Abstract
The isonicotinic acid containing porphyrin compounds 5-{4-[3-(4-pyridylcarbonyloxy)propoxy]phenyl}-10,15,20-triphenylporphyrin, 5-{4-[3-(4-pyridylcarbonyloxy)propoxy]phenyl}-10,15,20-tri(4-methoxyphenyl)porphyrin, and 5-{4-[3-(4-pyridylcarbonyloxy)propoxy]phenyl}-10,15,20-tri(4-chlorophenyl)porphyrin are prepared and characterized by 1H NMR, 13C NMR, MS, elemental analysis, IR, and UV-Vis. In addition, their spectroscopic properties are investigated through using the Raman spectroscopy, fluorescence, and surface photovoltage measurements. The influence of different substituents on Raman spectra is small, but their impact on the fluorescence spectra and surface photovoltage measurements is significant. Molecular dynamic simulations and UV-Vis diffuse-reflectance spectra show that these porphyrin compounds are potential semiconductor materials.

Introduction
In recent years, porphyrins have received considerable attention because of their significant chemical stability and important properties in many fields such as optoelectronic devices,1–3 molecular logic devices,4–7 optical sensors,8–10 photodynamic therapy,11–14 supramolecular self-assembly15–18 solar energy harvesting, and in storage devices.19–21 The extended π-conjugation system in the porphyrin skeleton22–24 leads to a wide range of visible light absorption and p-type properties as an electronic system. Some porphyrins have been tested for the photosensitization of wide-bandgap semiconductors, 25 and these p-type organic semiconductors are investigated because of their potential applications in electronic devices such as organic solar cells26–29 and organic field effect transistors. 30 Isonicotinic acid is an important drug intermediate with wide application prospects and has been used in the treatment of tuberculosis. 31 The synthesis of porphyrins combined with isonicotinic acid is expected to be beneficial in realizing the dual activity of porphyrins and isonicotinic acid.
It is important to study the electronic structure of porphyrins in the context of various applications such as photoluminescence and photoelectric conversion. The Raman spectroscopy, fluorescence spectra, surface photovoltage measurements, and UV-Vis absorption spectra are used widely to study the electronic structure of porphyrins.32–35 In addition, molecular dynamics simulations and UV-Vis diffuse-reflectance spectra are powerful tools for studying the chemical compositions and sample structures. All these techniques provide useful information for investigating the semiconductor properties of materials.36–38
In this work, we have synthesized asymmetric porphyrins possessing an isonicotinic acid moiety and phenyl, methoxyphenyl, or chlorophenyl groups. These porphyrins are characterized by 1H NMR, 13C NMR, MS, elemental analysis, IR, and UV-Vis. The effects of the substituents on these porphyrins are investigated through the Raman spectroscopy, fluorescence, and surface photovoltage measurements. Molecular dynamics simulations and UV-Vis diffuse-reflectance spectra show that these porphyrins are potential semiconductor materials.
Results and discussion
The synthetic procedure for the preparation of the porphyrin derivatives and their yields are illustrated in Scheme 1.
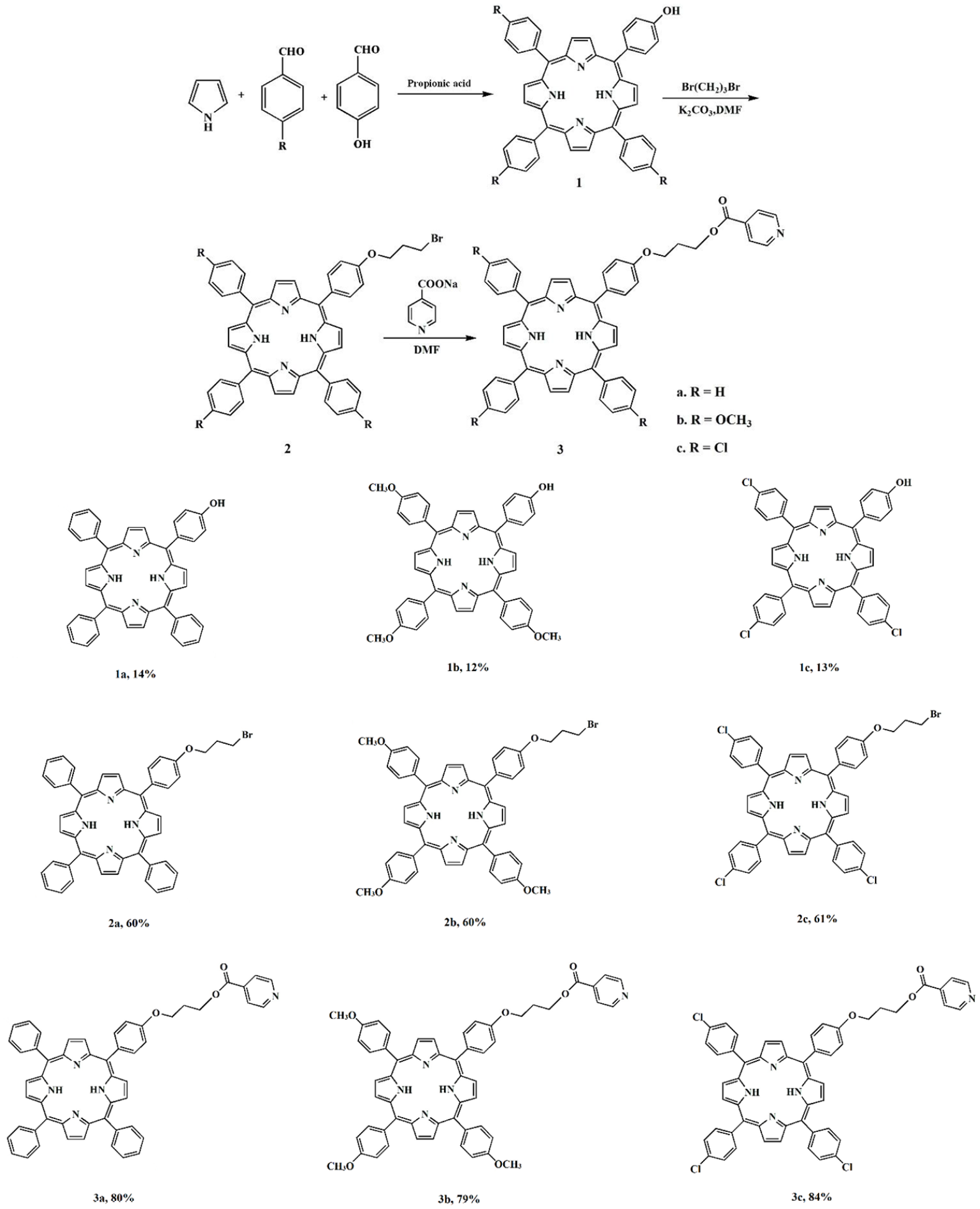
Synthesis route of
From the 1H NMR spectral data of
In the infrared spectra, the bands at 3317–3322 and 966 cm−1 are assigned to the N–H stretching vibrations and binding vibrations of the porphyrin core. The C–H stretching vibrations due to methylene and the C=O stretching vibrations appear in the range of 2931–3031 and 1720–1730 cm−1, respectively. The bands around 1508–1514 and 1471–1472 cm−1 are assigned to the skeletal vibrations of benzene and pyrrole, respectively. The C–O–C stretching vibrations appear in the range of 1222~1248 cm−1. In addition, the medium to strong band at 1089 cm−1 in the IR spectrum of 3c is assigned as overlapping bands due to the C=C (benzene) and C–Cl stretching vibrations which were characteristic of a phenyl with a para-chlorine substituent. 39
The UV–Vis absorption bands of porphyrins are determined by the electronic transitions from the ground state (S0) to the two lowest excited singlet states (S1 and S2). The S0 to S1 transition causes weak Q bands in visible region, while the transition of S0 to S2 produces a strong Soret band in the near UV region.40,41 From the UV–Vis absorption spectra data of compounds
From the Raman spectra (Figure 1), the Raman frequency data, and corresponding assignments (Table 1) of
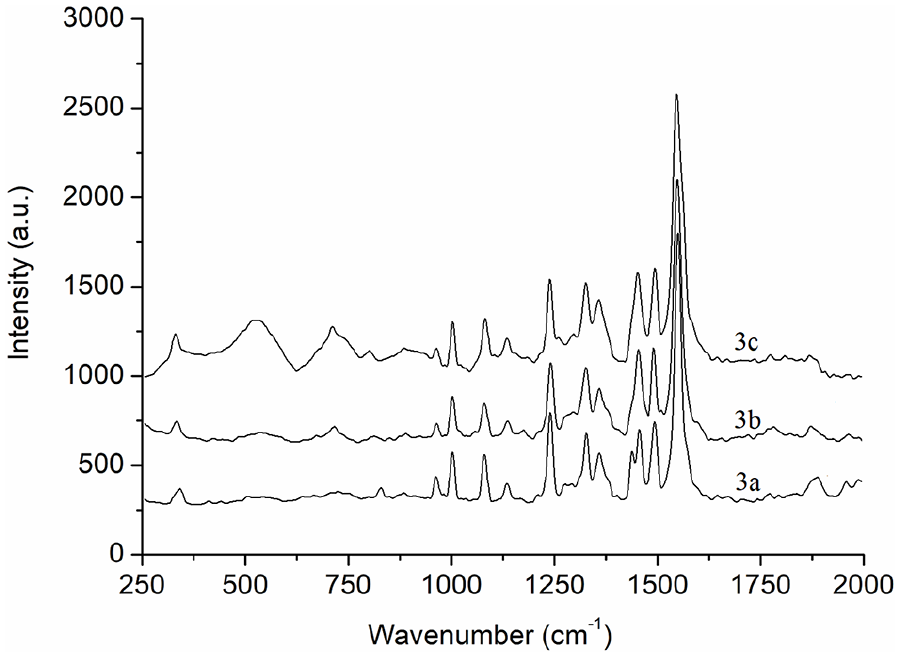
Raman spectra of
Raman frequencies and assignments of

vs: very strong; s: strong; m: medium; w: weak.
The fluorescence spectra of

Fluorescence spectra of
Emission spectral data of

The room-temperature fluorescence quantum yields of

where Fi is the integral areas of fluorescence, Ai represents the absorbance, and Φi represents the quantum yield at the same excitation wavelength. The fluorescence quantum yields of
Photogenerated electron transitions on the surface of the solid result in surface photovoltage. As shown in Figure 3, the surface photovoltage spectra of the

Surface photovoltage of
The geometric structures of the

(a) The stable conformation of

HOMO and LUMO distributions of
Molecular orbital energy distribution of

UV-Vis diffuse-reflectance spectra reflect the sample structure and composition through the interactions of light between the molecules such interactions include diffraction, reflection, refraction, and absorption. Quantitative analysis can be achieved by after UV-Vis diffuse-reflectance and correction via the Kubelka–Munk equation correction. At room temperature, and employing a scanning range of 200–800 nm, the UV-Vis diffuse-reflectance spectra of

where x and y are the λ and FR (Reflectance Function) in the UV-Vis diffuse-reflectance spectrum, respectively. X and Y are the Eg and FR
2
in the optical absorption spectrum, respectively. The Eg values are in the range of 1.74–1.82 eV. The experimental data are similar to that in the quantum chemistry theory, confirming our theoretical inference that
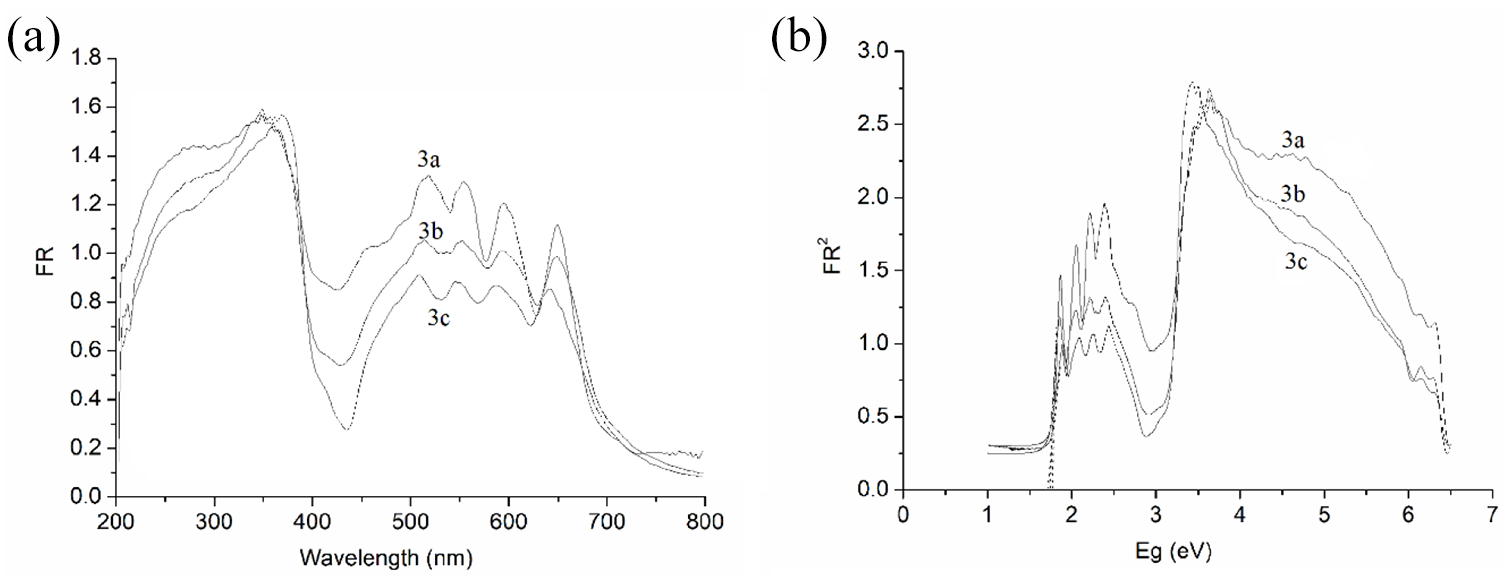
(a) UV-Vis diffuse-reflectance spectra of
Conclusion
In summary, three isonicotinic acid porphyrin compounds have been successfully synthesized and systematically characterized. Their spectroscopic properties have been investigated using Raman, fluorescence, and surface photovoltage spectra. These studies indicate that the fluorescence intensities and quantum yields are influenced by different substituents on the porphyrin ring. The fluorescence intensity and quantum yield of the porphyrin are increased by substituting with methoxy groups, while being decreased by the presence of chloro substituents. However, a competitive process is shown between fluorescence intensity and surface photovoltage intensity. The surface photovoltage intensity of the porphyrin is increased by connecting the chlorine groups, while it is decreased by incorporating methoxy groups. The chlorine (electron-withdrawing) groups have a stronger influence on the fluorescence intensity than methoxy (electron-donating) groups. The nature of the substituent has little influence on the Raman spectra of the isonicotinic acid porphyrin compounds. Furthermore, molecular dynamics simulations and UV-Vis diffuse-reflectance spectra indicate that these porphyrin compounds are potential semiconductor materials. In this paper, our research has focused on the synthesis and properties of three differently substituted isonicotinic acid porphyrin compounds, and the properties of the isonicotinic acid porphyrin metal complexes warrant further exploration in our future work.
Experimental
Apparatus and measurements
1 H NMR spectra were recorded on a Varian Unity 500 (MHz) NMR spectrometer in CDCl3. Chemical shifts are expressed in ppm with tetramethylsilane as the internal reference and reported as position (δH), relative integral. 13C NMR spectra were recorded on a Varian Unity 100 (MHz) NMR spectrometer in CDCl3. Mass spectra (fast atom bombardment (FAB)) were obtained using a VG-Quattro mass spectrometer. Infrared spectra were measured using a Nicolet-360 Fourier transform infrared (FTIR) spectrometer in the region 4000–400 cm−1 by incorporating the samples in KBr disks. The UV-Vis absorption spectra in the region 350–700 nm were obtained using a Shimadzu UV-3000 spectrometer using chloroform as the solvent. Resonance Raman spectra were recorded with a Renishaw RM1000 Raman spectrophotometer equipped with an integral microscopy and radiation of 514.5 nm was obtained from an Ar+ laser. Fluorescence spectra were recorded at room temperature with a Shimadzu RF-5301PC spectrofluorometer using chloroform as the solvent in the region of 500–800 nm. The excitation wavelength is 420 nm. Surface photovoltage spectra were recorded at room temperature with a 500-W Xe lamp, an SBP500 monochromator (30 nm/min), an FZ-A radiometer (<80 mW/cm2), a Stanford SR830-DSP lock-in amplifier (23 Hz), and a DSi200 Ultraviolet silicon detector (30 ms). Molecular dynamics simulations were achieved using the Material Studio 4.3. The UV-Vis diffuse-reflectance spectra in the region 200–800 nm were obtained using a Maypro 2000 spectrometer.
Materials
All the reagents and solvents were of analytical reagent grade. Pyrrole and N,N-dimethylformamide (DMF) were freshly distilled before using. Chloroform was dried using 4 Å molecular sieves (>12 h). Sodium isonicotinate was synthesized in our laboratory.
Synthetic procedures
5-(4-hydroxyphenyl)-10,15,20-triphenylporphyrin (
5-(4-Hydroxyphenyl)-10,15,20-triphenylporphyrin (1a )
A mixture of 4-hydroxybenzaldehyde (2.4 g, 0.021 mol), benzaldehyde (6.4 mL, 0.064 mol), and pyrrole (5.6 mL, 0.078 mol) was added dropwise to boiling propionic acid (250 mL). The reaction solution was refluxed for 0.5 h and then cooled to room temperature and placed in a refrigerator at −18 °C overnight. The mixture was then filtered and the obtained purple solid was washed with absolute ethanol. The crude product was dissolved in chloroform (50 mL) and chromatographed on a column of neutral alumina. The first band containing mesotetraphenylporphyrin was eluted by chloroform. The second band was eluted by chloroform containing 10% absolute ethanol and contained the desired porphyrin. The second fraction was concentrated and cooled. The precipitated product was dried in vacua. Yield: 14%. 1H NMR (500 MHz, CDCl3): δ −2.79 (s, 2H, inner NH), 7.65-7.80 (m, 9H, Ar-3,4,5-H), 7.91-7.96 (d, 2H, ArO-3, 5-H), 8.09-8.20 (m, 8H, Ar-2, 6-H), 8.78-8.86 (m, 8H, pyrrole, β-H), 9.99 (s, 1H, OH) in agreement with literature values. 50
5-(4-Hydroxyphenyl)-10,15,20-tri(4-methoxyphenyl)porphyrin (1b )
Porphyrin
5-(4-Hydroxyphenyl)-10,15,20-tri(4-chlorophenyl)porphyrin (1c )
Porphyrin
5-(4-Bromopropyloxy)phenyl-10,15,20-triphenylporphyrin (
5-[4-(3-Bromopropyloxy)phenyl]-10,15,20-triphenylporphyrin (2a )
A mixture of
5-[4-(3-Bromopropyloxy)phenyl]-10,15,20-tri(4-methoxyphenyl)porphyrin (2b )
Compound
5-[4-(3-Bromopropyloxy)phenyl]-10,15,20-tri(4-chlorophenyl)porphyrin (2c )
Compound
5-{4-[3-(4-pyridylcarbonyloxy)propoxy]phenyl}-10,15,20-triphenylporphyrin (3a )
A mixture of sodium isonicotinate (57 mg, 0.39 mmol) and compound
5-{4-[3-(4-pyridylcarbonyloxy)propoxy]phenyl}-10,15,20-tri(4-methoxyphenyl)porphyrin (3b )
Compound
5-{4-[3-(4-pyridylcarbonyloxy)propoxy]phenyl}-10,15,20-tri(4-chlorophenyl)porphyrin (3c )
Compound
Supplemental Material
sj-docx-1-chl-10.1177_17475198211032835 – Supplemental material for Synthesis and properties of unsymmetrical porphyrins possessing an isonicotinic acid moiety and phenyl, methoxyphenyl, or chlorophenyl groups
Supplemental material, sj-docx-1-chl-10.1177_17475198211032835 for Synthesis and properties of unsymmetrical porphyrins possessing an isonicotinic acid moiety and phenyl, methoxyphenyl, or chlorophenyl groups by Shiqi Wang, Yao Li and Binbin Wang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NSFC (U1804156 and 42002164), the China Postdoctoral Science Foundation (2018M632775), the Henan Province Colleges and Universities Key Research Project (18A620002 and 18A150005), the State Key Laboratory Cultivation Base for Gas Geology and Gas Control (Henan Polytechnic University) (WS2018A05), and the Natural Science Foundation of Henan Province (202300410171).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
