Abstract
Six pyrophosphate analogues are prepared from zerumbone, murrayafoline A, acridone, and 4-hydroxycoumarin via 1,3-dipolar cycloaddition reactions. Their in vitro cytotoxic activity is evaluated against HepG2, LU-1, and HeLa cancer cell lines. Among them, diisopropyl ((ethoxy((4-((1-methoxy-3-methyl-9H-carbazol-9-yl)methyl)-1H-1,2,3-triazol-1-yl)methyl)phosphoryl)methyl)phosphonate (

Introduction
The pyrophosphate group plays an important role in the biochemistry of living organisms. 1 A number of pyrophosphate analogues have been used as drugs for the treatment of osteoporosis, 2 for example, alendronate, risedronate, ibandronate, and zoledronate, and as human farnesyl pyrophosphate synthase inhibitors.3,4
Pyrophosphates and their analogues often have a high negative charge and subsequent poor bioavailability. 5 Because of their high charge at physiological pH, pyrophosphates and their analogues can have difficulty diffusing across cell membranes. 6 To remedy this, pyrophosphonates have been considered as replacements for pyrophosphates so enabling a problematic molecule to overcome a biological obstacle. 7 In addition, several studies have shown that the potencies of pyrophosphate analogues can be increased by administering them as prodrugs in which negative charges on the phosphate moieties are masked.7,8 Pyrophosphonate prodrugs such as tetrakis-pivaloyloxymethyl 2-(thiazole-2-ylamino)ethylidene-1,1-bisphosphonate 8 and tetrakis-pivaloyloxymethyl 2-(3,4-dibromophenyl)ethylidene1,1-bisphosphonate 9 have been found to potently inhibit the in vitro growth of a variety of tumor cell lines (Figure 1). A number of pyrophosphonate prodrugs, such as SRP3042, 10 also have the ability to accelerate the degradation of HMG-CoA reductase, which is a rate-limiting enzyme in the cholesterol biosynthetic pathway, and reduce serum cholesterol level (Figure 1).

Example of pyrophosphate analogues.
Specifically, pyrophosphonate analogues of nucleosides such as AOPCP and AB680 (Figure 1) express strong anti-cancer activity through inhibition of the enzyme CD7311–13 or ecto-5’-nucleotidase, an enzyme that is able to catabolize 5’-adenosine monophosphate (AMP) into adenosine at the extracellular level. The overexpression of CD73 has been linked to many diseases including breast cancer, bladder cancer, ovarian cancer, and melanoma. 14 Therefore, the CD73 enzyme is a potential target for cancer prevention and treatment.
In our previous report, a series of 1,2,3-triazoles containing a bisphosphonate unit (P-C-P-C) was prepared and their CD73 inhibitory and cytotoxic activities were documented. 5 In addition, the non-adenosine bisphosphonates (Figure 1) were reported to inhibit the growth of human cancer cell lines. These findings encouraged us to investigate the cytotoxicity of new non-adenosine pyrophosphate analogues. Therefore, in this paper, several novel pyrophosphonate analogues of murrayafoline A, azazerumbone, acridone, and 4-hydroxycoumarin are synthesized and evaluated for their cytotoxic activity against human carcinoma cell lines: HepG2, LU-1, and HeLa.
Results and discussion
The synthesis was initiated with acridone, 4-hydroxycoumarin, and two bioactive natural compounds, murrayafoline A and zerumbone, which can be easily isolated as the major active ingredients from the rhizomes of Glycosmis stenocarpa
15
and Zingiber zerumbet,
16
respectively. Compounds
First, the key intermediate diisopropyl {[(azidomethyl)(ethoxy)phosphoryl]methyl}phosphonate (

Synthesis of the key intermediate
In the next step, the terminal alkynes
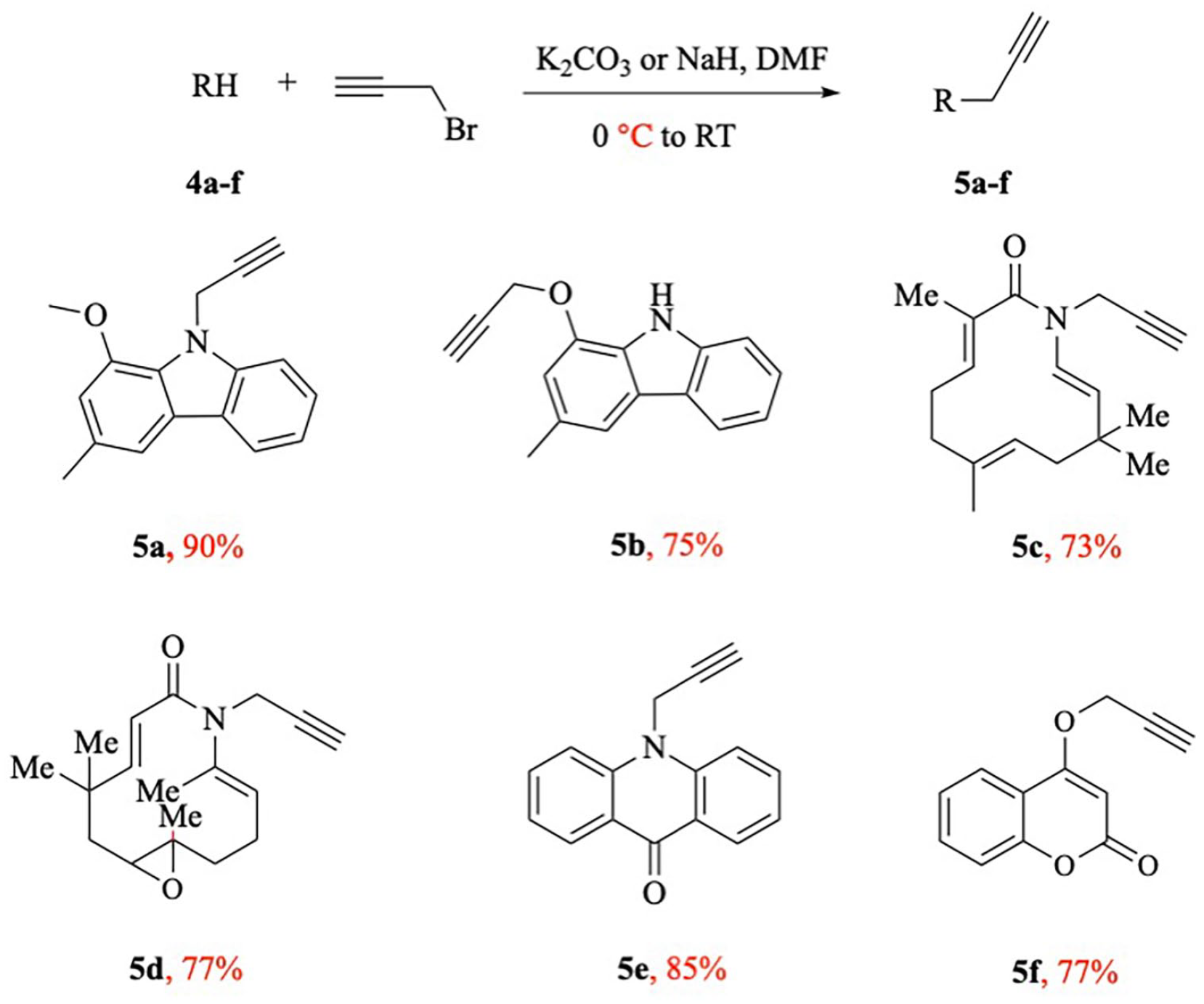
Synthesis of terminal alkynes
In the final step, a 1,3-dipolar cycloaddition reaction was used to couple the intermediate diisopropyl {[(azidomethyl)(ethoxy)phosphoryl]methyl}phosphonate (
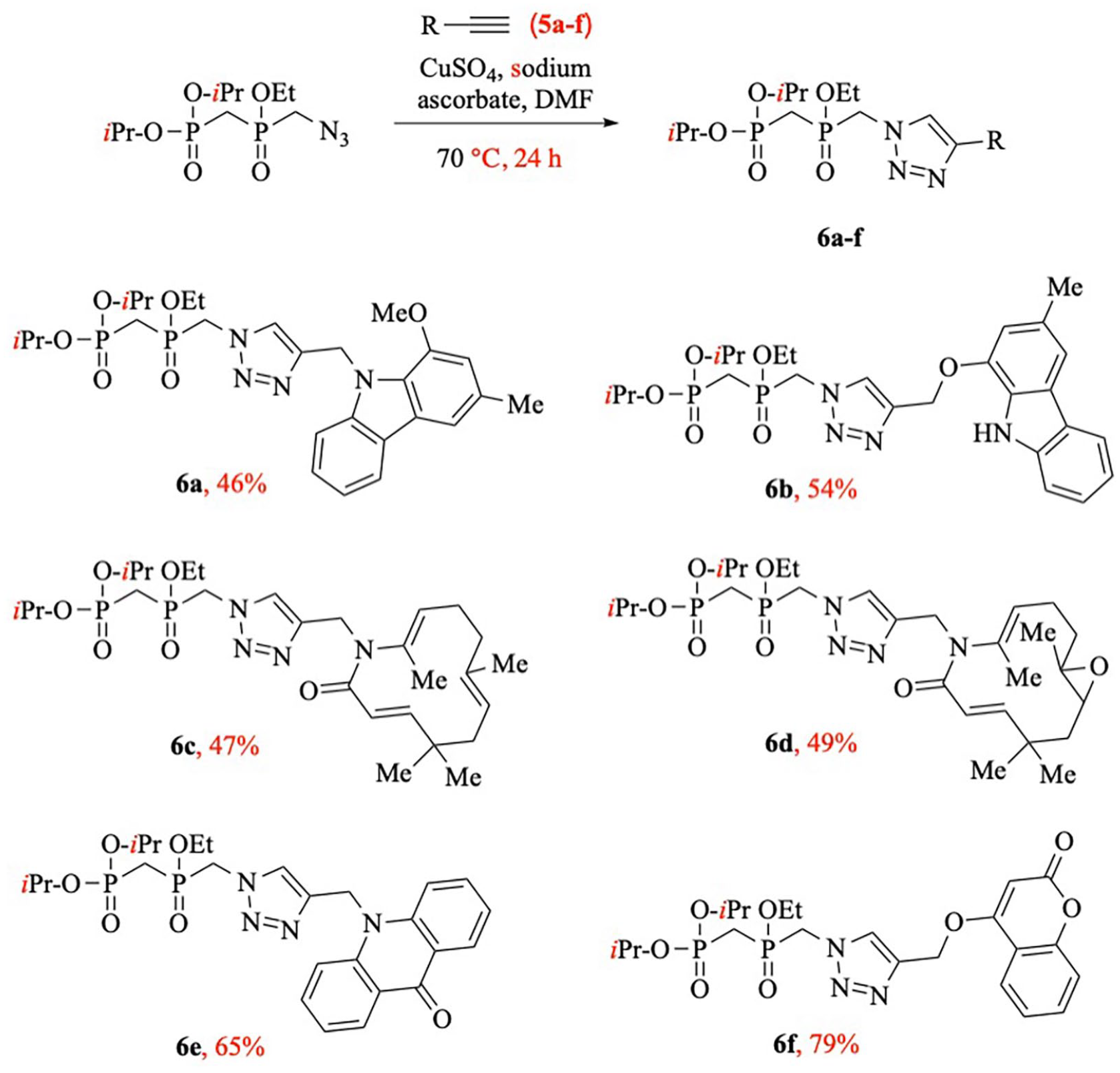
Synthesis of pyrophosphate analogues
All the synthesized compounds
Cytotoxic activity of compounds

As shown in Table 1, pyrophosphonates
The reference substance, ellipticine, exhibited cytotoxic activity against human cancer cells, including HepG2 (ATCC-HB-8065), LU-1 (ATCC-HTB-57), and HeLa (ATCC-CCL-2) cells with IC50 value of 0.35, 0.30, and 0.16 μM, respectively. The values shown for these compounds are the average of three determinations.
Conclusion
In this study, a five-step procedure was used to synthesize six pyrophosphonates
Experimental
General
1H NMR and 13C NMR spectra were recorded at ambient temperature on a Bruker Avance 500 MHz spectrometer in CDCl3. Chemical shifts δ are quoted in parts per million (ppm) referenced to the residual solvent peak (CDCl3 at 7.26 and 77 ppm) relative to TMS. Mass spectra were recorded using an Agilent LC/MSD Trap SL. Thin-layer chromatography was performed on precoated Silica Gel 60 F254 aluminum sheets (Merck), and products were visualized under a UV lamp at 254 nm. Column chromatography was carried out on Silica Gel (40–230 mesh). The terminal alkynes
Synthesis of alkynes 5a–f
Synthesis of intermediates: 1-methoxy-3-methyl-9-(prop-2-yn-1-yl)-9H-carbazole (
General procedure for the synthesis of compounds 6a–f
To a solution of the intermediate
Diisopropyl ((ethoxy((4-((1-methoxy-3-methyl-9H-carbazol-9-yl)methyl)-1H-1,2,3-triazol-1-yl)methyl)phosphoryl)methyl)phosphonate (6a ):
Compound
Diisopropyl ((ethoxy((4-(((3-methyl-9H-carbazol-1-yl)oxy)methyl)-1H-1,2,3-triazol-1-yl)methyl)phosphoryl)methyl)phosphonate (6b ):
Compound
Diisopropyl ((ethoxy((4-(((3E,7Z,11E)-5,5,8,12-tetramethyl-2-oxoazacyclododeca-3,7,11-trien-1-yl)methyl)-1H-1,2,3-triazol-1-yl)methyl)phosphoryl)methyl)phosphonate (6c ):
Compound
Diisopropyl ((ethoxy((4-(((4E,8E)-1,5,10,10-tetramethyl-7-oxo-13-oxa-6-azabicyclo[10.1.0]trideca-4,8-dien-6-yl)methyl)-1H-1,2,3-triazol-1-yl)methyl)phosphoryl)methyl)phosphonate (6d ):
Compound
Diisopropyl ((ethoxy((4-((9-oxoacridin-10(9H)-yl)methyl)-1H-1,2,3-triazol-1-yl)methyl)phosphoryl)methyl)phosphonate (6e ):
Compound
Diisopropyl ((ethoxy((4-(((2-oxo-2H-chromen-4-yl)oxy)methyl)-1H-1,2,3-triazol-1-yl)methyl)phosphoryl)methyl)phosphonate (6f ):
Compound
Cytotoxic evaluation
Stock solutions of the pyrophosphate analogues
Supplemental Material
sj-pdf-1-chl-10.1177_17475198211043439 – Supplemental material for Synthesis of pyrophosphate analogues and their cytotoxic activities
Supplemental material, sj-pdf-1-chl-10.1177_17475198211043439 for Synthesis of pyrophosphate analogues and their cytotoxic activities by Van-Tai Nguyen, Minh-Quan Pham, Thi-Ha Vu, Thi-Hong-Ha Tran, Duy-Tien Doan, Dinh-Luyen Nguyen, Phong Le and Van-Chinh Luu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institute does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was financially supported by the Vietnam Graduate University of Science and Technology under grant no. GUST.STS.DT2017-HH17.
Human and animal rights
This article does not contain any studies with human or animal subjects.
Informed consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
