Abstract
Photodynamic therapy is a promising cancer treatment with the advantages of low toxicity, high efficiency, and noninvasiveness. In this study, 23 novel porphyrin–chrysin derivatives are synthesized using alkyl carbon chains as bridges. We use human gastric cancer cells (MGC-803) and human cervical cancer cells to evaluate the in vitro antitumor activity of all the porphyrin–chrysin derivatives, with 5-fluorouracil (5-Fu) as a positive control. Several of the prepared compounds showed effective photodynamic killing effects, among which 5-hydroxy-2-phenyl-7-(2-(4-(10,15,20-tris(4-hydroxyphenyl)porphyrin-5-yl)phenoxy)ethoxy)-4H-chromen-4-one shows the highest antiproliferation activity on human cervical cancer cells, with a half maximal inhibitory concentration of 26.51 ± 1.15 µM. Flow cytometry analysis showed that human cervical cancer cell apoptosis might be induced by G1 phase arrest.

Introduction
Photodynamic therapy (PDT) is a new technology that was began to be applied to cancer therapy in the late 1970s. Thereafter, PDT has been accepted by the relevant departments of the United States, the United Kingdom, Germany, Japan, and other countries to enter clinical applications. Until now, this therapy has been used to treat a variety of malignancies successfully.1–3
The basic elements of PDT can be divided into photosensitizers, specific excitation light, and molecular oxygen. The photosensitizer transfers the absorbed energy to the surrounding oxygen molecules through light irradiation, so that the oxygen molecules become excited singlet oxygen (1O2) and other reactive oxygen species (ROS) to achieve the purpose of killing tumor cells. 4 In fact, the PDT beam is different from the usual clinical laser treatment in that its energy density is low, and its purpose is to activate the photosensitizer without causing tissue damage.
Although a number of photosensitizers have been suggested for PDT, 5 porphyrins, and their analogs remain the most extensively employed photosensitive compounds.6,7 Porphyrins have the following advantages for PDT. On one hand, they are satisfactory for singlet oxygen production, and on the other hand, they have the ability to preferentially aggregate in tumor tissues. There are two general theories about the mechanism of specific aggregation of porphyrins in tumor tissues. According to pH theory, most porphyrin molecules enter the cell through passive diffusion. Numerous experiments have shown that porphyrin compounds are more conducive to maintaining the molecular state in a weakly acidic tumor cell environment compared to normal tissues, thereby facilitating their entry into tumor cells in molecular form.8–10 Based on the theory of low-density lipoproteins (LDLs), the surface of rapidly growing tumor cells contains more LDL receptors that show strong activity. When the porphyrin compound binds to the LDLs, the LDLs transport the drug to bind to receptors on the cell membrane so that the drug can accumulate in the tumor tissue.11–14 Based on this, a large number of antitumor porphyrin derivatives have been reported.15–18
Chrysin is a natural flavonoid and has a wide range of pharmacological activities such as anti-inflammatory,19,20 antioxidant,21,22 antibacterial, 23 hypoglycemic, 24 antitumor,25–27 and so on. However, there are many shortcomings in the chemical structure of chrysin. Research by scientists in both China and abroad on chrysin has mainly focused on the modification and optimization of its chemical structure. On one hand, chrysin shows poor lipophilic and water-solubility properties, which limits its clinical application. Second, the 5- and 7-hydroxy groups of chrysin are readily inactivated by rapid glycosylation and metabolism in vivo. Therefore, the structural optimization of chrysin in order to improve its bioavailability and activity has been the subject of significant research.28–30
To this end, we have innovatively combined the antitumor compounds porphyrin and chrysin by appropriate methods. Finally, a series of novel porphyrin–chrysin derivatives have been obtained and the products tested for their performance and in vitro antitumor activity.
Results and discussion
Chemical synthesis
In this study, 23 novel substituted porphyrin–chrysin derivatives have been synthesized. First, we prepared the chrysin and porphyrin derivatives. The chrysin derivatives

Synthesis of the chrysin derivatives.

Synthesis of the porphyrin compounds.

Synthesis of the porphyrin–chrysin derivatives.
Performance testing
UV-Vis spectra
As shown in Table 1, the Soret band and the Q band are two characteristic absorption bands of the porphyrin compounds. The Soret band is generally around 418 nm and exhibits strong absorption. The Q band is generally between 500 and 700 nm and exhibits weak absorption. The II band is a characteristic absorption band of flavonoids.
38
We found that the Soret band absorption peaks of the derivatives
UV/Vis absorption spectral data of the porphyrin–chrysin derivatives. a

The structural formulae of the porphyrin–chrysin compounds are shown in Scheme 3.
Fluorescence spectra
The results of fluorescence spectroscopy showed that the maximum fluorescence emission wavelengths of the derivatives
Detection of singlet oxygen (1O2)
1O2 is a powerful ROS, which can effectively induce the apoptosis of tumor cells. 1,3-Diphenylisobenzofuran (DPBF) is a commonly used singlet oxygen–trapping agent, which can be oxidized by singlet oxygen to open the ring and destroy the conjugated structure, resulting in the disappearance of the fluorescence signal. We have used the magnitude and rate of its fluorescence intensity change around 456 nm to evaluate the ability of compounds to produce singlet oxygen.
39
As shown in Figure 1, compared with the DPBF control group, the fluorescence intensity of the DPBF solution and the mixed solution of the target compounds porphyrin–chrysin derivatives
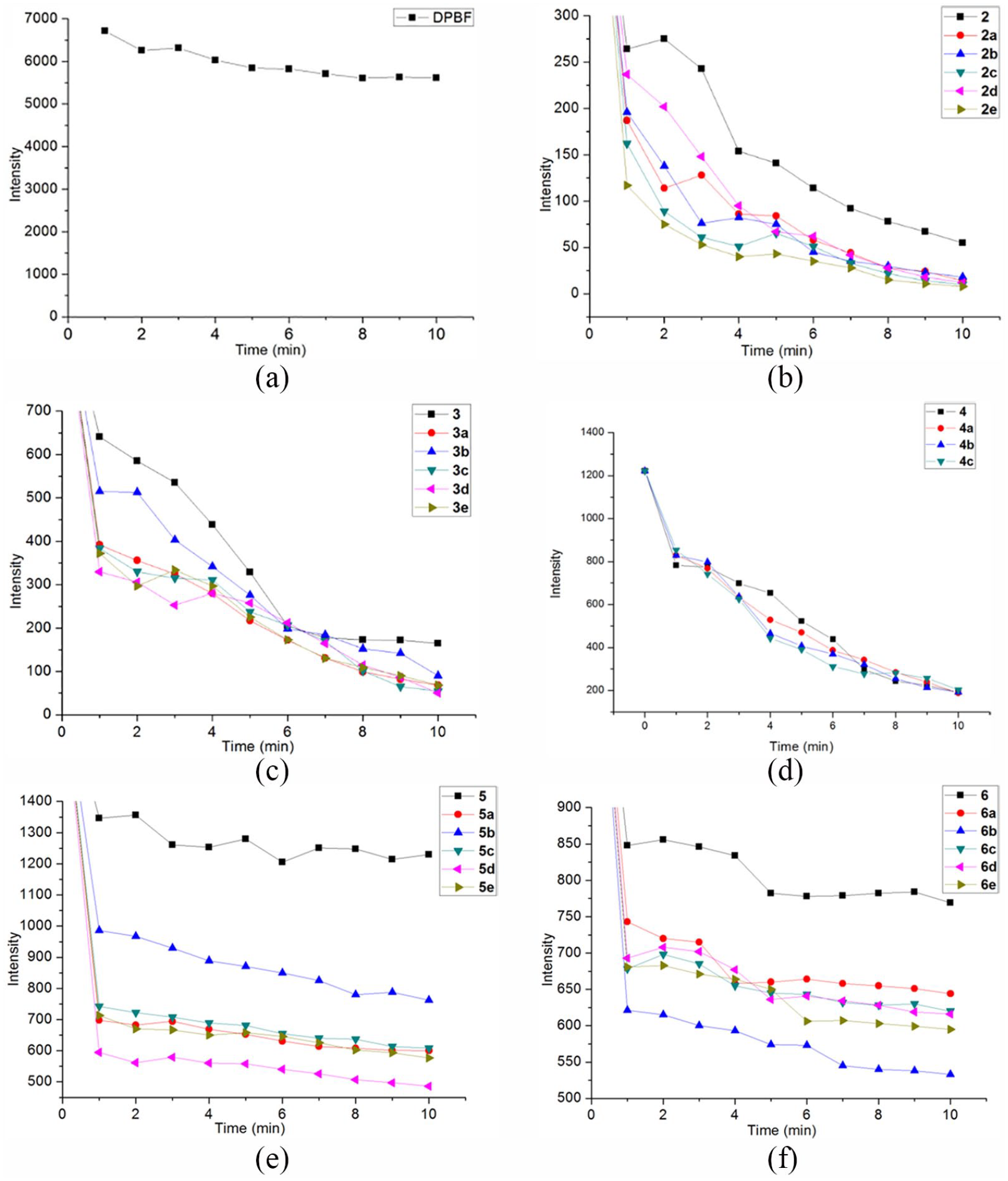
Singlet oxygen yields of the porphyrin–chrysin derivatives and DPBF (a) is the change of fluorescence intensity of DPBF within 10 min; (b)–(f), are the changes of the fluorescence intensities of DPBF within 10 min, respectively, in the derivatives
Comparing the presence of Figure 1(b)–(d) with (e) and (f), it can be seen that the free-base porphyrin derivatives
Biological activities
In vitro antiproliferative activity assay
The antiproliferative activity in vitro of compounds
According to the data in Table 2, the antiproliferative activity of the light group was significantly stronger than that of the dark group. Derivatives
Anti-proliferative activity against tumor cell lines.

ND = not detected.
Cell cycle assay
A cell cycle assay was conducted to check the distribution of HeLa cells, treated with compound
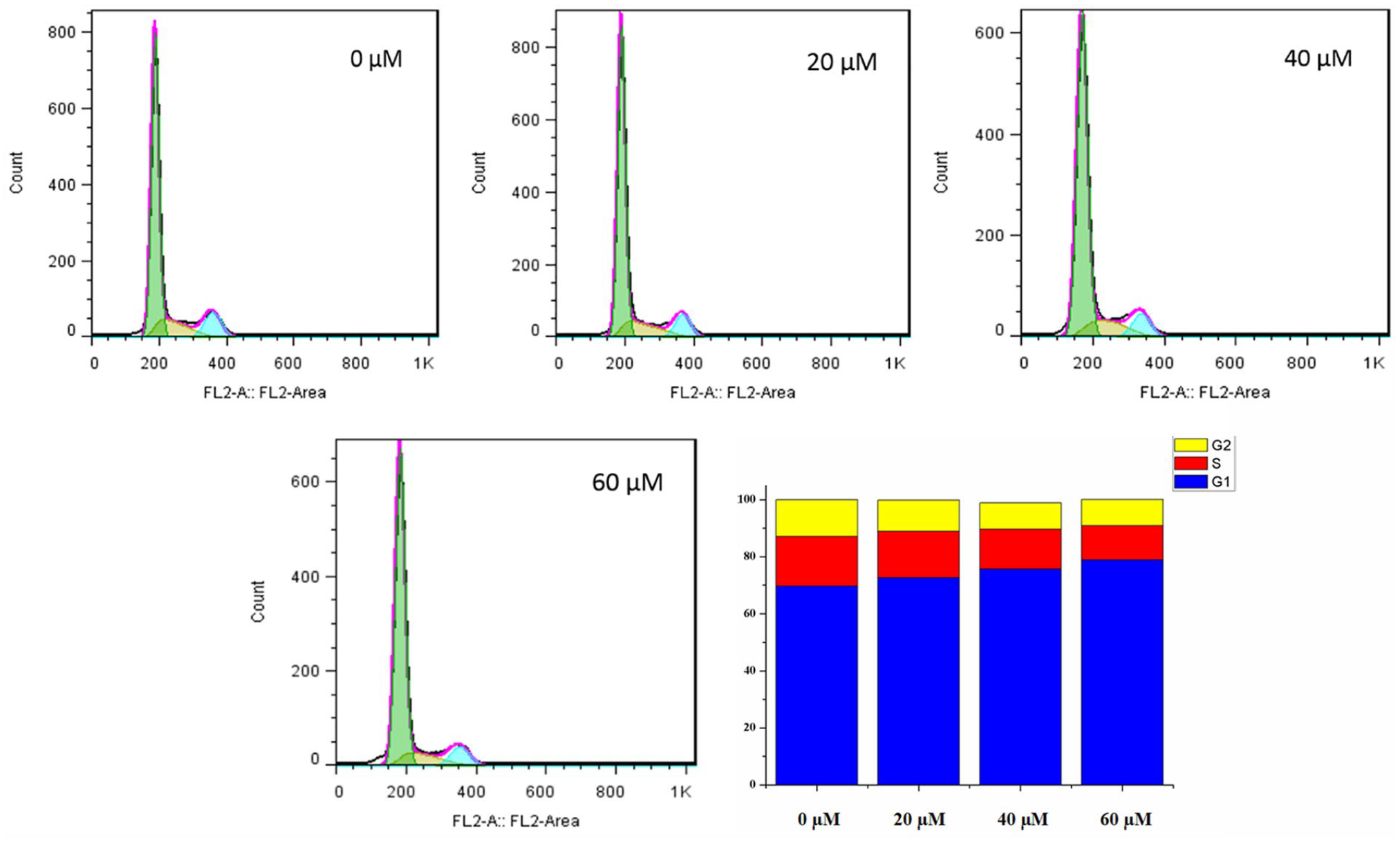
Inhibition of the cell cycle of HeLa cells by compound
Apoptosis assay
The flow cytometry assay determined the effect of compound

Effect of compound
Conclusion
In summary, a series of novel porphyrin–chrysin derivatives have been synthesized, and these compounds were evaluated for their 1O2 and antitumor activities. The results of MTT in vitro antiproliferative experiments showed that compounds
Experimental section
All the starting materials, solvents, and reagents were reagent grade and purchased from commercial sources unless otherwise stated. Chrysin was purchased from Aladdin. 1H NMR was recorded on a Bruker AVANCE instrument (400 and 500 MHz, Bruker, Germany). MALDI-TOF MS data were recorded using a Bruker Daltonics flex analyzer (Bruker, Germany).
Synthesis methods
Synthesis of chrysin derivatives 1a –1e. 31,32
The appropriate 1, n-dibromoalkane (10 mmol), K2CO3 (50 mmol), and KI (50 mmol) were dissolved in acetone (60 mL) and stirred at 60°C for 1 h under reflux. Next, chrysin (508.5 mg, 2 mmol) was slowly added to the reaction system in batches, and the reaction progress was monitored by thin-layer chromatography (TLC) until the reaction was complete. The reaction solution was extracted with dichloromethane and washed twice with water to remove impurities. The organic layer was dried over anhydrous Na2SO4, and then the solvent was distilled off under reduced pressure. The residue purified by silica gel column chromatography. The first color band was collected with dichloromethane/n-hexane (3: 1) as eluent, and yellow crystals were obtained by recrystallization from n-hexane.
Synthesis of porphyrin compounds 2 , 3 , and 4. 33–36
Compounds
Synthesis of nickel metalloporphyrin compounds 5 and 6.
37
Porphyrin
Synthesis of porphyrin–chrysin derivatives 2a –e , 3a –e , 4a –c , 5a –e , and 6a –e
Compound
Performance characterization
UV-Vis spectrometry
The test sample was dissolved in chloroform to prepare a working solution having a concentration of 10 μM, and the absorption spectra were scanned between 200 and 800 nm using a UV-Vis spectrophotometer.
Fluorescence spectrometry
The test compounds were accurately weighed using an analytical balance, and all the samples were dissolved in chloroform, and the sample was prepared as a test solution having a concentration of 10 μM. The samples were scanned using an F-7000 FL fluorescence spectrometer with a scanning range of 200–800 nm and an excitation wavelength of 418 nm.
1O2 detection
The test compounds were accurately weighed using an analytical balance and all the samples and DPBF were separately dissolved in chloroform. The sample was prepared as a test solution containing a sample (concentration: 10 μM) and DPBF (concentration: 100 μM). The samples were analyzed using an F-7000 FL fluorescence spectrometer. The excitation wavelength was set to 418 nm, the scanning speed was 2400 nm/min, the scanning range was 200–800 nm, and the scanning was performed at intervals of 1 min. The fluorescence changes of DPBF were recorded at 463 nm.
Biological activity detection
In vitro antitumor efficacy
The in vitro antitumor efficacy of the compounds was evaluated using the human tumor cells MGC-803 and HeLa cells in parallel with PDT. Each tested compound was dissolved in dimethyl sulfoxide (DMSO). The cells were plated in 96-well micro-titer plates at a density of 1 × 105 cells per well and incubated in a humidified atmosphere with 5% CO2 at 37°C for 48 h. One of the experimental groups was removed after the first 4 h. The upper culture medium was carefully removed with a pipette and refilled with the drug-free culture fluid. Light was supplied by a 12 W LED purple lamp located 20 cm from the 96-well plate. Irradiation was continued for 10 min, after which the cells were placed in an incubator to continue the culture incubation. Test compounds of different concentrations (16, 32, 64, 128 μM) were added into triplicate wells and DMSO was used as the control. After 48 h, 20 mL of MTT solution (5 mg mL−1) was added to each well, and incubation was continued for an additional 4 h. Formazan was dissolved in 150 mL of DMSO and added to the wells. The absorbance (optical density, OD) was monitored on a Wellscan MK-2 microplate reader (Lab-Systems) at 490 nm. The IC50 values were determined using the logit method. All experiments were performed three times.
Cell cycle analysis
The HeLa cells were plated in six-well plates (1 × 105 cells per well). After 24 h, the cells were incubated with different concentrations of compound
Cell apoptosis assay
The HeLa cells were plated in six-well plates (1 × 105 cells per well). After 24 h, the cells were incubated with different concentrations of compound
Supplemental Material
Supporting_information_2 – Supplemental material for Novel photosensitizing properties of porphyrin–chrysin derivatives with antitumor activity in vitro
Supplemental material, Supporting_information_2 for Novel photosensitizing properties of porphyrin–chrysin derivatives with antitumor activity in vitro by Ding Liu, Qizhi Zhang, Lang Zhang, Wenmei Yu, Huizhi Long, Jun He and Yunmei Liu in Journal of Chemical Research
Footnotes
Acknowledgements
Ding Liu and Qizhi Zhang are contributed equally to this work and should be regarded as co-first authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Hunan Provincial Department of Education Key Project (grant no. 18A244), the Hunan Province University Students Research Learning and Innovative Experimental Project (grant no. S201910555017), and the Hunan Natural Science Foundation (grant no. 2018JJ2349).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
