (m, 2H), 3.27–3.19 (m, 2H), 1.81–1.66 (m, 4H), 1.19 (t, J = 7.3 Hz, 3H), 1.15 (t, J = 7.3 Hz, 3H). 13C NMR (125 MHz, CDCl3): δ 168.9, 168.2, 144.0, 143.9, 139.4, 132.4, 130.0, 128.9 (2C), 128.0, 127.6, 127.0 (2C), 126.0, 124.4, 120.8 (2C), 120.6, 119.8, 108.8, 32.8, 29.9, 29.6, 25.8, 25.2, 14.7, 14.2; HRMS (ESI): m/z calcd for C27H27N2O2 [M + H]+: 411.2073, found: 411.2072.
6-Methyl-4,5-diphenylpyrrolo[3,4-c]carbazole-1,3(2H,6H)-dione (4a )
To a Schlenk tube was added compound
8-Chloro-6-methyl-4,5-diphenylpyrrolo[3,4-c]carbazole-1,3(2H,6H)-dione (4b )
According to the procedure used to prepare
6,8-Dimethyl-4,5-diphenylpyrrolo[3,4-c]carbazole-1,3(2H,6H)-dione (4c )
According to the procedure used to prepare
2,6-Dimethyl-4,5-diphenylpyrrolo[3,4-c]carbazole-1,3(2H,6H)-dione (4d )
A mixture of
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We gratefully acknowledge the Natural Science Foundation of Zhejiang (LY18H300009), the Postdoctoral Science Foundation of China (2014M550256), the State Key Laboratory of Drug Research (SIMM1601KF-04), and the National Natural Science Foundation of China (8150131067).



