Abstract
Sixteen conjugates of dihydroartemisinin and zerumbone with 2-mercapto-1,3,4-oxadiazoles were synthesized and structurally elucidated by 1D NMR, 2D NMR, and HRMS spectra. The cytotoxic screening results showed that all the conjugates of dihydroartemisinin with 2-mercapto-1,3,4-oxadiazoles (

Introduction
Oxadiazole rings constitute an important group of heterocycles. In general, oxadiazoles are synthetic and rarely found in nature, except for three oxadiazoles: phidiniadine A (

The chemical structure of oxadiazole isomers, natural oxadiazoles, and some oxadiazole drugs.
Artemisinin (
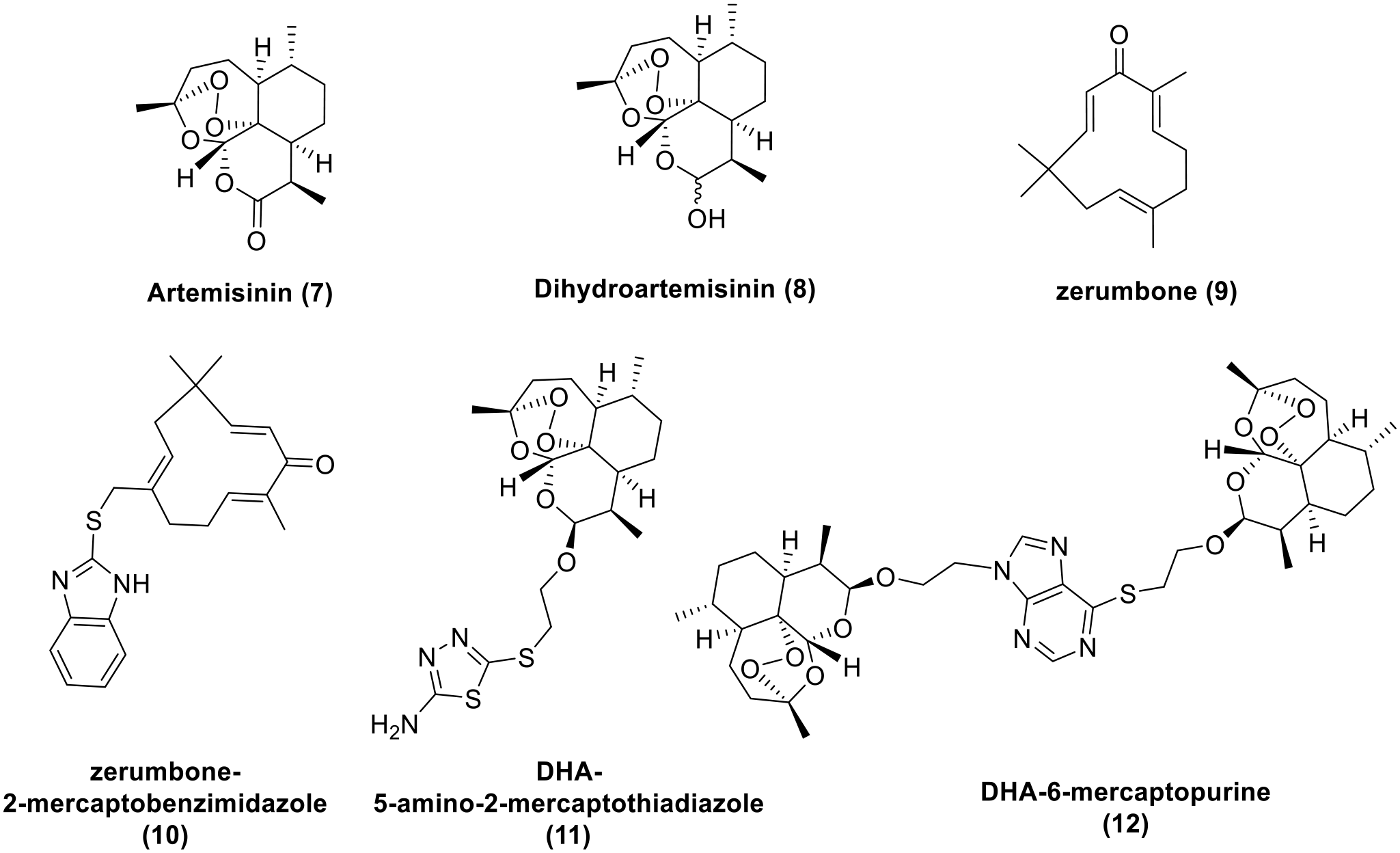
The chemical structures of artemisinin
Another bioactive center motif is the natural pentadienone moiety found in zerumbone (
Nowadays, discovering particular and efficient chemotherapy drugs is a task given to medicinal chemists. These medications work by interacting with certain molecular targets to limit the growth of cancer cells, severely harming the malignant cells in the process. However, various issues, including poor delivery, selectivity, and/or resistance, may slow down the medication development process. Understanding the cellular and molecular pathways that contribute to the process of cancer development and/or spread is, therefore, of significant interest. 37
Protein kinases (PKs) constitute crucial protein families in the process of diverse disease propagation, such as cancer, diabetes, and/or inflammation. 38 Therefore, they constitute a very promising target for new drug discovery due to their prominent roles in several cellular functions such as apoptosis, cell cycle, DNA damage/repair, and metabolism. 39 The overexpression of EGFR (epidermal growth factor receptor) causes a variety of cancer types, including breast, colon, and ovarian subtypes. EGFR is a transmembrane PK receptor that is responsible for cell proliferation and/or apoptosis through different signal transduction pathways. 40 It was recorded to be involved in the process of angiogenesis which increases the proliferation of tumor cells, invasiveness, and metastasis. EGFR inhibitors (e.g. erlotinib) 41 are one of the most important FDA-approved drugs for cancer treatment. Our initial in silico study shows that 2-mercapto-1,3,4-oxadiazole conjugates with zerumbone and DHA have better binding affinities to EGFR receptors than erlotinib due to the direct interactions of crucial amino acids in the active site of EGFR with the oxazole ring and other moieties of these conjugates.
As mentioned earlier, the strategy of building structural blocks based on 1,3,4-oxadiazole and these sesquiterpenes is reasonable. As a continuous result of our recent studies42–44 on the synthesis and biological activities of dihydroartemisinin (DHA) and zerumbone conjugates with the thiols, in the present work, sixteen conjugates of 2-mercapto-1,3,4-oxadiazoles with DHA and zerumbone via thioether linkage have been designed, synthesized, and evaluated for cytotoxic activity against HepG2, LU-1 cell lines, as well as in silico inhibitory activity against EGFR tyrosine kinase.
Results and discussion
Chemistry
The conjugates

Synthesis of DHA and zerumbone conjugates:
In the next step, 5-(hetero)aryl-2-mercapto-1,3,4-oxadiazoles
In the final step, 2-mercapto-1,3,4-oxadiazoles
The structures of
Biology
As shown in Table 1, the conjugates
In vitro cytotoxic activity of the synthesized conjugates.

Data represent the mean ± standard deviation of three independent wells.
In a series
Molecular docking study
The EGFR protein is found on the surface of some normal cells (including cancer cells) and is involved in the processes of cell development and division. Blocking the EGFR pathway can control the growth of cancer cells, and so EGFR inhibitors are used in cancer treatment. Recent studies have shown that tyrosine kinase inhibitors (TKIs) associated with mutations in EGFR are used to treat initial lung cancer.
49
The three-dimensional crystal structure of the inactive EGFR tyrosine kinase domain (PDB ID: 4HJO, resolution: 2.75 Å) was obtained from the RCSB protein data bank and was used for molecular docking and in silico analysis. Before virtual screening was conducted, the initial crystallographic ligand was re-docked into the active site of the EGFR protein with an RMSD value <2 Å (Figure 3) to show that the docking protocol was suitable for this study. The crystallographic ligand, Erlotinib, is a tyrosine kinase inhibitor known to exhibit activity by binding to the active conformation of the EGFR-TKD (tyrosine kinase domain) used to block EGFR (epidermal growth factor receptor) signaling in cancer. Therefore, this compound was chosen as the reference compound for the current study. Next, two series of compounds were subjected to molecular docking to evaluate their effects on the EGFR target. The binding energies, the number of hydrogen bonds, and the residues involved in the hydrogen bonding of sixteen compounds (

Plot illustrating a 3D representation of the superimposition of the co-crystallized (green) and the re-docked (yellow) Erlotinib in the active site of EGFR protein (PDB ID: 4HJO) with an RMSD value of 1.45 Å.
Binding energies, number of hydrogen bonds, and residues involved in hydrogen bonding of compounds (
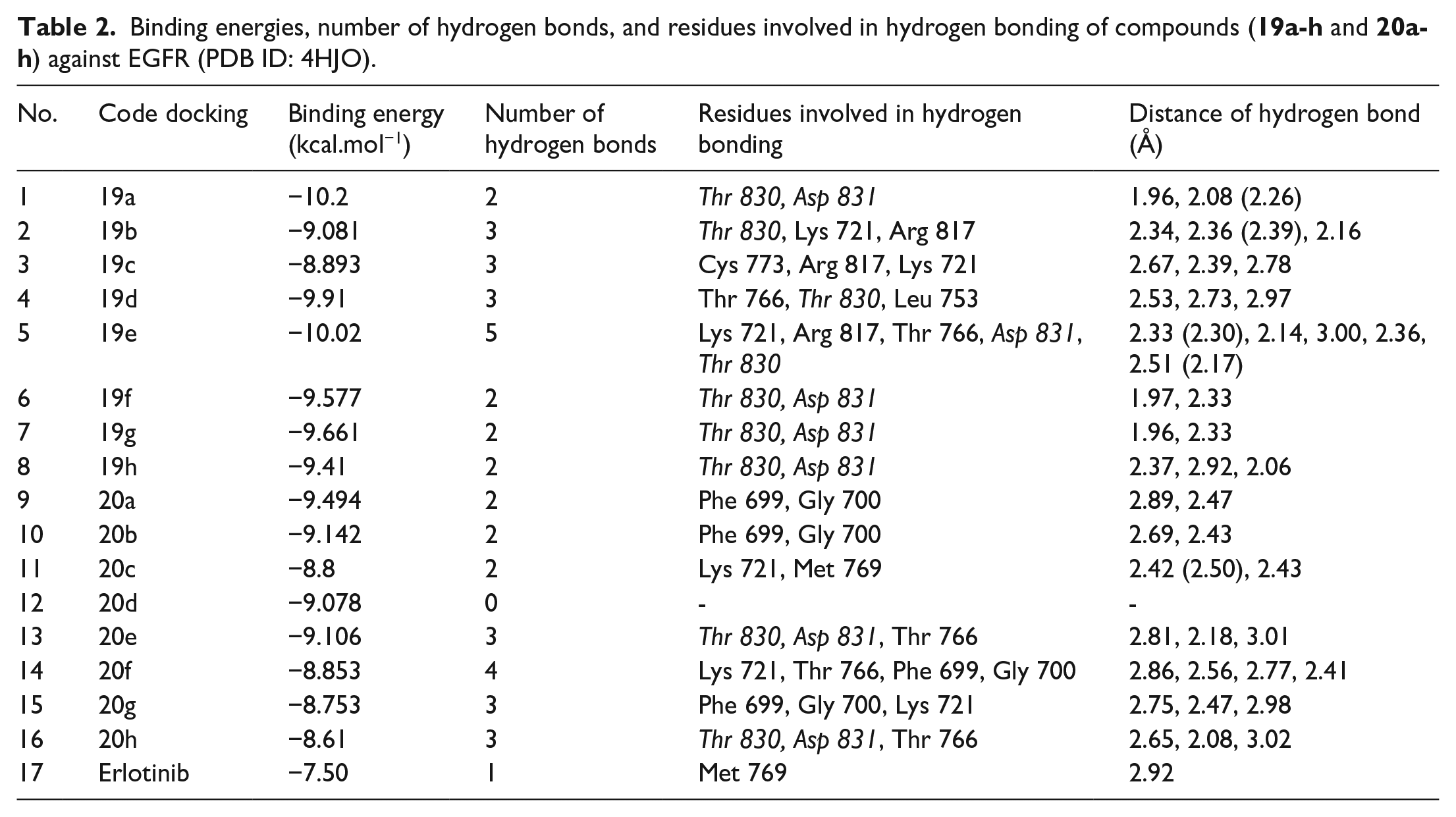
The details of the interactions presented in Table 2 and Figure 4 indicate that both tested compound series exhibit better interactions with EGFR than Erlotinib. Figure 4 shows the best fit of
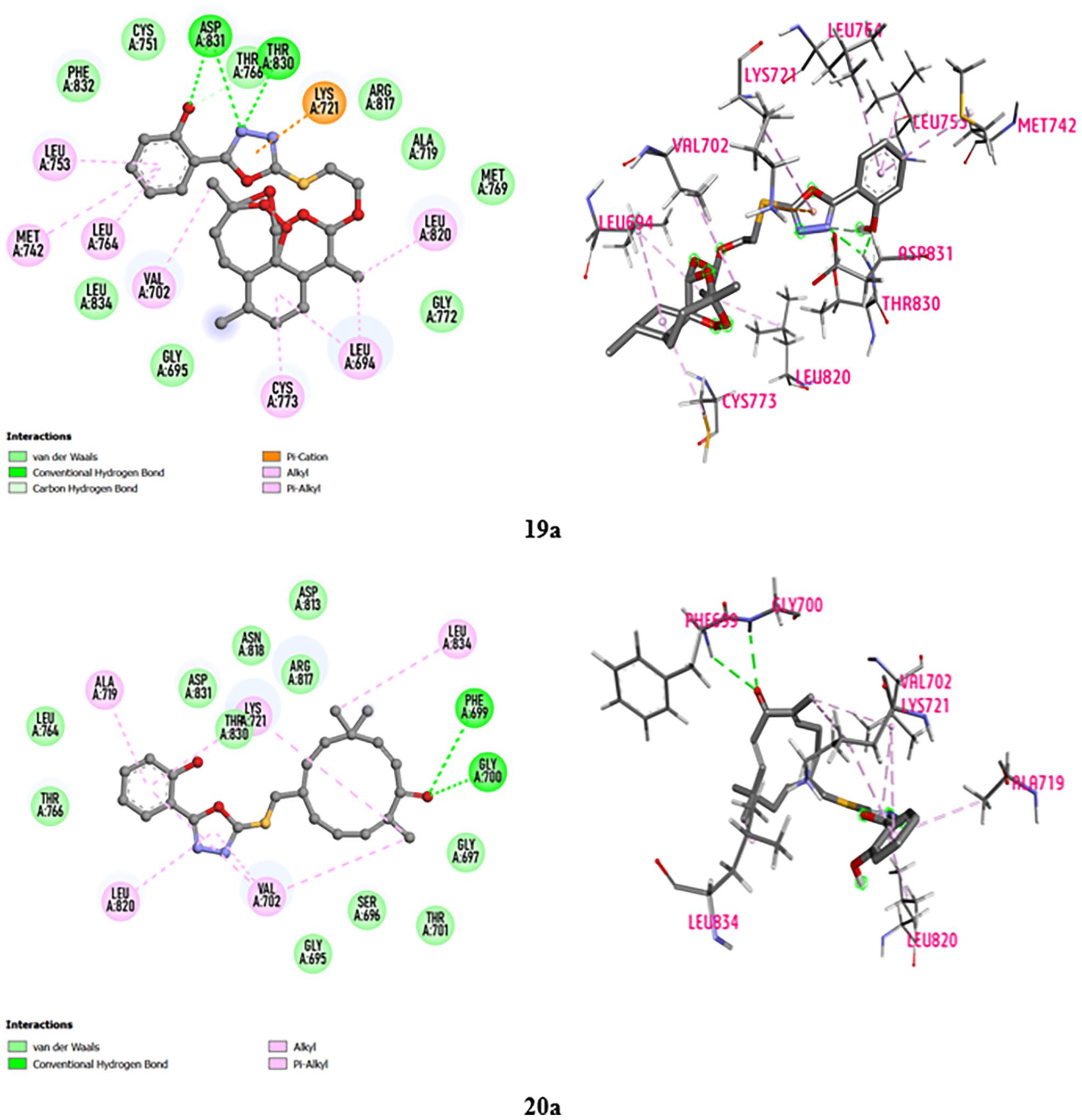
2D (the left) & 3D (the right) views of the top-leads
Conclusion
Sixteen novel conjugates
In addition, the subsequent docking studies revealed that conjugates
Experimental
Chemistry
While dihydroartemisinin is commercially available in Vietnam, zerumbone is isolated from the rhizome of Zingiber zerumbet according to a protocol described by Akhtar et al. 54 Other chemicals were purchased from Sigma Aldrich (Singapore) and used without further purification. 1H NMR and 13C NMR spectra were recorded at the ambient temperature on a Bruker Avance 600 MHz spectrometer (Biospin, Germany) in DMSO-d6. Chemical shifts δ are quoted in parts per million (ppm) referenced to the residual solvent peak, (DMSO at 2.50, 3.32 ppm, and 39.5 ppm) relative to TMS. Mass spectra were recorded using an Agilent LC/MSD Trap SL. Thin-layer chromatography was performed on precoated Silica Gel 60 F254 aluminum sheets (Merck, Germany), and products were visualized under a UV lamp at 254 nm. Column chromatography was carried out on silica gel (40-230 mesh).
2-mercapto-1,3,4-oxadiazoles
The 2-(bromo)ethyl ether of DHA
General procedure for the synthesis of conjugates 19a-h, and 20a-h
To a stirred solution of each 2-mercapto-1,3,4-oxadiazole (
2-(5-((2-(((3R,5aS,6R,8aS,9R,10S,12R,12aR)-3,6,9-trimethyldecahydro-12H-3,12-epoxy[1,2]dioxepino[4,3-i]isochromen-10-yl)oxy)ethyl)thio)-1,3,4-oxadiazol-2-yl)phenol (19a)
Yield 73%, white powder, m.p. 120-121 °C; 1
2-(4-methoxyphenyl)-5-((2-(((3R,5aS,6R,8aS,9R,10S,12R,12aR)-3,6,9-trimethyldecahydro-12H-3,12-epoxy[1,2]dioxepino[4,3-i]isochromen-10-yl)oxy)ethyl)thio)-1,3,4-oxadiazole (19b)
Yield 79%, white powder, m.p. 135-136 °C; 1
2-((2-(((3R,5aR,8aS,9R,10S,12R,12aR)-3,9-dimethyldecahydro-12H-3,12-epoxy[1,2]dioxepino[4,3-i]isochromen-10-yl)oxy)ethyl)thio)-5-(3,4,5-trimethoxyphenyl)-1,3,4-oxadiazole (19c)
Yield 78%, colorless oil; 1
2-(4-methoxybenzyl)-5-((2-(((3R,5aS,6R,8aS,9R,10S,12R,12aR)-3,6,9-trimethyldecahydro-12H-3,12-epoxy[1,2]dioxepino[4,3-i]isochromen-10-yl)oxy)ethyl)thio)-1,3,4-oxadiazole (19d)
Yield 74%, colorless oil; 1
2-(pyridin-2-yl)-5-((2-(((3R,5aS,6R,8aS,9R,10S,12R,12aR)-3,6,9-trimethyldecahydro-12H-3,12-epoxy[1,2]dioxepino[4,3-i]isochromen-10-yl)oxy)ethyl)thio)-1,3,4-oxadiazole (19e)
Yield 70%, white powder, m.p. 121-122 °C; 1
2-(pyridin-3-yl)-5-((2-(((3R,5aS,6R,8aS,9R,10S,12R,12aR)-3,6,9-trimethyldecahydro-12H-3,12-epoxy[1,2]dioxepino[4,3-i]isochromen-10-yl)oxy)ethyl)thio)-1,3,4-oxadiazole (19f)
Yield 71%, white powder, m.p. 130-131 °C; 1
2-(pyridin-4-yl)-5-((2-(((3R,5aS,6R,8aS,9R,10S,12R,12aR)-3,6,9-trimethyldecahydro-12H-3,12-epoxy[1,2]dioxepino[4,3-i]isochromen-10-yl)oxy)ethyl)thio)-1,3,4-oxadiazole (19g)
Yield 68%, white powder, m.p. 123-125 °C; 1
2-(furan-2-yl)-5-((2-(((3R,5aS,6R,8aS,9R,10S,12R,12aR)-3,6,9-trimethyldecahydro-12H-3,12-epoxy[1,2]dioxepino[4,3-i]isochromen-10-yl)oxy)ethyl)thio)-1,3,4-oxadiazole (19h)
Yield 63%, colorless oil; 1
(2E,6Z,10E)-6-(((5-(2-hydroxyphenyl)-1,3,4-oxadiazol-2-yl)thio)methyl)-2,9,9-trimethylcycloundeca-2,6,10-trien-1-one (20a)
Yield 61%, white powder, m.p. 129-130 °C; 1
(2E,6Z,10E)-6-(((5-(4-methoxyphenyl)-1,3,4-oxadiazol-2-yl)thio)methyl)-2,9,9-trimethylcycloundeca-2,6,10-trien-1-one (20b)
Yield 66%, colorless oil; 1
(2E,6Z,10E)-2,9,9-trimethyl-6-(((5-(3,4,5-trimethoxyphenyl)-1,3,4-oxadiazol-2-yl)thio)methyl)cycloundeca-2,6,10-trien-1-one (20c)
Yield 68%, white powder, m.p. 72-73 °C; 1
(2E,6Z,10E)-6-(((5-(4-methoxybenzyl)-1,3,4-oxadiazol-2-yl)thio)methyl)-2,9,9-trimethylcycloundeca-2,6,10-trien-1-one (20d)
Yield 60%, colorless oil; 1
(2E,6Z,10E)-2,9,9-trimethyl-6-(((5-(pyridin-2-yl)-1,3,4-oxadiazol-2-yl)thio)methyl)cycloundeca-2,6,10-trien-1-one (20e)
Yield 54%, white powder, m.p. 75-76 °C; 1
(2E,6Z,10E)-2,9,9-trimethyl-6-(((5-(pyridin-3-yl)-1,3,4-oxadiazol-2-yl)thio)methyl)cycloundeca-2,6,10-trien-1-one (20f)
Yield 54%, colorless oil; 1
(2E,6Z,10E)-2,9,9-trimethyl-6-(((5-(pyridin-4-yl)-1,3,4-oxadiazol-2-yl)thio)methyl)cycloundeca-2,6,10-trien-1-one (20g)
Yield 58%, white powder, m.p. 90-91 °C; 1
(2E,6Z,10E)-6-(((5-(furan-2-yl)-1,3,4-oxadiazol-2-yl)thio)methyl)-2,9,9-trimethylcycloundeca-2,6,10-trien-1-one (20h)
Yield 51%, colorless oil; 1
Biology
The in vitro cytotoxic evaluation against HepG2 and LU-1 cancer cell lines was carried out according to the described protocols.55,56 Accordingly, stock solutions of the conjugates
Molecular docking study
Molecular docking is one of the most effective computational approaches for drug development to determine the efficacy of investigated drugs. The interactions of novel 2-mercapto-1,3,4-oxadiazole-dihydroartemisinin (
Supplemental Material
sj-pdf-1-chl-10.1177_17475198231199428 – Supplemental material for Synthesis and cytotoxic activity evaluation of novel dihydroartemisinin and zerumbone conjugates with 2-mercapto-1,3,4-oxadiazoles as potential EGFR inhibitors
Supplemental material, sj-pdf-1-chl-10.1177_17475198231199428 for Synthesis and cytotoxic activity evaluation of novel dihydroartemisinin and zerumbone conjugates with 2-mercapto-1,3,4-oxadiazoles as potential EGFR inhibitors by Duc Quan Tran, Ngoc Hung Truong, Thi Hoang Anh Nguyen, Thi Thuy Trinh, Thi Cham Ba, Thi Thuy Linh Nguyen, Xuan Ha Nguyen, Manh Cuong Nguyen and Van Chinh Luu in Journal of Chemical Research
Supplemental Material
sj-pdf-2-chl-10.1177_17475198231199428 – Supplemental material for Synthesis and cytotoxic activity evaluation of novel dihydroartemisinin and zerumbone conjugates with 2-mercapto-1,3,4-oxadiazoles as potential EGFR inhibitors
Supplemental material, sj-pdf-2-chl-10.1177_17475198231199428 for Synthesis and cytotoxic activity evaluation of novel dihydroartemisinin and zerumbone conjugates with 2-mercapto-1,3,4-oxadiazoles as potential EGFR inhibitors by Duc Quan Tran, Ngoc Hung Truong, Thi Hoang Anh Nguyen, Thi Thuy Trinh, Thi Cham Ba, Thi Thuy Linh Nguyen, Xuan Ha Nguyen, Manh Cuong Nguyen and Van Chinh Luu in Journal of Chemical Research
Footnotes
Acknowledgements
We thank Bachelor student, Van Tu Ngo for his help in the preparation of the references and the supplemental materials.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Vietnam Academy of Science and Technology under the project with the code: KHCBHH.02/ 21-23.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
