Abstract
A novel one-pot 1,3-dipolar cycloaddition/oxidation reaction of 1,4-quinones, aromatic aldehydes, and N-substituted amino esters to construct polysubstituted benzo[f]isoindole-4,9-diones induced by benzoic acid is reported. Based on optimized reaction conditions, various polysubstituted benzo[f]isoindole-4,9-diones derivatives are obtained in moderate yields. This straightforward methodology employs mild reaction conditions, a green oxidant, and readily available starting materials. Notably, a wide range of substrates is tolerated.
Introduction
Pyrrole and its derivatives are an important class of five-membered nitrogen-containing heterocyclic compounds that are present in numerous natural products and serve as intermediates in the synthesis of fine chemicals in the fields of medicine,1,2 food, common chemicals,3,4 printing and dyeing,5–7 and so on. The benzo[f]isoindole framework is the core structure of natural products exhibiting important biological activities. Examples include bioactive molecules such as bhimamycins C and D (active against human ovarian cancer cell lines),8–10 GR30921 (for solid tumors), 11 a potent anti-inflammatory that also exhibited analgesic activity, 12 and an azanaphthoquinone-pyrrole that plays an important role against different cancers 10 (Scheme 1).

Representative examples of natural products containing a biological activity.
Due to their wide utility as biologically active agents and key intermediates in organic synthesis, pyrrole derivatives have attracted significant attention from organic chemists, and a variety of efficient methods have been developed for their synthesis. Examples include Hantzsch,13–18 Trofimov,19–22 Mannich23–26 and Clauson–Kaas reactions,27–30 Friedel–Crafts,31–34 and [3+2] cycloadditions.35–39 In recent years, metal-free catalysis of various organic transformations has gained more and more attention.40,41 Benzoic acid, a high catalytically active, nontoxic, inexpensive, and readily available catalyst for the construction of five-membered heterocycles, has attracted the interest of numerous chemists.42,43
Recently, compared with the present diversified routes to pyrrole derivatives, synthetic protocols to the benzo[f]isoindole-4,9-dione framework are relatively limited with only a few methods having been reported.44–46 Lin et al.
45
have reported the silver(II)-mediated radical reactions of 2-substituted-1,4-naphthoquinones and α-keto acids to produce benzo[f]isoindole-4,9-diones. Our own team
46
has reported a molecular iodine induced 1,3-dipolar cycloaddition between quinone structures and diethyl N-substituted iminodiacetates to construct benzo[f]isoindole-4,9-diones, as shown in Scheme 2. Encouraged by this achievement and our ongoing study of quinones,42,46–48 we herein report a straightforward and effective method for the construction of polysubstituted benzo[f]isoindole-4,9-dione

Examples of benzo[f]isoindole-4,9-dione synthesis.
Results and discussion
Our initial investigations focused on examining the feasibility of the [3+2] cycloaddition reaction of 1,4-naphthoquinone (
Optimization of the reaction conditions a .
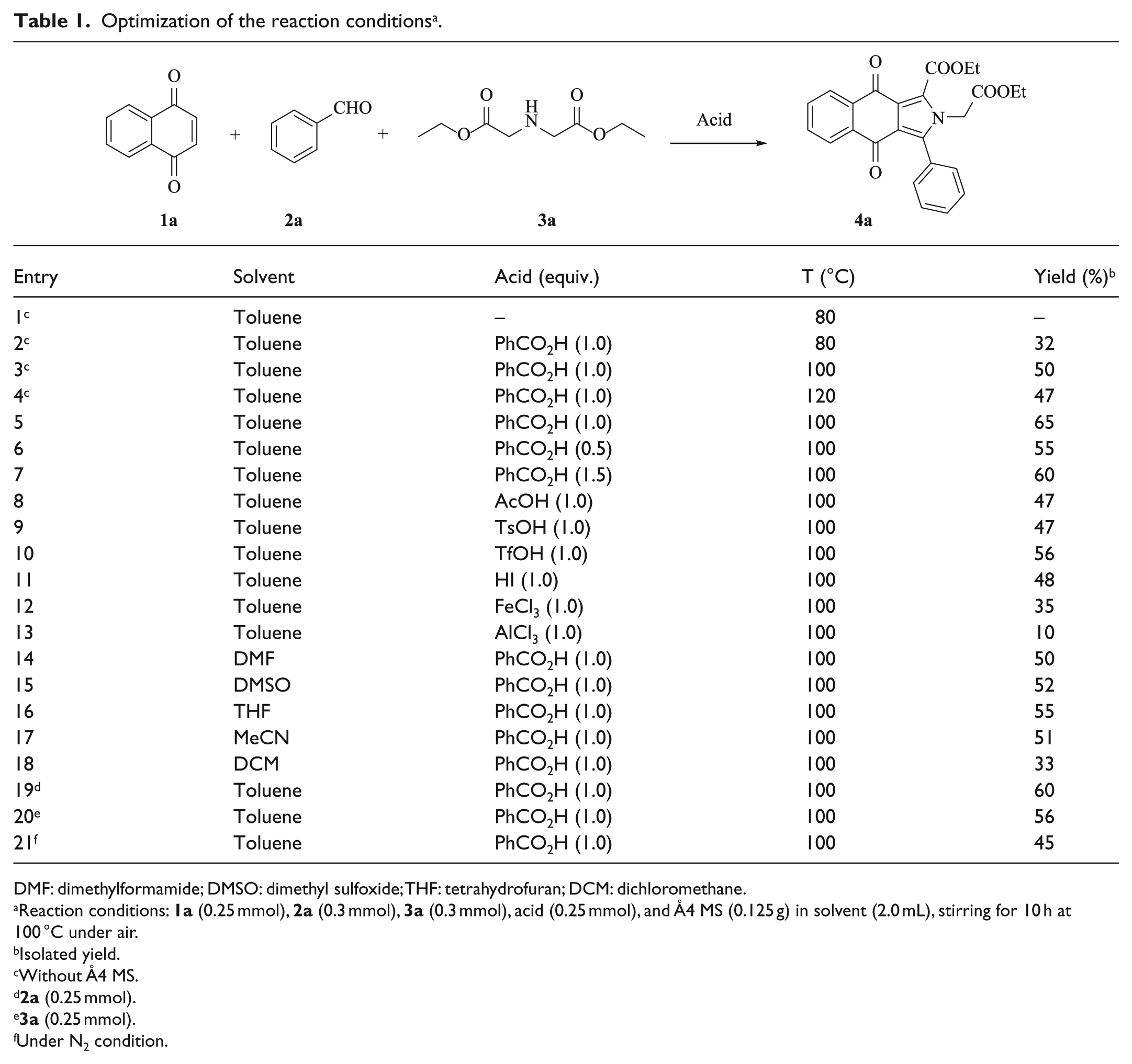
DMF: dimethylformamide; DMSO: dimethyl sulfoxide; THF: tetrahydrofuran; DCM: dichloromethane.
Reaction conditions:
Isolated yield.
Without Å4 MS.
Under N2 condition.
No target compound
With optimized reaction conditions in hand, we next evaluated the scope of the reaction for the construction of various benzo[f]isoindole-4,9-diones. A wide range of aromatic aldehydes
Reaction conditions:
Isolated yield.
In addition, benzaldehydes bearing electron-donating groups (e.g. 4-NMe2, 4-Me, 4-MeO, 3-MeO, 2-MeO, 2,5-2MeO) gave similar results, that is, products
In order to expand the application of this reaction, different amino acid esters were also investigated under the optimized reaction conditions, as shown in Table 3. When ethyl (methylamino)acetate (
Reaction conditions:
Isolated yield.
Taken together with previous results from the literature,
46
the above results have led us to propose a possible mechanism (Scheme 3). Initially, benzaldehyde would react with diethyl iminodiacetate in the presence of PhCO2H to afford zwitterionic intermediate

A possible reaction mechanism for the synthesis of
Conclusion
In conclusion, we have successfully developed a simple and novel method for the synthesis of polysubstituted benzo[f]isoindole-4,9-diones through a PhCO2H-induced [3+2] cycloaddition/oxidation tandem reaction. Under optimized reaction conditions, various easily accessible aromatic aldehydes, N-substituted amino esters, and 1,4-quinones were reacted to give the polysubstituted benzo[f]isoindole-4,9-diones derivatives in moderate-to-good yields. Moreover, the benzo[f]isoindole-4,9-dione products contain one or two ester groups, which can be further transformed into useful building blocks. Finally, thanks to the important biological activities of benzo[f]isoindole-4,9-diones, further studies in our laboratory will focus on expanding the scope of this method.
Experimental section
General information
All chemicals were purchased from commercial vendors and were used as received without further purification. The 1H and 13C nuclear magnetic resonance (NMR) spectra were recorded with a Bruker AM 500 spectrometer at 500 and 125 MHz, respectively, in CDCl3 using tetramethylsilane (TMS) as an internal standard. Chemical shifts (δ) are reported in parts per million (ppm) and the following abbreviations are used to identify the multiplicities: s (singlet), d (doublet), t (triplet), q (quartet), m (multiplet), b (broad), or combinations thereof. High-resolution mass spectrometry (HRMS) data were obtained with a Thermo Scientific LTQ Orbitrap XL mass spectrometer (ESI). Thin-layer chromatography was performed on pre-coated glass plates and visualized with UV light at 254 nm. Flash column chromatography was performed on brand silica gel (200–300 mesh size).
General procedure for the synthesis of 4a
1,4-Naphthoquinone (
Ethyl 2-(2-ethoxy-2-oxoethyl)-4,9-dioxo-3-phenyl-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 2-(2-ethoxy-2-oxoethyl)-3-(4-fluorophenyl)-4,9-dioxo-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 3-(4-chlorophenyl)-2-(2-ethoxy-2-oxoethyl)-4,9-dioxo-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 3-(4-bromophenyl)-2-(2-ethoxy-2-oxoethyl)-4,9-dioxo-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 2-(2-ethoxy-2-oxoethyl)-3-(4-nitrophenyl)-4,9-dioxo-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 3-(4-cyanophenyl)-2-(2-ethoxy-2-oxoethyl)-4,9-dioxo-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 3-(2-chlorophenyl)-2-(2-ethoxy-2-oxoethyl)-4,9-dioxo-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 2-(2-ethoxy-2-oxoethyl)-3-(3-nitrophenyl)-4,9-dioxo-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 3-(4-(dimethylamino)phenyl)-2-(2-ethoxy-2-oxoethy-l)-4,9-dioxo-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 2-(2-ethoxy-2-oxoethyl)-4,9-dioxo-3-(p-tolyl)-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 2-(2-ethoxy-2-oxoethyl)-3-(4-methoxyphenyl)-4,9-dioxo-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 2-(2-ethoxy-2-oxoethyl)-3-(3-methoxyphenyl)-4,9-dioxo-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 2-(2-ethoxy-2-oxoethyl)-3-(2-methoxyphenyl)-4,9-dioxo-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 3-(2,5-dimethoxyphenyl)-2-(2-ethoxy-2-oxoethyl)-4,9-dioxo-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 2-(2-ethoxy-2-oxoethyl)-3-(naphthalen-1-yl)-4,9-dioxo-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 2-(2-ethoxy-2-oxoethyl)-4,9-dioxo-3-(thiophen-2-yl)-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 2-(2-ethoxy-2-oxoethyl)-4,9-dioxo-3-propyl-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 2-methyl-4,9-dioxo-3-phenyl-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 2-benzyl-4,9-dioxo-3-phenyl-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 4,9-dioxo-2,3-diphenyl-4,9-dihydro-2H-benzo[f]isoindole-1-carboxylate (
Ethyl 2-(2-ethoxy-2-oxoethyl)-4,11-dioxo-3-phenyl-4,11-dihydro-2H-naphtho[2,3-f]isoindole-1-carboxylate (
Supplemental Material
Supplementary_Material-revised – Supplemental material for Acid-induced intermolecular [3+2] cycloaddition/oxidation to synthesize polysubstituted benzo[f]isoindole-4,9-diones
Supplemental material, Supplementary_Material-revised for Acid-induced intermolecular [3+2] cycloaddition/oxidation to synthesize polysubstituted benzo[f]isoindole-4,9-diones by Zhang-qi Lin, Chao-dong Li, Yu-jin Li, Yong-jian Zhang, Zi-chun Zhou, Qing Ye and Jian-rong Gao in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Natural Science Foundation of China (Grant No. 21606201), the National Natural Science Foundation of Zhejiang (Grant No. LY13B020016), and the Technological Innovation Program of Zhejiang Province (Zhejiang Xinmiao Talents Program; Grant No. 2017R403066).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.







