Abstract
To improve the photocatalytic activity of TiO2, a series of NiO–TiO2 nanotubes (NTbs) is prepared by impregnating TiO2 nanotubes in a solution of NiCl2·6H2O at different concentrations. Self-organized TiO2 nanotubes are prepared by a two-step anodization process. Scanning electron microscopy images show that large particle agglomerates are formed for higher precursor concentrations, and X-ray energy-dispersive spectroscopy results indicate that the atomic percentages of Ni in the NiO–TiO2 NTbs prepared with precursor concentrations of 100 and 300 mM are 1.95% and 4.23%, respectively. Electronic lifetime measurements show that the recombination rate of photogenerated electron–hole pairs is lower for NiO–TiO2 NTbs compared to that of TiO2. Specifically, the recombination rate of the sample prepared at 50 mM is the lowest, which is associated with the longest electron lifetime. Compared to unmodified TiO2 nanotubes, NiO–TiO2 NTbs exhibit improved results for the photocatalytic degradation of rhodamine B.

Introduction
The world is currently facing a major crisis regarding environmental pollution and sustainable development. With regard to ecological issues and environment protection, nanoscale semiconductor photocatalytic materials have attracted significant interest. In particular, titanium dioxide (TiO2) has been widely studied because of its stable chemical properties, non-toxicity, low cost, and excellent photocatalytic properties.1–3 Furthermore, titanium dioxide can be synthesized in various forms, such as TiO2 nanoparticles, TiO2 nanotubes, TiO2 nanowires, and TiO2 nanospheres.4–6 In particular, TiO2 nanotubes have a highly ordered structure and a large surface area, which can effectively increase the transfer rate of photogenerated electrons, reduce the recombination of photogenerated electron–hole pairs, and thus improve the photocatalytic reaction activity. 7 TiO2 nanotubes are prepared using a simple and widely used method; in addition, they can be used in dye-sensitized batteries, and are employed in the photodegradation of organic compounds, and the photolysis of aquatic hydrogen 8 and biological materials. 9 Although TiO2 nanotubes are widely used in many fields, the fact that pure TiO2 has a wide band gap (Eg = 3.2 eV for anatase) means that it can only be activated by UV light sources with wavelengths lower than 387 nm; this is 3%–4% of the whole spectrum of solar light. 10 However, photogenerated electron–hole pairs are easily recombined inside or on the surface of TiO2. 11 Previous studies have shown that most of the photogenerated electron–hole pairs (60%–80%) in a TiO2 colloid recombine in nanoseconds. Hence, they are lost in the form of heat and the utilization rate of photogenerated electrons is reduced. Therefore, in order to reduce the band gap, researchers have focused on the preparation and modification of TiO2 nanotubes.12,13 This can promote the photoresponse of TiO2 nanotubes in the visible light spectrum and increase the use of solar light, which in turn will improve the utilization rate of photoelectrons, inhibit the recombination of photogenerated electron–hole pairs, and improve the photocatalytic activity. In recent years, numerous researchers have developed new techniques for the fabrication of TiO2 nanotubes, such as the sol-gel process, the template method, hydrothermal synthesis, and anodic oxidation.14–16 Among these, TiO2 nanotubes prepared by anodic oxidation have gained more attention due to their large specific surface areas and highly ordered structures.
The anodic oxidation method generally uses pure titanium foil as the anode and an inert metal or a non-metal with good conductivity as the cathode, which are oxidized and etched in a specific electrolyte. In addition, the morphology and diameter of the TiO2 nanotubes can be controlled by parameters such as the anodizing voltage, the anodizing temperature, the anodizing time, the electrolyte composition, and the electrolyte pH. Sergiu et al. 17 alternately used high (120 V) and low (40 V) voltages during the preparation process. The anodic oxidation times during the high and low voltage were fixed at 1 and 5 min, respectively. TiO2 nanotube arrays with a geometrical morphology similar to a bamboo structure were successfully fabricated. Compared with smooth cylindrical TiO2 nanotubes formed by anodic oxidation under constant-voltage, the bamboo-like TiO2 nanotube arrays have larger specific surface areas. Meanwhile, in order to improve the usability of the TiO2 nanotubes, many studies have focused on the modification of TiO2 nanotube arrays. The photoelectron transfer efficiency and visible light response range can be effectively improved by doping with non-metal elements such as N, C, and F, with metal elements such as Pt, Ag, La, and Ni, or with metal oxides such as NiO, MoO3, and Cu2O. Likewise, several semiconductors—such as CdS, α-Fe2O3, PbS, and WO3—have been adopted to enhance the photocatalytic activity of TiO2. 18 The doping method is an important parameter. At present, doping methods include impregnation deposition, electrochemical deposition, photochemical deposition, ion-sputtering deposition, and chemical bath deposition.
As a p-type semiconductor material, NiO has unique catalytic, electrical, magnetic, and hole transport properties. When it is compounded with n-type TiO2 semiconductor materials, a p–n heterostructure composite material can be constructed. When the p–n heterojunction is formed, the built-in electric field promotes the transfer of photogenerated electrons and holes to the conduction band of the n-type semiconductor and the valence band of the p-type semiconductor, respectively, which then promotes separation of the photogenerated charge carrier. 19 Liang et al. 20 used an electrodeposition method to deposit Ni into TiO2 nanotubes with a periodic pulsed current. Each period had three steps (200 mA/cm2 for 10 ms, 200 mA/cm2 for 4 ms, and 0 mA/cm2 for 1 s, successively). The result showed that a Schottky barrier was formed between the Ni and TiO2 nanotubes, which had a good effect on suppressing the recombination of photogenerated electron–hole pairs. Therefore, the photocatalytic efficiency of Ni-doped TiO2 nanotubes was found to be significantly higher than that of pure TiO2 nanotubes.
In this work, we have prepared TiO2 nanotubes by anodic oxidation, that is, we used a direct impregnation–calcination method to insert NiO nanoparticles into TiO2 nanotubes. The schematic diagram of the processes was shown in Figure 1. In addition, the effects of the precursor concentration on the micromorphology, elemental composition, crystalline structure, and photocatalytic activity of NiO/TiO2 nanotubes were studied.

A schematic diagram of the processes involved in product formation.
Results and discussion
Micromorphology of NiO–TiO2 NTbs
Figure 2 shows the top-view scanning electron microscope (SEM) images of anodized TiO2 NTb arrays and NiO–TiO2 NTbs prepared with different concentrations of NiCl2·6H2O. Figure 2(a) clearly shows a self-ordered structure, where vertically aligned TiO2 nanotube arrays are formed on the Ti substrate, with an average diameter of 100 nm and a wall thickness of 13 nm. When the concentration of NiCl2·6H2O is relatively low (50–150 mM, Figure 2(b)–(d)), no significant micromorphology changes are observed in the TiO2 nanotubes. Moreover, there is no obvious accumulation of NiO particles; only small fragments formed by cracking of the tube walls were dispersed on the surface of the TiO2 nanotubes. However, when the concentration of NiCl2·6H2O is relatively high (200–300 mM, Figure 2(e) and (f)), a large amount of NiO particles accumulated on the surface of the TiO2 nanotubes. In summary, the precursor concentration has a significant effect on the micromorphology of the TiO2 nanotubes.

SEM top-view images of TiO2 NTbs and NiO–TiO2 NTbs on Ti foil surface prepared with different NiCl2·6H2O concentrations: (a) TiO2 NTbs, (b) 50 mM, (c) 100 mM, (d) 150 mM, (e) 200 mM, and (f) 300 mM.
Figure 3 shows the cross-sectional SEM images of the as-prepared samples. It is apparent that the tube length did not change before or after the NiO deposition and that the average length is 4.2 μm. In addition, after the impregnation and annealing processes, the nanotubular structure is maintained and no significant morphological change is observed. Consequently, when the metal or metal oxide is deposited by the impregnation method, the precursor concentration has a significant effect on the amount of doped product, and large particle agglomerates can be identified at higher precursor concentrations.

SEM cross-sectional images of TiO2 NTbs and NiO–TiO2 NTbs on Ti foil surface prepared with different NiCl2·6H2O concentrations: (a) TiO2 NTbs, (b) 150 mM, and (c) 300 mM.
Figure 4 demonstrates that the NiO–TiO2 NTbs were composed of Ti, O, C, and Ni. The EDS element analysis results were shown in Table 1. The atomic ratio between Ti and O is approximately 1:2. This indicates that the products formed by anodization are TiO2 nanotubes. The carbon is possibly derived from external contamination during testing. There are no N and F elements in the TiO2 nanotubes after annealing. This is because any organic substances adsorbed on the surface of TiO2 nanotubes, along with N and F, are removed effectively via annealing at a high temperature.
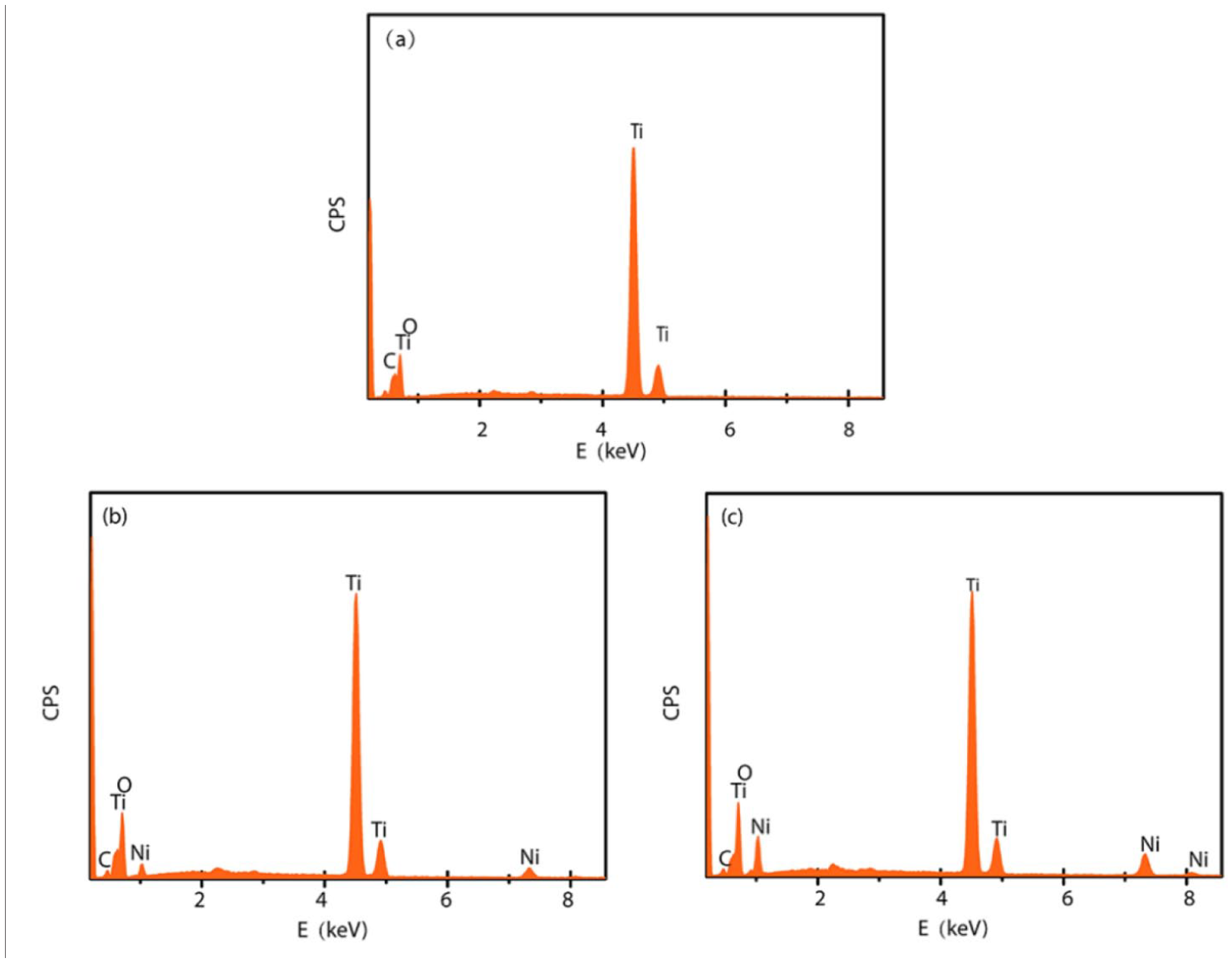
EDS spectra of NiO–TiO2 NTbs prepared using different NiCl2·6H2O concentrations: (a) TiO2 NTbs, (b) 100 mM, and (c) 300 mM.
EDS element analysis results.

EDS: energy-dispersive spectroscopy.
Crystallite structure of NiO–TiO2 NTbs
X-ray diffraction (XRD) analysis was employed to characterize the crystal phase of the NiO–TiO2 composite nanotube arrays (Figure 4). All samples showed diffraction peaks at 25.3°, 47.7°, and 54.8°. These diffraction peaks can be assigned to the (101), (200), and (211) planes of the anatase phase, respectively. 21 This indicates that the crystal phase of TiO2 NTbs transformed from the amorphous phase into anatase only after annealing and that no rutile phase appeared. However, no obvious peak for NiO was observed in the XRD spectra; this is possibly because of the small amount of assembled NiO or poor crystallization. However, after impregnation, the diffraction peaks of anatase and titanium were shifted, which may be due to Ni entering the lattice of TiO2, leading to a change in the lattice size. With the increase in precursor concentration, the magnitude of the diffraction peaks at 25.3°, 47.7°, and 54.8° gradually decreases. This may be due to the NiO particles deposited on the surface of TiO2 nanotubes or the variation of the lattice parameters of TiO2 after modification.
Electron lifetime of NiO–TiO2 NTbs
The open-circuit voltages (Voc) of NiO–TiO2 NTbs prepared using different precursor concentrations were recorded and the results are shown in Figure 5.

Open-circuit voltage decay (OCVD) of NiO–TiO2 NTbs prepared using different precursor concentrations.
The electron lifetime curves of the NiO–TiO2 NTbs prepared at different precursor concentrations were calculated using the results from Figure 5 and equation (1), and the results are shown in Figure 6. Compared to those for pure TiO2 nanotubes, the electron lifetimes of NiO–TiO2 NTbs prepared with precursor concentrations of 50, 100, and 200 mM were improved. This indicates that the recombination rate of photogenerated electron–hole pairs was lower. Specifically, the recombination rate of the sample prepared at 50 mM was the lowest. Hence, it has the highest electron lifetime.

Electronic lifetimes of NiO–TiO2 NTbs prepared using different precursor concentrations.
Photocatalytic activity of NiO–TiO2 NTbs
The absorbance of pure TiO2 nanotubes and NiO–TiO2 nanotubes was tested with a UV-Vis spectrophotometer, and the efficiency of rhodamine B (RhB) degradation was calculated to study the photocatalytic reaction activity of the catalyst. The results are shown in Figures 7 and 8.

Degradation of RhB under UV irradiation by NiO–TiO2 NTbs prepared with different precursor concentrations. Blank test 1 is the degradation curve with only TiO2 nanotubes, and blank test 2 is the degradation curve with UV light only.
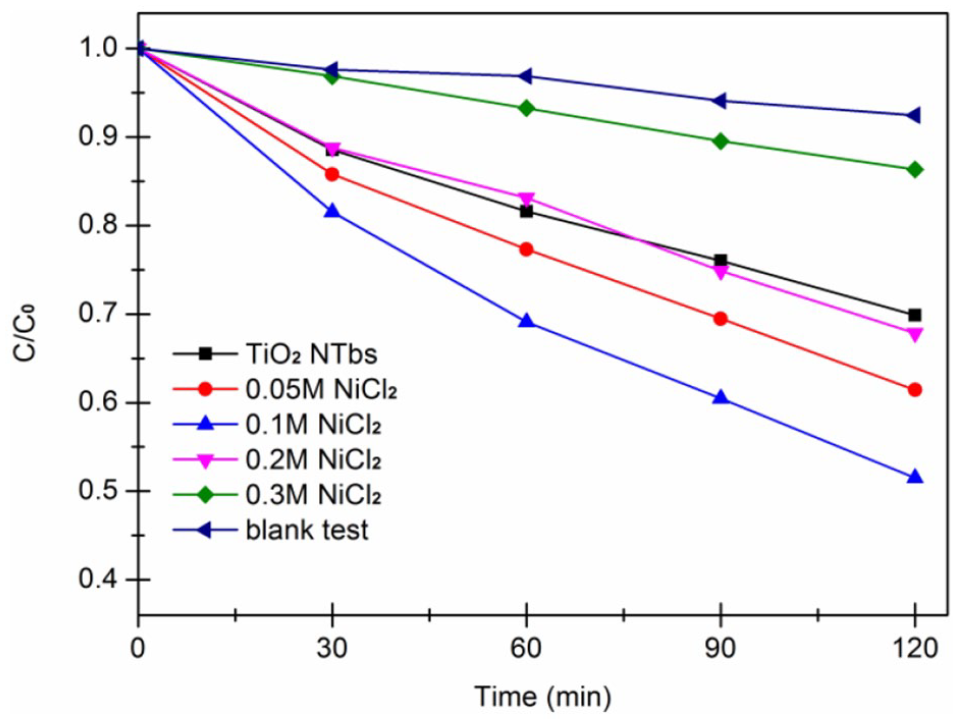
Degradation of RhB under visible light irradiation by NiO–TiO2 NTbs prepared with different precursor concentrations. Blank test is the degradation curve with visible light only.
It is apparent from Figure 7 that the pure TiO2 nanotubes have rather a low photocatalytic activity. When the concentration of the precursors was increased from 50 to 100 mM, the photocatalytic activity of the NiO–TiO2 nanotubes significantly improved. The precursor concentration of 100 mM results in the best degradation effect, possibly due to NiO entering the TiO2 lattice, which promotes carrier separation and improves carrier utilization. When the concentration of precursors was increased, the degradation rate of the NiO–TiO2 NTbs decreased. This can be attributed to the increase of the amount of NiO particles on the surface of TiO2 nanotubes, which affected not only the absorption of photons by NiO–TiO2 NTbs, but also the adsorption of RhB. Figure 8 shows the degradation of RhB by catalysts prepared using different precursor concentrations under visible light. From the above analysis, it can be seen that visible light has a great impact on the photocatalytic effect of composite semiconductors prepared under different precursor concentrations. In summary, compared to the case with pure TiO2 nanotubes, a precursor concentration of 100 mM during impregnation had a positive effect on the photocatalytic activity.
Conclusion
In this work, highly ordered TiO2 nanotubes evenly modified by NiO particles have been successfully prepared by an impregnation deposition method. SEM images show that the precursor concentration has a significant effect on the micromorphology of the TiO2 nanotubes. The surface of the TiO2 nanotubes does not significantly change with relatively low precursor concentrations (50–150 mM). NiO particles accumulated on the surface and filled the pores for relatively high precursor concentrations (200–300 mM). The formed NiO–TiO2 NTbs were mainly composed of Ti, O, and Ni, with trace amounts of C from external contamination during the test. The electron lifetimes of NiO–TiO2 NTbs prepared at different precursor concentrations were improved compared to those of pure TiO2 nanotubes. The catalyst prepared at 50 mM concentration had the highest electron lifetime. XRD results (Figure 9) indicated that the crystal phase of NiO–TiO2 NTbs transformed from an amorphous phase to anatase after annealing. The photocatalytic activity of the NiO–TiO2 NTbs was compared by evaluating the degradation of RhB under UV-Vis light. The best photocatalytic effect was observed for the sample prepared with a precursor concentration of 100 mM.

XRD patterns of TiO2 NTbs and NiO–TiO2 NTbs prepared at different NiCl2·6H2O concentrations. All samples were annealed at 450 °C under a nitrogen atmosphere for 3 h.
The modified nanotubes have improved photocatalytic performance and can be used in environmental protection and energy fields such as organic degradation and photocatalytic hydrogen production, but there are still some shortcomings: (1) most research stays in the laboratory stage, the repeatability is unknown, and the materials have not been applied in practice. (2) Some mechanisms for the preparation of TiO2 nanomaterials remain unclear. (3) It is necessary to further explore green, environmentally friendly, inexpensive, fast, and efficient modification methods.
Experimental
Preparation of TiO2 nanotubes (NTbs)
Titanium foils (0.1 mm × 30 mm × 40 mm, 99.60% purity) were ultrasonically cleaned in acetone and ethanol, and then rinsed with deionized water for 15 min to remove impurities. The foils were then dried under a nitrogen stream before anodization. A two-electrode system was used for the anodic oxidation process. Ti foil was used as the anode and a carbon rod as the cathode. The Ti foil was anodized at 50 V for 30 min in an ethylene glycol/deionized water solution (10 vol% of H2O), citric acid (0.11 mol L−1), and NH4F (0.15 mol L−1). The first anodization process was carried out in a water bath at 60 °C under magnetic stirring. Next, the Ti foil was sonicated in deionized water for 20 min in order to remove the oxide layer. The second anodization process was conducted at room temperature (25 °C) for 2 h, also in a water bath under magnetic stirring. Subsequently, the anodized Ti foil was rinsed in deionized water and dried under a nitrogen stream. The as-prepared TiO2 nanotubes were further annealed at 450 °C before using.
Preparation of NiO–TiO2 NTbs
The prepared TiO2 NTbs were immersed in different concentrations (50, 100, 150, 200, and 300 mM) of a NiCl2·6H2O solution for 20 min at room temperature (25 °C). The samples were then rinsed with deionized water and dried at 60 °C. Finally, the prepared samples were annealed in a muffle furnace under a nitrogen atmosphere at 450 °C for 3 h to convert the amorphous phase into anatase.
Characterization of the NiO–TiO2 NTbs
The morphology and microstructure of the NiO–TiO2 NTbs were studied with a SEM (ZEISS-IGMA, HD/VP, Carl Zeiss). X-ray energy-dispersive spectroscopy (EDS) was used to analyze the element species and content. The accelerating voltage used in the test is 5.0 kV. The electron microscope images at different magnifications were compared, and Rasmus software was used to measure the diameter, wall thickness, tube length, and particle diameter of the catalyst.
The crystal phase structures of the NiO–TiO2 NTbs were determined by XRD (D/max-2200PC, Rigaku, Shimadzu). The test wavelength is λ = 0.15406 nm, the current is 30 mA, the working voltage is 40 kV, and the scanning range is 10–80°. The XRD spectrum is compared with the MDI Jade5 database software to obtain the crystal structure of the sample.
Electrochemical tests were carried out on an electrochemical workstation (CHI604E, Shanghai, China) with a three-electrode arrangement. The prepared NiO–TiO2 NTbs (10 × 10 mm2), a platinum net (20 × 20 mm2), and an Ag/AgCl (3 M KCl) electrode were used as the working electrode, counter electrode, and reference electrode, respectively. Zaban et al. 22 proposed an effective model to study the electron lifetime of semiconductors by analyzing the photo-induced open-circuit voltage decay (OCVD). The photovoltage is recorded under open-circuit photostationary conditions. Once the illumination is interrupted, the photogenerated carriers decay due to recombination, leading to a decrease in the photogenerated open-circuit voltage. The effective electron lifetime is related to the reciprocal of the change of the open-circuit voltage and the time via the following equation

where kB is the Boltzmann constant, T is the absolute temperature, and e is the elementary charge. The electrolyte used in the experiment is 1 mol L−1 Na2SO4 solution. Using a 365-nm UV light source, the OCVD was tested under the conditions of an applied voltage of 0.
The photocatalytic activity of the pure and modified TiO2 nanotubes was tested using a UV-Vis absorption spectrometer (UV1900, Youke, Shanghai). The light source was UV light, and the wavelength range was λ = 400–650 nm. In this experiment, RhB was selected as the target degradation product. The specific experimental steps are as follows.
The catalyst was cut to a size of 1 × 1 cm2, placed upright in a cuvette and irradiated under 500 W UV light for 30 min; the cuvette was 3 cm away from the light source to increase the hydrophilicity of the catalyst surface, and then colorimetric test was performed. RhB solution (2.5 mL with a concentration of 10 µm L−1) was added to the dish, and allowed to stand in a dark environment for 20 min to reach an adsorption–desorption equilibrium. A UV-Vis spectrophotometer within the wavelength range of 400–650 nm was used to test the absorbance of the RhB solution before starting the irradiation process. The photocatalytic degradation was continued, and the absorbance of the RhB solution was recorded after irradiation times of 30, 60, 90, and 120 min, respectively.
Footnotes
Acknowledgements
The authors are also grateful for the measurement assistants from the Analysis & Testing Center of Northeast Petroleum University.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant no. 21905042), the Heilongjiang Youth Innovative Talents Training Project (grant no. UNPYSCT-2018042), and the Cultivating Foundation and Scientific Research Foundation of Northeast Petroleum University (grant no. 2017PYQZL-06).
