Abstract
A series of novel methylated (–)-epigallocatechin-3-gallate-4β-triazolopodophyllotoxin derivatives is synthesized by utilizing the click reaction. Evaluation of their cytotoxicity against a panel of five human cancer cell lines (HL-60, SMMC-7721, A-549, MCF-7, and SW480) using the MTT assay shows that most of these compounds exhibit weak cytotoxicity. It is observed that compound
Introduction
Natural products are valuable materials with various structures, unique biological activities, and specific selectivity that can aid in the discovery of anticancer drugs.
1
However, many potential drugs lack tumor selectivity and often display significant toxic side effects, which hampers their development for clinical use.2,3 In order to reduce cytotoxicity and maintain activity of these natural compounds, appropriate structural modification can be adopted in order to develop drugs. Podophyllotoxin (PPT,

Chemical structures of podophyllotoxin (PPT,
Green tea (Camellia sinensis leaves) is an extremely popular drink,
12
and the major catechins found in green tea are (–)-epicatechin (EC,
Typically, tubulin inhibitors with a trimethoxyphenyl (TMP) moiety, which is essential for interactions with tubulin, are interesting compounds. In our previous studies, 27 we reported a group of (–)-epigallocatechin-3-gallate-4β-triazolopodophyllotoxin conjugates as a new class of antitumor compounds. Herein, we report the chemical synthesis of a series of methylated (–)-epigallocatechin-3-gallate-4β-triazolopodophyllotoxin derivatives from the natural compounds PPT and EGCG by click reactions, and have evaluated their in vitro anticancer activity against five human cancer cell lines: including HL-60 (leukemia), SMMC-7721 (hepatoma), A-549 (lung cancer), MCF-7 (breast cancer), and SW480 (colon cancer). Besides, molecular docking analyses of these compounds were studied and correlated with their anticancer activity.
Results and discussion
Chemistry
As shown in Scheme 1, the first part of the synthesis of the methylated (–)-epigallocatechin-3-gallate-4β-triazolopodophyllotoxin derivatives required the preparation of the terminal alkynes
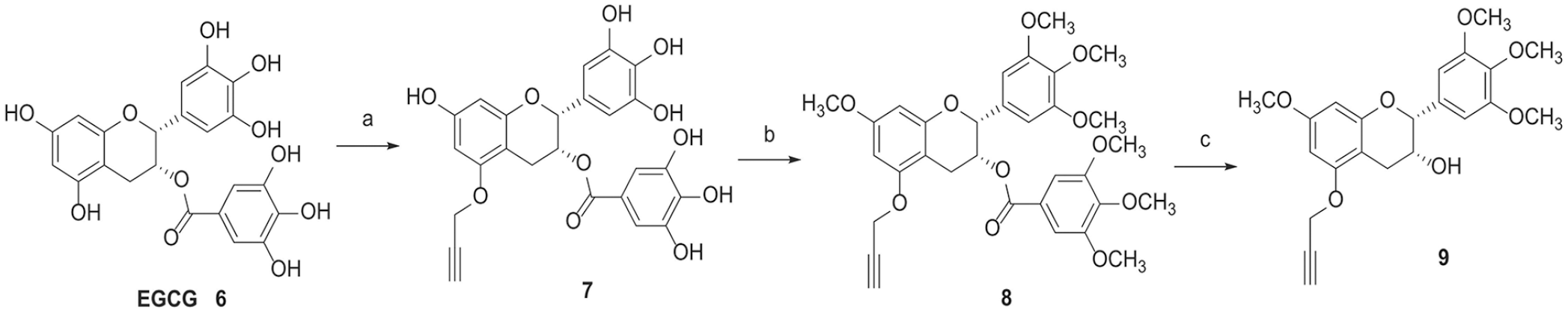
Synthesis of the terminal alkynes

Synthesis of methylated (–)-epigallocatechin-3-gallate-4β-triazolopodophyllotoxin derivatives
All the synthesized compounds were characterized by 1H NMR, 13C NMR, and ESI-MS analytical data. The formation of the triazole ring was confirmed by the resonance of its C14-H signal (7.8–8.2 ppm) in the aromatic region in the 1H NMR spectra, and which was further supported by two characteristic carbon signals at around 145 and 126 ppm in the 13C NMR spectra.
In vitro antiproliferative activity
The antiproliferative activity of methylated (–)-epigallocatechin-3-gallate-4β-triazolopodophyllotoxin derivatives
In vitro anticancer activity (IC50, μM) of methylated (–)-epigallocatechin-3-gallate-4β-triazolopodophyllotoxin derivatives
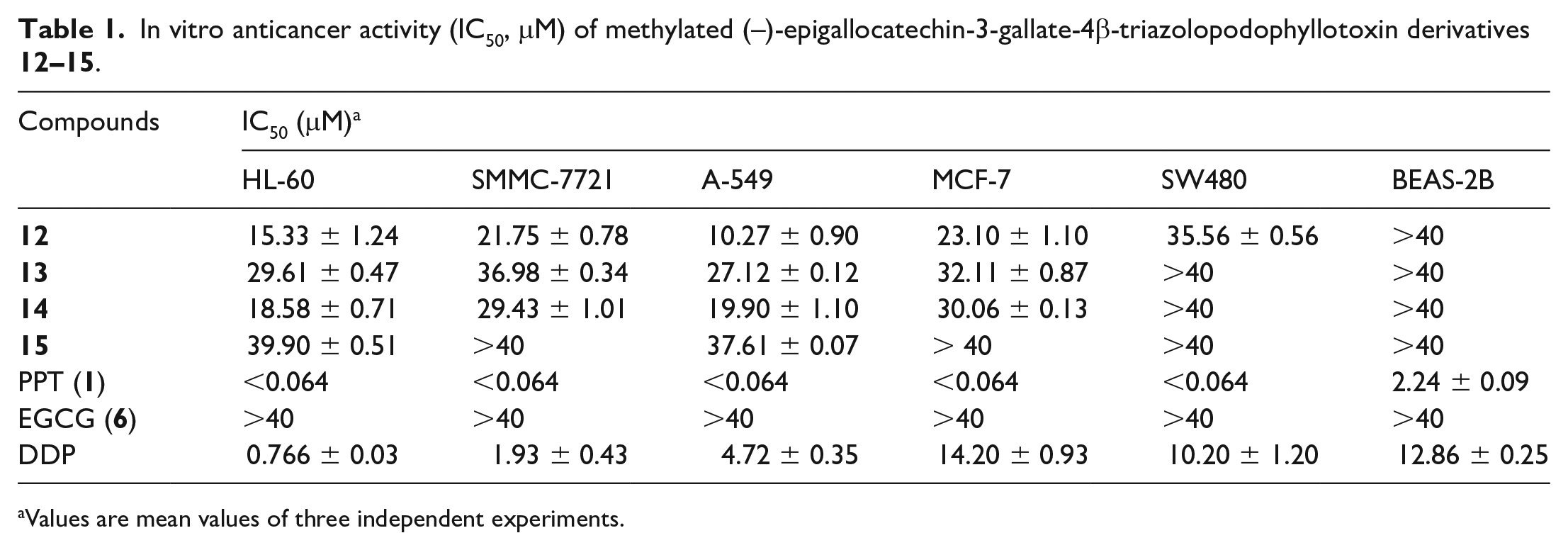
Values are mean values of three independent experiments.
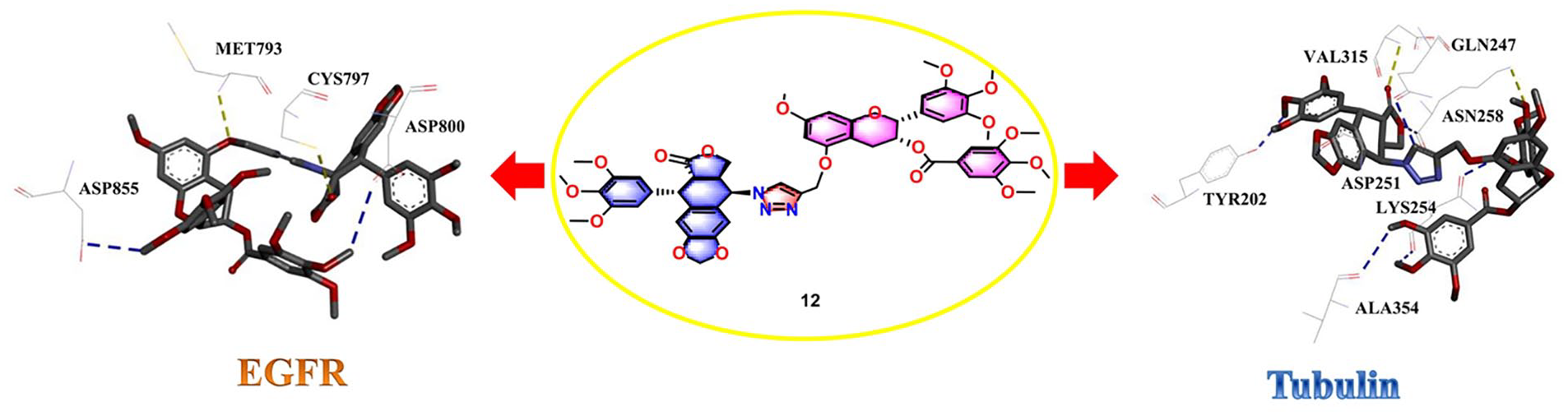
Molecular docking studies
To further investigate the potential binding between tubulin (PDB code: 3UT5)/EGFR (PDB code: 3IKA) and compound

Binding sits of proteins and compound
The residues for molecular interaction between tubulin/

Conclusion
In summary, a series of novel (–)-epigallocatechin-3-gallate-4β-triazolopodophyllotoxin derivatives has been synthesized by utilizing the click-reaction, and the cytotoxicity of these compounds has been screed against a panel of five human cancer cells (HL-60, SMMC-7721, A-549, MCF-7, and SW480) using the MTT assay. Compound
Experimental
General
All cancer cells (HL-60, SMMC-7721, A-549, MCF-7, and SW480) were obtained from the Shanghai Cell Bank, China. (–)-Epigallocatechin-3-gallate and podophyllotoxin were purchased from Chengdu Proifa Technology Development Co., Ltd (Chengdu, China); 3-(4,5-dimethyl-thiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) was obtained from Sigma-Aldrich (St. Louis, MO, USA). All reagents were commercially available and were used without further purification unless indicated otherwise. Melting points were measured with an X-4 melting point apparatus and are uncorrected. MS data were obtained in the ESI mode on an API Qstar Pulsar instrument; HRMS data were obtained in the ESI mode on an LCMS-IT-TOF (Shimadzu, Kyoto, Japan) instrument. 1H NMR and 13C NMR spectra were recorded on Bruker AVANCE III 500 MHz (Bruker BioSpin GmbH, Rheinstetten, Germany) instrument, using tetramethylsilane (TMS) as an internal standard: chemical shifts (δ) are given in ppm, coupling constants (J) in Hz, and the solvent signals were used as references (CDCl3: δC = 77.2; residual CHCl3 in CDCl3: δH = 7.26; CD3OD: δC = 49.0; residual CH3OH in CD3OD: δH = 4.78). Column chromatography (CC) was performed using silica gel (200–300 mesh; Qingdao Makall Group Co., Ltd.; Qingdao; China). All reactions were monitored by thin-layer chromatography (TLC), and samples were visualized under ultraviolet light (254 nm) and/or using 10% phosphomolybdic acid/EtOH.
(–)-Epigallocatechin-3-gallate-1-propyne (7)
(–)-Epigallocatechin-3-gallate (2.3 g, 5.0 mmol) was dissolved in N,N-dimethylformamide (DMF) (20 mL), and then sodium hydride (300 mg, 7.5 mmol) was added at 0 °C under nitrogen and the mixture was stirred at room temperature for 0.5 h. Propargyl bromide (0.5 mL, 5.0 mmol) was quickly added, and the reaction was stirred at 80 °C for 12 h. After cooling, the mixture was concentrated under vacuum and the resulting residue was purified by silica gel chromatography with CHCl3/CH3OH, (9:1 → 4:1) to afford the product
Methylated (–)-epigallocatechin-3-gallate-1-propyne (8)
To a solution of (–)-epigallocatechin-3-gallate-1-propyne (
Methylated (–)-epigallocatechin-1-propyne (9)
To a solution of methylated (–)-epigallocatechin-3-gallate-1-propyne (
General procedure for the synthesis of methylated catechin-4β-triazolopodophyllotoxin derivatives 12–15
To a solution of methylated catechin-1-propyne
Methylated (–)-epigallocatechin-3-gallate-[4β-(1,2,3-trizaol-1-yl-4-deoxypodophyll-otoxin)] ether (12)
White amorphous powder; yield 82%; m.p. = 180–182 °C; 1H NMR (500 MHz, CD3OD): δ 7.92 (s, 1H, C14-H), 6.98 (s, 2H, C2'', C6''-H), 6.82 (s, 2H, C2', C6'-H), 6.68 (s, 1H, C5'''-H), 6.65 (s, 1H, C8'''-H), 6.30 (s, 2H, C2'''', C6''''-H), 6.23 (d, 1H, J = 2.3 Hz, C6-H), 6.21 (d, 1H, J = 2.3 Hz, C8-H), 6.17 (d, 1H, J = 2.3 Hz, C3-H), 6.02-5.97 (m, 2H, OCH2O), 5.55 (s, 1H, C2-H), 5.25 (s, 2H, OCH2), 5.03 (d, 1H, J = 3.0 Hz, C4'''-H), 4.71 (d, 1H, J = 5.0 Hz, C1'''-H), 4.27-4.25 (m, 2H, C11'''-CH2), 3.72 (s, 6H, C3'', C5''-OCH3), 3.71 (s, 3H, C7-3OCH), 3.66-3.60 (m, 21H, C3', C4', C5', C4'', C3''''', C4'''', C5''''-OCH3), 3.40 (dd, 1H, J = 4.6, 12.0 Hz, C4-Ha), 3.03 (dd, 1H, J = 4.6, 12.0 Hz, C4-Hb), 2.89-2.86 (m, 1H, C3'''-H), 2.83-2.81 (m, 1H, C2'''-H); 13C NMR (125 MHz, CD3OD): δ 173.5 (C-12'''), 164.3 (C-11), 159.4 (C-7), 158.4 (C-5), 155.1 (C-9), 153.0 (C-3'''', C-5''''), 152.4 (C-3'', C-5''), 152.1 (C-3', C-5'), 148.2 (C-7'''), 147.0 (C-6'''), 142.9 (C-4''), 140.0 (C-14), 137.0 (C-4''''), 136.5 (C-4'), 135.2 (C-1''''), 133.8 (C-9'''), 132.9 (C-10'''), 126.0 (C-1'), 125.3 (C-13), 125.1 (C-1''), 109.8 (C-5'''), 108.7 (C-8'''), 108.1 (C-2'''', C-6''''), 106.5 (C-2'', C-6''), 103.8 (C-2', C-6'), 101.7 (OCH2O), 99.5 (C-10), 93.4 (C-8), 91.8 (C-6), 76.6 (C-2), 69.0 (OCH2), 67.1 (C-3, C-11'''), 65.0 (C-4'''), 60.2 (4''''-OCH3), 60.0 (4''-OCH3), 59.9 (4'-OCH3), 57.3 (7-OCH3), 56.0 (3''''-OCH3), 55.8 (5''''-OCH3), 55.7 (3''-OCH3), 55.6 (5''-OCH3), 55.4 (3'-OCH3), 55.2 (5'-OCH3), 42.9 (C-1'''), 40.7 (C-2'''), 36.4 (C-3'''), 25.3 (C-4); MS (ESI): m/z = 1034 [M + H]+.
Methylated (–)-epigallocatechin-3-gallate-[4β-(1,2,3-trizaol-1-yl-4-deoxy-4’-demethylpodophyllotoxin)] ether (13)
White amorphous powder; yield 78%; m.p. = 185–186 °C; 1H NMR (500 MHz, CD3OD): δ 7.91 (s, 1H, C14-H), 6.09 (s, 2H, C2'', C6''-H), 6.82 (s, 2H, C2', C6'-H), 6.65 (d, 1H, J = 2.3 Hz, C5'''-H), 6.64 (d, 1H, J = 2.3 Hz, C8'''-H), 6.26 (s, 2H, C2'''', C6''''-H), 6.23 (d, 1H, J = 2.3 Hz, C6-H), 6.21 (d, 1H, J = 2.3 Hz, C8-H), 6.17 (d, 1H, J = 2.3 Hz, C3-H), 6.02-5.97 (m, 2H, OCH2O), 5.55 (brs, 1H, C2-H), 5.25 (s, 2H, OCH2), 5.03 (d, 1H, J = 3.0 Hz, C4'''-H), 4.66 (d, 1H, J = 5.0 Hz, C1'''-H), 4.27-4.25 (m, 2H, C11'''-CH2), 3.74 (s, 6H, C3'', C5''-OCH3), 3.72 (s, 3H, C7-3OCH), 3.71-3.60 (m, 18H, C3', C4', C5', C4'', C3''''', C4'''', C5''''-OCH3), 3.34-3.31 (m, 1H, C4-Ha), 3.04-3.01 (m, 1H, C4-Hb), 2.89-2.87 (m, 1H, C3'''-H), 2.83-2.81 (m, 1H, C2'''-H); 13C NMR (125 MHz, CD3OD): δ 173.5 (C-12'''), 164.3 (C-11), 159.4 (C-7), 158.4 (C-5), 155.1 (C-9), 153.0 (C-3'''', C-5''''), 152.7 (C-3'', C-5''), 152.1 (C-3', C-5'), 148.1 (C-7'''), 147.3 (C-6'''), 142.9 (C-4''), 140.0 (C-14), 137.0 (C-4''''), 134.9 (C-4'), 133.8 (C-9'''), 133.3 (C-10'''), 129.6 (C-1''''), 126.0 (C-1'), 125.3 (C-13), 125.1 (C-1''), 109.8 (C-5'''), 108.6 (C-8'''), 108.4 (C-2'''', C-6''''), 106.5 (C-2'', C-6''), 103.8 (C-2', C-6'), 101.6 (OCH2O), 99.5 (C-10), 93.4 (C-8), 91.8 (C-6), 76.6 (C-2), 69.0 (OCH2), 67.0 (C-3, C-11'''), 65.0 (C-4'''), 60.2 (4''-OCH3), 60.0 (4'-OCH3), 57.4 (7-OCH3), 56.00 (3''''-OCH3), 55.9 (5''''-OCH3), 55.8 (3''-OCH3), 55.7 (5''-OCH3), 55.5 (3'-OCH3), 55.2 (5'-OCH3), 42.8 (C-1'''), 40.8 (C-2'''), 36.3 (C-3'''), 25.3 (C-4); MS (ESI): m/z = 1029 [M + H]+.
Methylated (–)-epigallocatechin-[4β-(1,2,3-trizaol-1-yl-4-deoxypodophyllotoxin)] ether (14)
White amorphous powder; yield 84%; m.p. = 182–184 °C; 1H NMR (500 MHz, CD3OD): δ 7.58 (s, 1H, C14-H), 6.78 (s, 2H, C2'', C6''-H), 6.65 (s, 1H, C5'''-H), 6.62 (s, 1H, C8'''-H), 6.40 (s, 2H, C2', C6'-H), 6.22 (d, 1H, J = 2.3 Hz, C6-H), 6.17 (d, 1H, J = 2.3 Hz, C8-H), 6.14 (d, 1H, J = 2.3 Hz, C3-H), 5.97-5.96 (m, 2H, OCH2O), 5.25-5.20 (m, 3H, C2-H, OCH2), 5.13 (d, 1H, J = 3.0 Hz, C4'''-H), 4.74 (d, 1H, J = 5.0 Hz, C1'''-H), 4.27-4.25 (m, 2H, C11'''-CH2), 3.78 (s, 3H, C7-3OCH), 3.78-3.72 (m, 18H, C3', C4', C5', C3''''', C4'''', C5''''-OCH3), 3.29-3.27 (m, 1H, C4-Ha), 3.10-3.08 (m, 1H, C4-Hb), 2.88-2.86 (m, 1H, C3'''-H), 2.84-2.82 (m, 1H, C2'''-H); 13C NMR (125 MHz, CD3OD): δ 175.9 (C-12'''), 161.1 (C-7), 160.6 (C-5), 156.9 (C-9), 154.6 (C-3'''', C-5''''), 154.0 (C-3', C-5'), 150.6 (C-7'''), 149.3 (C-6'''), 138.3 (C-4''''), 137.3 (C-14), 136.7 (C-1''''), 135.7 (C-4'), 134.7 (C-9'''), 134.7 (C-10'''), 126.7 (C-13), 126.4 (C-1'), 111.2 (C-5'''), 109.9 (C-8'''), 109.4 (C-2'''', C-6''''), 105.4 (C-2', C-6'), 103.3 (OCH2O), 102.1 (C-10), 94.7 (C-8), 92.5 (C-6), 80.0 (C-2), 68.9 (OCH2), 67.3 (C-3, C-11'''), 65.7 (C-4'''), 61.1 (4''''-OCH3), 59.8 (4'-OCH3), 56.8 (7-OCH3), 56.7 (3''''-OCH3), 56.6 (5''''-OCH3), 55.9 (3'-OCH3), 55.8 (5'-OCH3), 44.9 (C-1'''), 42.5 (C-2'''), 38.6 (C-3'''), 29.4 (C-4); MS (ESI): m/z = 840 [M + H]+.
Methylated (–)-epigallocatechin-[4β-(1,2,3-trizaol-1-yl-4-deoxy-4’-demethylpodophyllotoxin)] ether (15)
White amorphous powder; yield 80%; m.p. = 187–188 °C; 1H NMR (500 MHz, CD3OD): δ 7.58 (s, 1H, C14-H), 6.77 (s, 2H, C2'', C6''-H), 6.62 (d, 2H, J = 3.0 Hz, C5''', C8'''-H), 6.36 (s, 2H, C2', C6'-H), 6.20 (d, 1H, J = 2.3 Hz, C6-H), 6.17 (d, 1H, J = 2.3 Hz, C8-H), 6.14 (d, 1H, J = 2.3 Hz, C3-H), 5.96-5.95 (m, 2H, OCH2O), 5.25-5.20 (m, 3H, C2-H, OCH2), 5.13 (s, 1H, C4'''-H), 4.74 (d, 1H, J = 5.0 Hz, C1'''-H), 4.36-4.33 (m, 2H, C11'''-CH2), 3.78 (s, 3H, C7-3OCH), 3.78-3.72 (m, 15H, C3', C4', C5', C3''''', C5''''-OCH3), 3.18 (dd, 1H, J = 4.6, 12.0 Hz, C4-Ha), 3.08-3.06 (m, 1H, C4-Hb), 2.85-2.83 (m, 1H, C3'''-H), 2.82-2.80 (m, 1H, C2'''-H); 13C NMR (125 MHz, CD3OD): δ 176.0 (C-12'''), 161.1 (C-7), 160.6 (C-5), 156.9 (C-9), 154.5 (C-3'''', C-5''''), 150.6 (C-7'''), 149.2 (C-6'''), 148.7 (C-3', C-5'), 137.3 (C-14), 136.9 (C-4''''), 136.0 (C-4'), 136.7 (C-1''''), 135.7 (C-1'), 135.1 (C-9'''), 131.3 (C-10'''), 126.7 (C-13), 111.3 (C-5'''), 109.8 (C-8'''), 109.3 (C-2'''', C-6''''), 105.4 (C-2', C-6'), 103.3 (OCH2O), 102.1 (C-10), 94.7 (C-8), 92.6 (C-6), 80.0 (C-2), 68.9 (OCH2), 67.3 (C-3, C-11'''), 65.7 (C-4'''), 59.8 (4'-OCH3), 56.8 (7-OCH3), 56.7 (3''''-OCH3), 56.6 (5''''-OCH3), 55.9 (3'-OCH3), 55.8 (5'-OCH3), 44.7 (C-1'''), 42.7 (C-2'''), 38.6 (C-3'''), 29.4 (C-4); MS (ESI): m/z = 826 [M + H]+.
Cytotoxicity assays
The cytotoxicity of all the compounds against human myeloid leukemia (HL-60), lung cancer (A-549), breast cancer (MCF-7), colon cancer (SW480), and hepatocellular carcinoma (SMMC-7721) cell lines was measured using the MTT method. 32 Adherent cells (100 μL) were seeded into each well of a 96-well cell culture plate and allowed to adhere for 12 h before drug addition, while suspended cells were seeded just before drug addition, both with an initial density of 1 × 105 cells/mL in 100 μL of medium. Each tumor cell line was exposed to the test compound at various concentrations in triplicate for 48 h. After the incubation, MTT (100 μg) was added to each well, and incubation was continued for 4 h at 37 °C. The cells were lysed with SDS (200 μL) after removal of 100 μL of medium. The optical density of the lysate was measured at 595 nm in a 96-well microtiter plate reader (Bio-Rad 680). IC50 values were calculated by Reed’s method. 33
Molecular docking studies
In this study, the crystal structure of tubulin (PDB ID: 3UT5) 34 and EGFR (PDB ID: 3IKA) 35 was obtained from the in Protein Data Bank (https://www.rcsb.org/). DiscoveryStudio 4.0 software was used for the modeling of the ligand and receptor. AutoDock Tools v1.56 was used for grid and docking according to the literature. 36 Docking parameters were set as the defaults values, except The Number of GA Runs, which was set to 20 and the maximum number of medium was set to 5,000,000 on AutoGrid v4.2 and AutoDock v4.2 (http://autodock.scripps.edu). Docking conformations were classified into different clusters by binding energy, and the cluster with the lowest binding energy was selected. In the selected cluster, conformations with the lowest binding energy and root mean square deviation (RMSD) (< 2.0 Å) were finally chosen to analyze the receptor–ligand interactions.
Supplemental Material
sj-pdf-1-chl-10.1177_17475198211027328 – Supplemental material for Synthesis, cytotoxicity, and molecular docking of methylated (–)-epigallocatechin-3-gallate-4β-triazolopodophyllotoxin derivatives as novel antitumor agents
Supplemental material, sj-pdf-1-chl-10.1177_17475198211027328 for Synthesis, cytotoxicity, and molecular docking of methylated (–)-epigallocatechin-3-gallate-4β-triazolopodophyllotoxin derivatives as novel antitumor agents by Cheng-Ting Zi, Ze-Hao Wang, Jing Shi, Bo-Ya Shi, Ning Zhang, Yi-Long Wu, Yin-Rong Xie, Lu Zhou, Chun Xiao, Xuan-Jun Wang and Jun Sheng in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by grants from the National Nature Science Foundation of China (grant nos 21602196, 31960075, and 31760226), the Science and Technology Project of Yunnan Province (grant nos 2017ZF003 and 2017FG001-046), and the Yunnan Provincial Key Programs of Yunnan Eco-friendly Food International Cooperation Research Center Project (grant nos 2019ZG00904 and 2019ZG00909).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
