Abstract
The synthesis of surface defect TiO2-x nanobelts with excellent visible light absorption is important to reduce costs, increase stability, and improve photocatalytic activity. In this work, we report that nitrogen-doped surface defect TiO2-x nanobelts are synthesized by annealing pristine TiO2 nanobelts in Ar/H2 (95%/5%) atmosphere followed by subsequent heat treatment in NH3 at various temperatures. The aim is to study the effect of the extent of the N-doping amount in the lattice of TiO2-x nanobelts on light absorption and photocatalytic activity. Considering the increase in organic pollutants in wastewater, the photocatalytic activity is measured by degrading rhodamine B (RhB) dye in water. The results demonstrate that the calcination temperature affects the doping level of N, and the b-TiO2-N550 sample exhibits higher photocatalytic performance than that of other samples under visible-light irradiation for the degradation of rhodamine B, which is up to 96.11%. The enhancement is ascribed to the synergistic effect of N-doping and self-doping oxygen vacancy (which extend the visible light absorption) and the separation efficiency of photogenerated carriers, which improves the photocatalytic activity.
Nitrogen-doped surface defect TiO2-x nanobelts are synthesized, and were used to degrade organic pollutants by photocatalytic.
Introduction
TiO2 nanobelts have attracted considerable attention owing to their large surface areas, high stability, one-dimensional structures, and many applications in the photocatalytic degradation of organic pollutants.1–3 However, the band gap of TiO2 limits their photocatalysis efficiency. Extensive efforts have been made to modify TiO2 materials through doping with various elements (e.g. S, B, F, Mn, and Fe), heterocoupling techniques, and high-temperature sintering.4–9 An inherent limitation of these approaches is the low lattice doping level of dopants owing to the limited solubility of substitutional dopants in the bulk. However, excessive amounts of introduced dopants often act as carrier recombination centers, which impair the photocatalytic properties of TiO2 materials. 10 Therefore, we need to develop an appropriate method to achieve a moderate amount of heavy non-metal doping into TiO2. To solve the abovementioned issues, TiO2 nanobelts are heat-treated in NH3 to achieve N-doping of the TiO2 lattice. One approach is to dope TiO2 with N, which results in p-states near the valence band similar to other deep donor levels in semiconductors.11–13 Nitrogen doping has been shown to extend light absorption from the ultraviolet (UV) to the visible light range and to produce possible photocatalytic activity under visible light irritation. However, visible light absorption was not remarkably enhanced. 14
Recently, black TiO2 nanobelts have been reported to improve visible light absorption and have shown excellent photocatalytic activity owing to the introduction of crystal defects in the TiO2-x nanobelts.15–20 Herein, pristine anatase TiO2 nanobelts are synthesized via hydrothermal processing and annealing in an Ar/H2 (95%/5%) atmosphere; then, the sample is heat-treated in NH3 at different sintering temperatures to introduce N-doping into the TiO2 lattice. The doping level of N in the TiO2 lattice increases on increasing the treatment temperature in an NH3 atmosphere. In this study, we prepared a series of black TiO2 nanobelts photocatalysts with N-doping (denoted as b-TiO2-N). The innovation of this study is to replace ordinary TiO2 nanotube arrays with black TiO2 nanotube arrays. The aim of this work is to combine black TiO2 nanotube arrays with oxygen vacancies with N-doping in the TiO2 lattice to maximize the capability of degrading rhodamine B. It is determined that the N-doping modification of black TiO2 nanobelts stirred at 550 °C showed excellent photocatalytic performance.
Results and discussion
X-ray diffraction (XRD) was employed to identify the crystal phase compositions of the samples at room temperature. Figure 1 shows the XRD patterns of the different samples. All diffraction peaks corresponded well with the anatase crystalline phase of TiO2. 21 The XRD patterns showed a broad and decreased anatase (101) peak in b-TiO2-N samples compared with that of black TiO2 nanobelts, which may be due to lattice distortion; this result indicated that nitrogen species were successfully doped into the TiO2 lattice. 22 The size and shape of the NH3-treated samples did not show any clear change. Thus, it is inferred that the order of the crystal lattice of anatase decreased with nitrogen doping. However, there were no peaks related to nitrogen-containing compounds or other titania polymorphs in the XRD patterns. This possibly occurred because the concentration of the dopants was too low to be detected by XRD. 23

XRD patterns of black TiO2 nanobelts and b-TiO2-N samples heated at different temperatures.
To further study the crystal structures, the samples were characterized by Raman spectroscopy, as shown in Figure 2. The Raman spectra of all the samples showed five vibrational modes at approximately 142, 196, 394, 517, and 637 cm−1, which were assigned to the Eg, Eg, B1g, A1g + B1g, and Eg Raman-active modes, respectively, and indicated that anatase was the predominant phase of all the samples. 24 The main anatase peaks of the b-TiO2-N samples were similar to those of black TiO2 nanobelts, which suggested that the low N content did not affect the Raman spectra of anatase. 25 However, compared with black TiO2 nanobelts, the intensity of the N-doped characteristic peak decreased and showed a blue shift in the Raman spectrum, which suggested that N-doping affected the structure. 26 In addition, black TiO2 nanobelts appeared to have a broadened intensity because of oxygen vacancies. 27

Raman spectra of black TiO2 nanobelts and b-TiO2-N samples heated at different temperatures.
As shown in Figure 3(a) and (b), there were no clear changes in the scanning electron microscopy (SEM) images of black TiO2 nanobelts and b-TiO2-N samples. Their composition was identified by energy-dispersive X-ray spectroscopy (EDS; transmission electron microscopy (TEM)-EDS) analysis. The Cu signals were determined to be strong in black TiO2 nanobelts and b-TiO2-N samples owing to the copper grid. However, N elements were observed only in the b-TiO2-N sample, which indicated the incorporation of N atoms into the black TiO2 nanobelt lattice. TEM was used to further characterize the structures of the b-TiO2-N samples. Figure 3(c) shows that b-TiO2-N retains the same nanobelt structure as black TiO2 nanobelts. These nanobelts were 5–30 nm wide and up to tens of micrometers long. Figure 3(d) shows the crystal lattice fringes with the interplanar spacing of 0.35 nm, which correspond to the (1 0 1) crystal facet of anatase TiO2, implying that N-doping did not change the crystal phase of the samples. 28 The b-TiO2-N sample has an approximately few nanometer-thick disordered surface layer owing to the existence of Ti3+ and oxygen vacancies; this layer helps to separate e–h and inhibits rapid recombination, which improves electron transfer and photocatalytic activation. 29

SEM images of (a) black TiO2 nanobelts and (b) TiO2-N; TEM images of (c) TiO2-N and (d) TiO2-N; and EDS of (e) black TiO2 nanobelts and (f) b-TiO2-N.
As shown in Figure 4, the absorption edges of black TiO2 nanobelts are larger than those of b-TiO2-N samples. For black TiO2 nanobelts, the absorption edges are approximately 420 nm, which is attributed to oxygen vacancies in the lattice. 30 However, the b-TiO2-N samples show a considerable absorption range of 420–550 nm, which increased with an increase in the NH3 treatment temperature; this occurred because interstitial N introduced local states near the valence band edge. 31 This absorption becomes more intense with an increase in the NH3 treatment temperature, which may be related to the N dopant content. In addition, these samples appear black owing to the introduction of oxygen vacancies, 32 which affects visible light absorption. Hence, visible light absorbance is higher for the b-TiO2-N samples treated at higher temperature.

UV-Vis diffuse reflectance absorption spectra for black TiO2 nanobelts and b-TiO2-N samples.
The X-ray photoelectron spectroscopy (XPS) analysis of b-TiO2-N550 is used as an example, and the results are shown in Figure 5. The binding energy of the XPS spectra was calibrated with reference to the C 1s peak at 284.8 eV. As shown in Figure 5(a), the photoelectron peaks of Ti, O, N, and C are observed in the XPS survey spectra for the b-TiO2-N550 sample. Clearly, no peaks attributed to impurities are observed, except for the C 1s peak, which may originate from CO2 being adsorbed on the surface of the samples when they are exposed to air or from the XPS instrument. 33 The survey spectra indicates that the obtained sample contains Ti, O, and N, and the relative content of nitrogen is 0.59 at%, which indicates that nitrogen is successfully doped into TiO2. Figure 5(b) shows the compared Ti2p XPS spectrums of b-TiO2-N550 sample and TiO2 standard materials. We can see that the two pictures are exactly the same, which may suggest that this Ti2p spectrum is characteristic of a single Ti4+ environment. This is not so surprising, as XPS looks only at the surface, which may be more oxidized, or the Ti3+ may be below the detection limit of XPS, which is only 0.5 at% or so. The O1s peak in Figure 5(c) can be further divided into two different peaks with binding energies at 529.8 and 531.2 eV, which can be indexed to the O lattice in Ti–O (TiO2) and surface hydroxyl groups, respectively. 34 The atom ratio of lattice oxygen to Ti content was 1.5, lower than 2 of TiO2, indicating the presence of oxygen vacancy. The XPS spectra of N 1s is shown in Figure 5(d) with binding energies located at 395.8 and 399.8 eV. The N 1s peak at 395.8 eV is characteristic of N3− that corresponds to TiN.30,31 The N 1s peak at 395–397 eV obtained from N-doped TiO2 has also been assigned to the Ti-N bonding by other researchers.18,32,33,35 The N 1s peak at ∼399.8 eV is ascribed to Ti-O-N or Ti-N-O oxynitride, and the result was also confirmed by Wang et al. 14 Therefore, the XPS results show that the N/Ti3+ co-doped triphasic TiO2 layer was successfully deposited on the surface of black TiO2 nanobelts during the hydrothermal process.

XPS spectra for the b-TiO2-N550 sample: (a) the full XPS spectrum; the spectra of (b) Ti 2p, (c) O 1s, and (d) N 1s.
To detect oxygen vacancies, electron paramagnetic resonance (EPR) was conducted at room temperature and at low temperature (at 123 K). Figure 6 shows the EPR data obtained at room temperature. Black TiO2 nanobelts and b-TiO2-N550 showed strong signals at

EPR spectra of black TiO2 nanobelts and b-TiO2-N550.
Electrochemical impedance spectroscopy is a useful technique to clarify the origin of the different photoelectrochemical performance of the samples. Z′ and Z″ represent the real and imaginary components of the impedance, respectively. The semicircle diameter corresponds to the charge transfer resistance of the interfaces inside the photoanode and at the photoanode/electrolyte interfaces. As shown in Figure 7(a), under illumination, the arch size of the samples follows the order of black TiO2 nanobelts > b-TiO2-N500 > b-TiO2-N525 > b-TiO2-N575 > b-TiO2-N550. Transient photocurrent responses for black TiO2 nanobelts and b-TiO2-N samples were recorded to evaluate the photogenerated charge separation and electron transfer. As shown in Figure 7(b), the b-TiO2-N550 sample exhibits a considerably enhanced photocurrent density compared with those of black TiO2 nanobelts and other b-TiO2-N samples, and it had the largest photocurrent density up to 0.28 mA cm−2. In this section, a higher photocurrent represents a higher e–h separation efficiency of the composite photocatalyst. Thus, the b-TiO2-N550 sample can efficiently separate the photogenerated carriers.

Comparison of the photoelectrochemical performance of black TiO2 nanobelts and b-TiO2-N electrodes in the electrolyte of a 1-M NaCl aqueous solution: open circuit potential under visible light illumination. (a) Electrochemical impedance spectra of the Nyquist plots under dark conditions. (b) I–t curves.
To evaluate the visible light photocatalytic efficiency of black TiO2 nanobelts and b-TiO2-N samples, the degradation of rhodamine B was carried out. As shown in Figure 8(a), the photocatalytic activity of the samples decreased according to the following order: b-TiO2-N550, b-TiO2-N525, b-TiO2-N575, b-TiO2-N500, and black TiO2 nanobelts. Compared with black TiO2 nanobelts (79.59%), approximately 96.11% of RhB was degraded by b-TiO2-N550 after 200 min. The photocatalytic efficiency of b-TiO2-N550 was much higher than that of black TiO2 nanobelts owing to the introduction of oxygen vacancies and N co-doping. For the b-TiO2-N samples, visible light absorption is promoted from the N 2p levels near the valence band maximum, and the color centers are induced by the oxygen vacancies and Ti3+. Only the introduction of an appropriate amount of defects can help improve the photocatalytic performance. However, the N content and the defects concentration of the b-TiO2-N samples are relevant to the NH3 treatment temperature. The modifications extended visible light absorption, resulting in high photoconversion efficiency, and produced a one-dimensional (1D) structure with a large surface area with more surface active sites, which was beneficial for improving the photocatalytic activity. The stability of a photocatalyst should be considered an important factor in practical applications. Thus, a degradation process recycling experiment was carried out to investigate the stability of the b-TiO2-N550 sample. As shown in Figure 8(b), no deactivation of the photocatalytic degradation of organic contaminates activity occurred after five cycles in 1000 min, which indicates the stability of the b-TiO2-N550 sample.

(a) Photocatalytic activities of RhB over black TiO2 nanobelts and the b-TiO2-N samples under visible light irradiation. (b) Stability of photocatalytic activity for degradation of the b-TiO2-N550 sample.
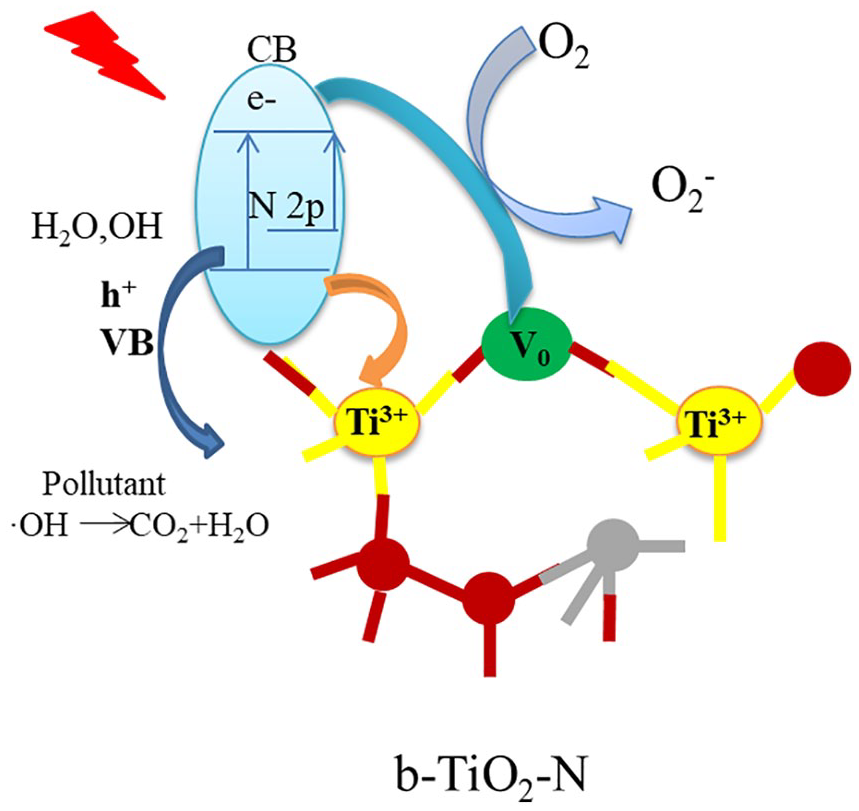
A possible mechanism for the photocatalytic activity for degradation of b-TiO2-N samples is schematically shown in Figure 9. The introduction of oxygen vacancies and nitrogen doping in the lattice of TiO2 modifies the band structure of TiO2. The N2p form impurity states above the valance band (VB), which narrows the band gap energy of TiO2. Accord-ing to literature reports,37,38 Ti4+ is reduced to Ti3+ and oxygen vacancies can significantly prolong the lifetime of photo-electrons and restrain the recombination of photogenerated electrons and holes. The holes of the VB react with hydroxyl groups (OH−) to generate ·OH radicals. The electrons can react with O2 to give O2−, which can further create the reactive hydroxyl radicals. The OH radical is considered a powerful oxidant and is responsible for the decomposition of many pollutants during the photocatalytic process. b-TiO2-N exhibits superior photocatalytic activity owing to the synergistic effects of N and Ti3+ co-doping, which extends the optical absorption to the visible light region and results in enhanced photocatalytic activity.

Schematic diagram of the mechanism of photocatalytic degradation of organic contaminates by b-TiO2-N.
Conclusion
We have successfully synthesized the b-TiO2-N nanobelts. Compared with black TiO2 nanobelts, the b-TiO2-N nanobelt samples exhibited excellent photocatalytic performance. Specifically, b-TiO2-N550 displayed the best photocatalytic properties among the resulting b-TiO2-N nanobelt samples, because of the formation of a considerable amount of oxygen vacancies and N/Ti3+ co-dropping. b-TiO2-N exhibits superior photocatalytic activity owing to the synergistic effect of N and Ti3+ co-dropping. It may have potential applications in many fields such as wastewater treatment, dye degradation, and voc removal.
Materials and methods
Synthesis of b-TiO2-N samples
Pristine TiO2 nanobelts were synthesized via the hydrothermal reaction method. In a conventional synthesis, anatase TiO2 particles (2.0 g, 35 ± 5 nm) were added to 80 mL of a 10 M NaOH aqueous solution. The mixture was magnetically stirred for 2 h and then transferred to a Teflon-lined stainless steel autoclave (100 mL). The autoclave was sealed and placed in a preheated oven to perform the hydrothermal reaction at 150 °C for 24 h. After hydrothermal processing, a white precipitate was collected by filtration and then washed with deionized water and 0.1 M HCl. When the washing solution had a pH <7, a white fluffy powder was obtained. Thus, H2Ti3O7 nanobelts were produced. The product was dried in an oven overnight at 80 °C. Black TiO2 nanobelts were prepared by annealing H2Ti3O7 nanobelts at 450 °C for 2 h in an Ar/H2 (95%/5%) atmosphere. Subsequently, the black TiO2 nanobelts were heat-treated in an NH3 flow (80 mL min−1) in a tubular furnace equipped with a gas flow controller. Four b-TiO2-N samples were obtained by NH3 treatment at different temperatures ranging from 500 °C to 575 °C for 3 h at a ramp rate of 10 °C min−1, and the four samples are named as b-TiO2-N500, b-TiO2-N525, b-TiO2-N550, and b-TiO2-N575, respectively. A pristine sample was also annealed at 450 °C under ambient air for comparison.
Characterization of b-TiO2-N samples
The morphology of the samples was investigated with field-emission scanning electron microscopy (FE-SEM, S-4800, Japan) and transmission electron microscopy (TEM, JEM-2100F, Japan). The crystal structures of the samples were determined by powder XRD (XRD Bruker D8-Focus diffractometer with monochromatized CuKα radiation, λ = 1.5418 Å, Germany). The UV-Vis diffuse reflectance spectra were measured on a UV-Vis spectrophotometer (UV-2700, Shimadzu, Japan) and converted into absorption spectra via the Kubelka–Munk transformation. The Raman spectra of the samples were measured on a confocal system with an excitation wavelength of 530 nm (Bruker, RENISHAW). XPS spectra were collected using a Kratos Axis Ultra.
Photoelectrochemical tests and photocatalytic evaluation
The photoelectrochemical properties of the samples were determined using a CHI660E (Shanghai, Chenhua, China) electrochemical workstation with a three-electrode system (working electrode: the samples were deposited on FTO; counter electrode: Pt wire; reference electrode: Ag/AgCl). A total of 40 mL of 1 mol L−1 NaCl was used as the supporting electrolyte, and the illumination source was 300 W xenon lamp equipped with a simulated solar light filter AM 1.5 (WXS-80C-3 AM 1.5G, 100 mW cm−2). High purity nitrogen was purged to remove dissolved oxygen from the system approximately 1 h before testing.
The photocatalytic activity of the photocatalysts was evaluated by investigating the degradation performance of rhodamine B (RhB) under the visible light irradiation. The solar light source was a 300 W Xe lamp (HSX-F/Uv300, NBeT). First, 0.03 g of the as-prepared photocatalyst was added into 30 mL of 10 mg L−1 dye solution to obtain the reaction suspension. Then, the mixture was magnetically stirred in the dark for 0.5 h to achieve an adsorption-desorption equilibrium. Finally, the mixture was illuminated with visible light under magnetic stirring. At given time intervals, 5 mL of the suspension was collected and centrifuged to obtain a clear solution. The filtrates were analyzed using a Shimadzu Model UV2550 spectrophotometer at the characteristic wavelength of RhB 553 nm.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by State Key Research and Development Plan Funding of China (2017YFB0602500) and Fundamental Research Grant Scheme (CF9300190032) by National Institute of Clean-and-Low-Carbon Energy, China.
