Abstract
The solubilities of benzoic acid and its nitrated derivatives (3-nitrobenzoic acid and 3,5-dinitrobenzoic acid) in seven pure solvents—water, methanol, ethanol, acetonitrile, dichloromethane, toluene, and ethyl acetate—were determined experimentally over a temperature range from 273.15 K to 323.15 K under 101.3 kPa. The solubility of the above substances in these solvents increased with temperature. The solubility values of benzoic acid in these seven solvents follow the following order: ethanol > ethanol > acetonitrile > ethyl acetate > dichloromethane > toluene > water, while the solubility values of its nitrification derivatives in these seven solvents follow the following order: methanol > ethanol > ethyl acetate > acetonitrile > dichloromethane > toluene > water. The solubility of benzoic acid, 3-nitrobenzoic acid, and 3,5-dinitrobenzoic acid were significantly different in the same solvent. The solubilities obtained are very helpful in improving the recrystallization and yields of 3-nitrobenzoic acid and 3,5-dinitrobenzoic acid.

Introduction
Benzoic acid and its nitro-derivatives (3-nitrobenzoic acid and 3,5-dinitrobenzoic acid) are important intermediates in organic synthesis,1–4 for example, in the production of the diagnostic drug amidotrizoic acid, a positive contrast agent for X-ray diagnosis (see Figure 1). As shown in Figure 2, amidotrizoic acid is obtained by reduction, iodination, and acetylation of 3,5-dinitrobenzoic acid. It is used in the preparation of diatrizoate meglumine, a contrasting agent. In addition, it is also used for urography, cardiovascular and cerebrovascular angiography, and various injection angiographies such as into joint cavities. 5 3,5-diaminobenzoic acid is used in the dye industry 6 and in the manufacture of liquid crystal intermediates such as the diphenyl ester of 3,5-diaminobenzoic acid. These are new fine chemical products with high added values. 7

The chemical structure of amidotrizoic acid.

Synthesis of amidotrizoic acid.
Nitration of benzoic acid gives 3-nitro- and 3,5-dinitro-benzoic acids with batch reactors being used in industry. However, although the conversion rate has been improved, the isolated yields of 3-nitrobenzoic acid and 3,5-dinitrobenzoic acid remain relatively low.8,9 Advanced microchannel reactors have a very high specific surface areas, thousands of times that of a conventional reactor and can realize the rapid exchange of reaction heat and so avoid reactions getting out of control. 10 Although the current microchannel reactors make up for the shortcomings of traditional kettle reactions and improve the rate of nitration of benzoic acid, the yield and purity of the products obtained remain relatively low because a large number of products are dissolved in the nitration mother liquor and cannot be easily removed.11,12 It is necessary to find better recrystallization solvents to improve the purities of the products.
In this paper, the solubility of benzoic acid and its nitro-derivatives was investigated in seven common pure solvents, including water, methanol, ethanol, acetonitrile, toluene, dichloromethane, and ethyl acetate; these were measured by a static method13,14 in the temperature range from 273.15 K to 323.15 K under P = 0.1 MPa. The results will help compare the solubility of benzoic acid, 3-nitrobenzoic acid, and 3,5-dinitrobenzoic acid and the optimization of the nitration of benzoic acid in industry.
Results and discussion
Solubility data
The measured solubilities of benzoic acid, 3-nitrobenzoic acid, and 3,5-dinitrobenzoic acid in water, methanol, ethanol, acetonitrile, dichloromethane, toluene, and ethyl acetate within the temperature range from 273.15 K to 323.15 K are presented in Table 1 and graphically shown in Figures 3–5. The figures show that the solubilities of these three compounds in the different solvents increase with increasing temperature.
Solubilities of benzoic acid, 3-nitrobenzoic acid, and 3,5-dinitrobenzoic acid.
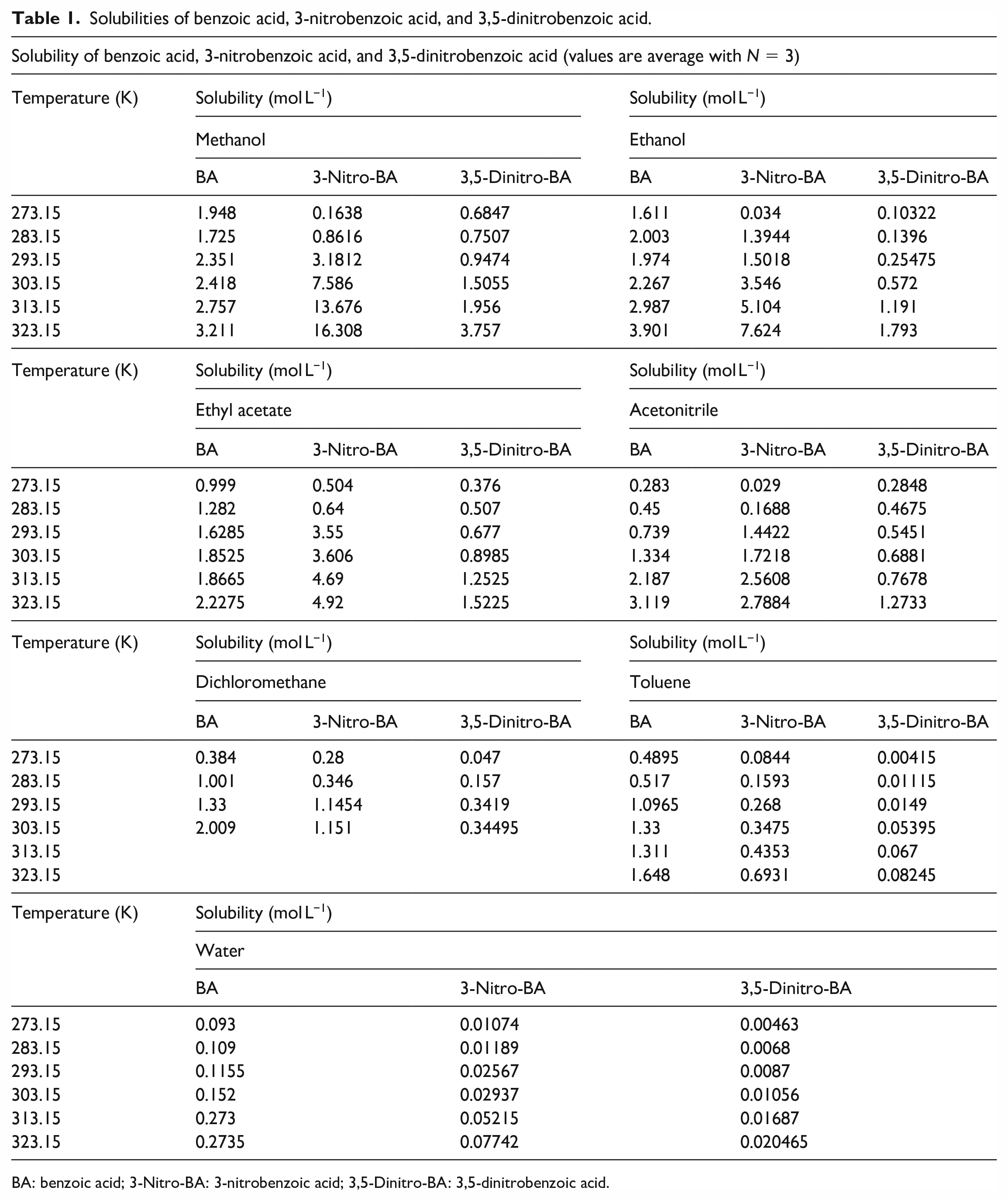
BA: benzoic acid; 3-Nitro-BA: 3-nitrobenzoic acid; 3,5-Dinitro-BA: 3,5-dinitrobenzoic acid.

The solubility of benzoic acid in seven solvents.
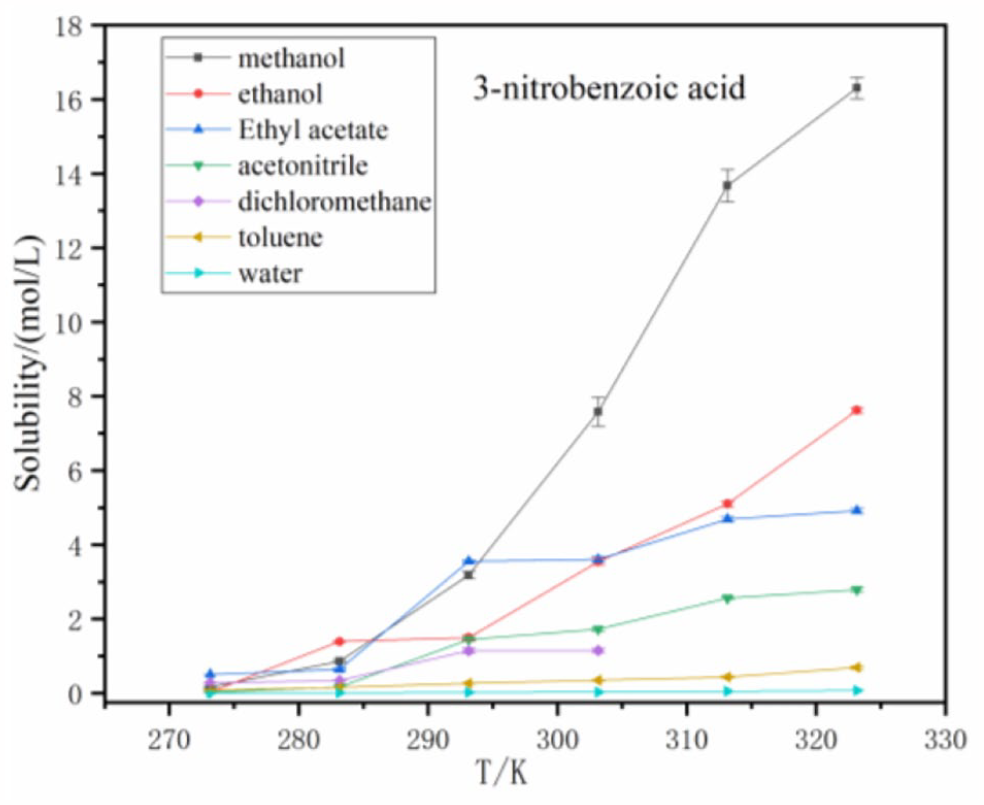
The solubility of 3-nitrobenzoic acid in seven solvents.
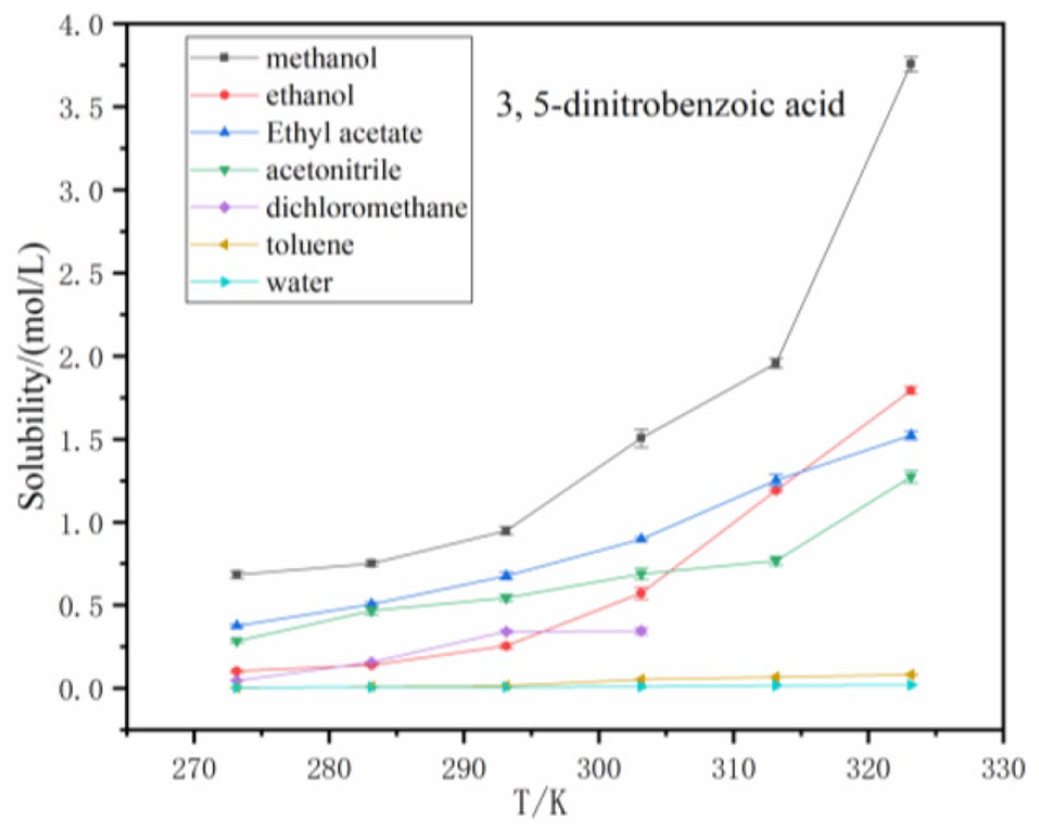
The solubility of 3,5-dinitrobenzoic acid in seven solvents.
Figure 3 further demonstrates that the solubility of benzoic acid in methanol and ethanol are greater than those in the other solvents according to the following order: ethanol > methanol > acetonitrile > ethyl acetate > dichloromethane > methylbenzene > water. The solubility of 3-nitrobenzoic acid in all solvents is larger than 3,5-dinitrobenzoic acid within the temperature range from 273.15 K to 323.15 K. In Figure 4, the solubility values of 3-nitrobenzoic acid in the seven solvents are in the following order: methanol > ethanol > ethyl acetate > acetonitrile > dichloromethane > methylbenzene > water. As shown in Figure 5, the order of the solubilities of 3,5-dinitrobenzoic is the same as 3-nitrobenzoic acid, that is, methanol > ethanol > ethyl acetate > acetonitrile > dichloromethane > methylbenzene > water.
A comparison of the solubilities of benzoic acid, 3-nitrobenzoic acid, and 3,5-dinitrobenzoic acid in the same solvent (Figures 6–9) show that in methanol, ethanol, and ethyl acetate, the solubility of benzoic acid is higher than that of 3,5-dinitrobenzoic acid and 3-nitrobenzoic acid between 273.15 K and 283.15 K. When acetonitrile is the solvent, the solubility of benzoic acid is almost the same as 3,5-dinitrobenzoic acid and also higher than that of 3-nitrobenzoic acid over the temperature range from 273.15 K to 283.15 K. However, the solubility of 3-nitrobenzoic acid is higher than those of benzoic acid and 3,5-dinitrobenzoic acid above 283.15 K in the above four solvents.
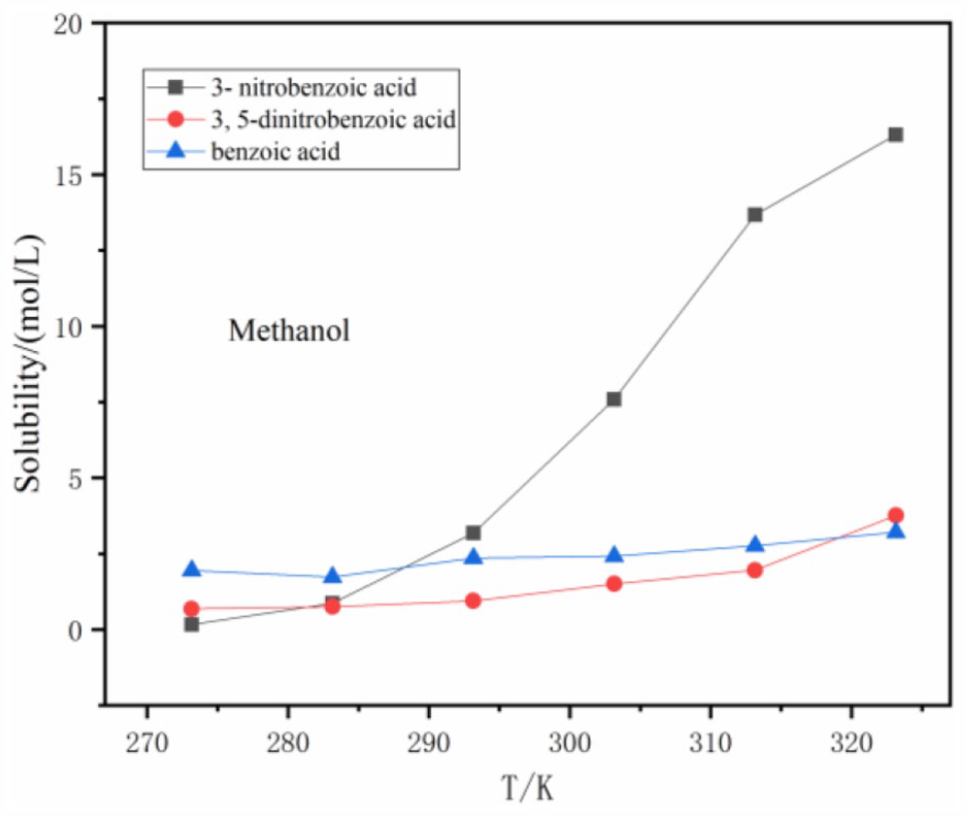
Solubility comparison in methanol.

Solubility comparison in ethanol.

Solubility comparison in ethyl acetate.

Solubility comparison in acetonitrile.
However, Figures 10–12 show that the solubility of benzoic acid is higher than that of 3,5-dinitrobenzoic acid and 3-nitrobenzoic acid at temperature between 273.15 K and 323.15 K in dichloromethane, methylbenzene, and water (as dichloromethane has a boiling point of 312.9 K and because of its volatile nature, it was only studied up to 303.15 K). With increased temperature, the solubility of benzoic acid in three solvents changed significantly, but the solubility of the three compounds in water increased only slightly. From the general trend that was observed, the solubility of 3-nitrobenzoic acid is greater than that of 3,5-dinitrobenzoic acid.

Solubility comparison in toluene.

Solubility comparison in dichloromethane.

Solubility comparison in water.
Discussion and analysis
The dipole moments of benzoic acid, 3-nitrobenzoic acid, and 3,5-dinitrobenzoic acid are 1.27D, 5.08D, and 4.38D, respectively. According to the solubility data measured by experiment, the solubility of substances with larger polarities in polar solvents is generally higher than that of substances with lower polarity.15,16 For example, the solubilities of 3-nitrobenzoic acid and benzoic acid in polar solvents such as methanol and ethanol are greater than that of 3,5-dinitrobenzoic acid. In a practical application, when nitro-derivatives are prepared from benzoic acid by nitration, the mother liquor should be cooled by ice water but at this point the yield is not high. At this time, the aqueous phase still contains more products, so a more polar solvent should be used to extract the organic matter from the aqueous phase.17–19 The solvent can then be removed by vacuum rotary evaporation to obtain the product. 20
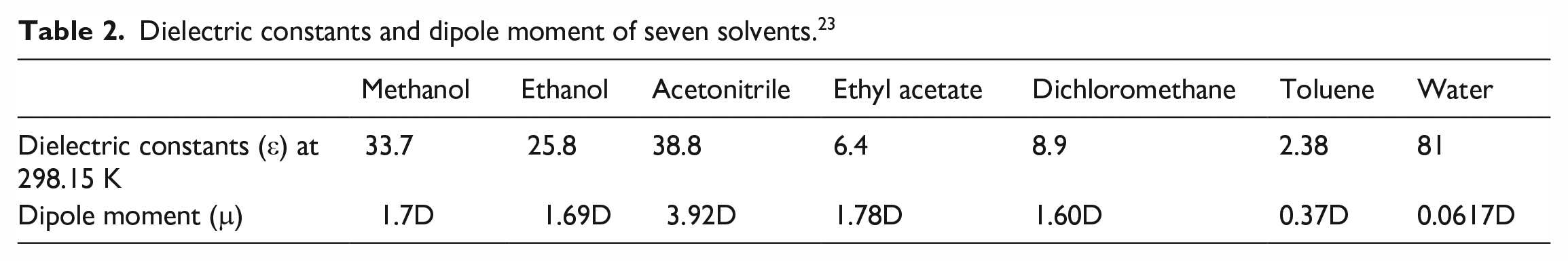
If the purity of the product obtained is not high, combinations of an appropriate efficient and a poor solvent can be chosen for recrystallization of the products according to the differences in solubility of the three substances. The dielectric constants and dipole moment of seven solvents are shown in Table 2. From Figure 13, we know that the solubility of 3-nitrobenzoic acid is large, despite changes in temperature, in methanol and ethanol but its solubility in water is very small. Therefore, methanol or ethanol together with the poor solvent water can be used for its recrystallization giving 3-nitrobenzoic acid with >98% purity. According to Figure 14, 3,5-dinitrobenzoic acid has a greater solubility in methanol. The solubility of 3,5-dinitrobenzoic acid in toluene and water is very small, if the 3,5-dinitrobenzoic acid is recrystallized, the efficient solvent methanol can be selected with the poor solvent toluene to obtain 3,5-dinitrobenzoic acid with a purity greater than >98.5%.
Dielectric constants and dipole moment of seven solvents. 23

Chemical structures of (a) benzoic acid, (b) 3-nitrobenzoic acid, and (c) 3,5-dinitrobenzoic acid.

Schematic diagram of experimental apparatus: I, smart thermostatic water bath; II, mercury-in-glass thermometer; III, magnetic stirrer; IV, stirrer controller; V, jacketed glass vessel; VI, sampling port; VII, condenser.
Conclusion
In this work, the equilibrium solubilities were obtained experimentally for benzoic acid, 3-nitrobenzoic acid, and 3,5-dinitrobenzoic acid in seven pure solvents within the temperature range from 273.15 K to 323.15 K under 101.3 kPa. The solubilities of the solutes in the selected pure solvents increased with increasing temperatures. At any given temperature, the solubility of benzoic acid in these seven solvents was in the following order: ethanol > methanol > acetonitrile> ethyl acetate > dichloromethane> toluene > water, and the solubility values of 3-nitrobenzoic acid and 3,5-dinitrobenzoic acid in these seven solvents followed the slightly different order: methanol > ethanol > ethyl acetate > acetonitrile > dichloromethane> toluene > water. This study also compared the solubilities of benzoic acid, 3-nitrobenzoic acid, and 3,5-dinitrobenzoic acid in same solvent. On the whole, the solubility of 3-nitrobenzoic acid is higher than that of 3,5-dinitrobenzoic acid in same solvent. All three compounds are the most soluble in methanol and ethanol.
Experimental
Materials and apparatus
Benzoic acid, 3-nitrobenzoic acid, and 3,5-dinitrobenzoic acid having a mass fraction of 0.99 were provided by Nanjing Wanqing Glass Instrument Co. Ltd. The solvents (water, methanol, ethanol, acetonitrile, dichloromethane, toluene, and ethyl acetate) were purchased from Sinopharm Chemical Reagent Co. Ltd., China, and they were of analytical grade. They were used without any further purification. Detailed information on these materials is presented in Table 3.
Detailed information on the materials used in the work, p = 101.3 KPa.

HPLC: high-performance liquid phase chromatograph; GC: gas chromatography.
This work. The standard uncertainties u are u(T) = 2 K, u(p) = 400 Pa, u(ρ) = 1.4 kg m−3.
An analytical balance (model SQP), which had a standard uncertainty of 0.0001 g provided by Satorius Scientific Instrument (Beijing), was employed to determine the mass of the saturated solutions. The system temperature was controlled using a low temperature and thermostatic ethanol bath (Model: DFY-5/20; standard uncertainty: 0.05 K), which was provided by Nanjing Kobor Instrument Equipment Co. Ltd. The experimental apparatus comprised of a 50 mL jacketed glass vessel with a magnetic stirrer and a circulating ethanol bath used for controlling the temperature of the system. To avoid escape of the solvent, a condenser was employed.
Solubility determination
In this work, the solid−liquid equilibrium for the binary systems of benzoic acid, 3-nitrobenzoic acid, and 3,5-dinitrobenzoic acid were obtained using the isothermal saturation method12,13 in the temperatures ranging from 273.15 K to 323.15 K under 101.3 kPa. As shown in Figure 14, the method used for the experimental measurements is same as that in the literature.21,22
In each experiment, about 20 mL of solvent and excess solute were added into the 50 mL glass vessel. The temperature was maintained at the desired value by circulating water from the thermostatic ethanol-circulator bath through the outer jacket. The actual dissolution system temperature was displayed by a mercury glass micro thermometer (standard uncertainty: 0.05 K) inserted in the inner chamber of the glass vessel jacket. A magnetic stirrer was used to mix the solid and solvent adequately. To obtain the accurate equilibrium time, about 0.1 mL was removed from the liquid phase system every hour using a microsampler, and then analyzed by HPLC. The results illustrated that 6 h was enough to achieve the solution system equilibrium. When the solid−liquid system arrived at equilibrium, the stirring was stopped, and the mixture was permitted to settle for 4 h before sampling. Then, the supernatant was withdrawn by a 1 mL preheated microsampler (to avoid precipitation of solids when they encounter a cool solvent), and the sample transferred quickly into a 50 mL preweighed volumetric flask. The sample was then diluted 50 ml using the corresponding solvent, and 20 μL of the solution was removed for analysis by HPLC. When the solubility was determined at one temperature, the residue, including any excess solid, was heated to another temperature, and the determination process was repeated.
Analytical method
The amounts of benzoic acid, 3-nitrobenzoic acid, and 3,5-dinitrobenzoic acid were determined using a WuFeng-LC-100 HPLC apparatus, which was equipped with a reverse phase column having a type of LP-C18 (150 mm × 4.6 mm). The column temperature was set to 303.15 K during the analysis. The wavelength of the UV detector was 254 nm. The mobile phase was a mixture of acetonitrile and water (acetonitrile: water = 42:58 containing 1 g L−1 tetrabutylammonium bromide) with a flow rate of 1.0 mL·min−1. Three samples were taken for each mixture at a certain temperature. The average value was considered as the final solubility data for the three compounds.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of the article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
