Abstract
Without any metal catalyst, an efficient transformation of a variety of sulfonyl hydrazides into the corresponding thiosulfonates mediated by NBS/DABCO under air is developed. The method utilizes mild reaction conditions, affords moderate to good yields of product, and tolerates a broad substrate scope. A plausible mechanism is proposed for the decomposition of the sulfonyl hydrazides and the construction of S(O2)–S bonds to form thiosulfonates.
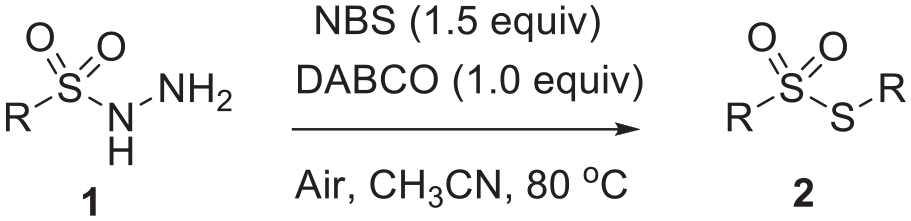
Introduction
Thiosulfonates play key roles in pharmaceuticals and possess rich biological activities such as antimicrobial, antiviral, and fungicidal.1–8 Owing to the wide applications of thiosulfonates, a variety of approaches have been developed for their preparation. The most common approaches for establishing the S–S bonds of thiosulfonates mainly include the oxidation of disulfides/mercaptans9–15 and the sulfuration of sulfinic acid salts.16–19 To synthesize unsymmetrical thiosulfonates, sulfonyl chlorides, sulfinyl chlorides, sulfinates, or sulfonyl hydrazides are commonly reacted with disulfides or mercaptans in the presence of a metal catalyst and an oxidant (Scheme 1(a)).18–22 To synthesize symmetrical thiosulfonates, in the last 15 years, the direct oxidation and coupling of disulfides or mercaptans has been highly favored (Scheme 1(b)).9,23,24 However, many of these methods suffer from harsh reaction conditions involving the use of strong oxidizing agents and toxic catalysts, and give low yields with numerous byproducts. Sulfonyl hydrazides possess a number of advantages such as easily availability, stability, and low sensitivity to air.25–27 In the past 3 years, they have attracted the attention of chemists for the preparation of symmetrical thiosulfonates by cleaving S–N bonds to construct S–S bonds. In 2017, Li et al. 14 reported that sulfonyl hydrazides underwent decomposition and were transformed into thiosulfonates with Pd/ZrO2 as the catalyst and O2 as the oxidant under visible light at ambient temperature for 24 h (Scheme 1(c)). In 2018, Wei et al. 28 showed that thiosulfonates could be obtained from sulfonyl hydrazides using K2S2O8 as a stoichiometric oxidant (Scheme 1(c)). In 2020, Kim et al. 29 reported that sulfonyl hydrazides were transformed to thiosulfonates in the absence of an oxidant at 90 °C for 15 h (Scheme 1(c)). Recently, Lv et al. 30 described that symmetrical/unsymmetrical thiosulfonates could be prepared from sulfonyl hydrazides using versatile heteropoly acid H3PMo12O40 as a catalyst at 90 °C for 3 h in CH3NO2 (Scheme 1(d)). Nevertheless, these methods suffer from environmental and economical concerns as they utilize strong oxidants, transition–metal catalysts, high reaction temperatures, and long reaction times, which impede the applicability of these methodologies. Avoiding these drawbacks, we have successfully developed a procedure to symmetrical thiosulfonates from sulfonyl hydrazides in moderate to good yields (57%–88%) using NBS (N-bromosuccinimide)/DABCO (1,4-diazabicyclo[2.2.2]octane) without any metal catalyst at 80 °C for 2 h under air (Scheme 1(e)).

Preparation of thiosulfonates.
Results and discussion
p-Tolylsulfonyl hydrazide (
Optimization of the reaction conditions for the synthesis of

Reaction conditions:
Isolated yield.
Under N2.
Under O2.
At room temperature for 10 h.
NBS (1.0 equiv.).
NBS (2.0 equiv.).
DABCO (0.5 equiv.).
DABCO (1.5 equiv.).
With optimized conditions in hand, we set out to explore the substrate scope. Several sulfonyl hydrazides with different substituents on the phenyl ring were investigated and the corresponding symmetrical thiosulfonates were obtained in moderate to good yields ranging from 68% to 85% (Table 2, Entries 1–14). The nature of the substituents affected the reaction yields to some degree. Compounds substituted with electron-donating groups (Table 2, Entries 1–3) gave slightly lower yields than those with electron-withdrawing groups (Table 2, Entries 6–8). When electron-donating substituents were attached to the ortho, meta, or para positions of the phenyl ring (Table 2, Entries 2, 9, and 11), the order of the product yields was para > ortho > meta, the same as with electron-withdrawing substituents (Table 2, Entries 6, 10 and 12). Multisubstituted sulfonyl hydrazides gave the corresponding thiosulfonates in good yields (75%–81%) (Table 2, Entries 13 and 14). Moreover, it was noteworthy that naphthyl and benzyl sulfonyl hydrazides also provided the desired products in 76% and 57% yields, respectively, (Table 2, Entries 15 and 16).
Synthesis of thiosulfonates

Confirmed by 1H and 13C NMR spectroscopy.
Isolated yield.
A scale-up experiment (20 mmol) of our synthesis provided an 82% yield of
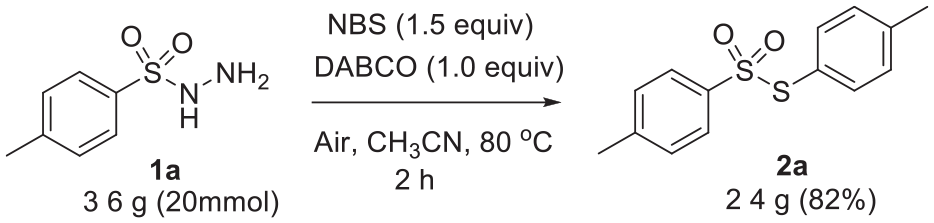
A scale-up experiment.
To further explore the mechanism of the decomposition of the sulfonyl hydrazides and the construction of S(O2)–S bonds to form thiosulfonates, several control experiments were carried out. First, when adding the radical scavenger TEMPO (1.0 equiv.) to the standard reaction, only a trace amount of the target product

Control experiments.
Based on these observations and relevant references,28,31–34 a mechanism can be proposed in Scheme 4. Initially, sulfonyl hydrazide
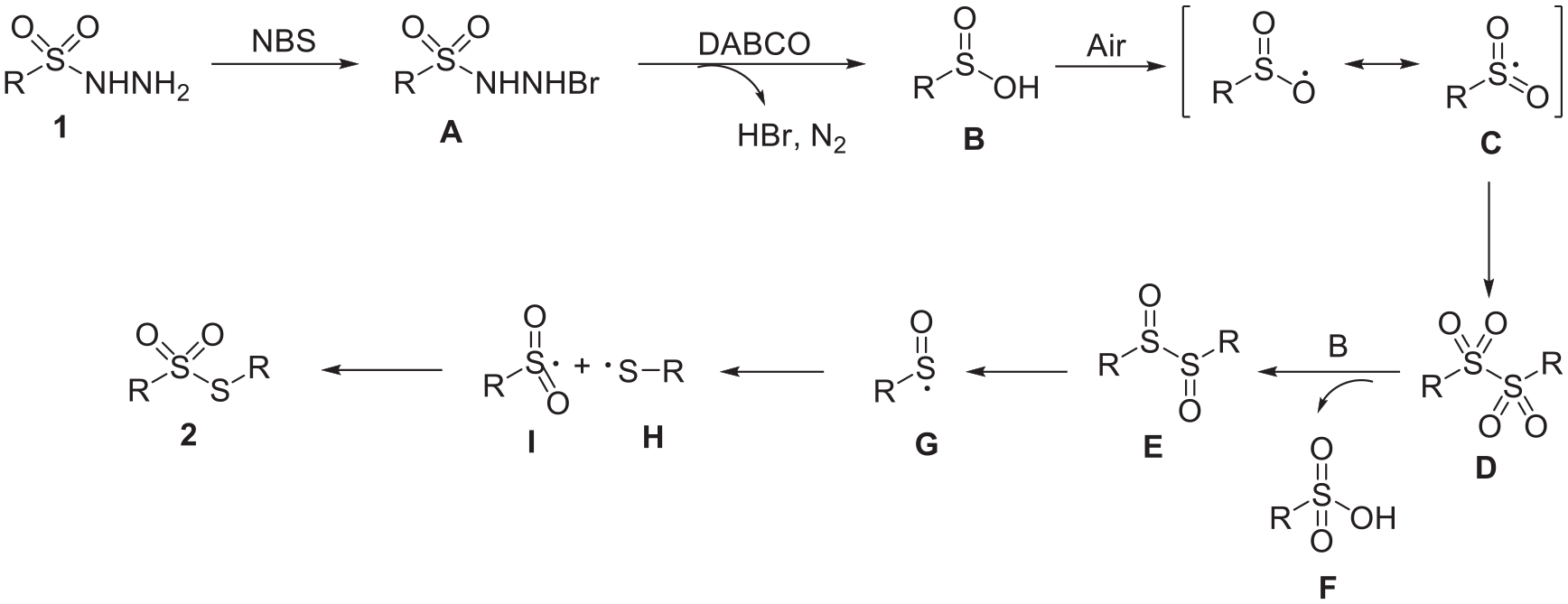
A plausible mechanism for the formation of
Conclusion
In summary, we have developed an efficient approach for the transformation of a variety of sulfonyl hydrazides into the corresponding thiosulfonates mediated by NBS/DABCO under air without any metal catalyst. The approach provides mild reaction conditions, moderate to good yields of products, and a broad substrate scope. A plausible mechanism has been proposed for the decomposition of the sulfonyl hydrazides and the construction of S(O2)–S bonds to form the target thiosulfonates. The reaction is limited to only symmetrical thiosulfonates, which should be as a limitation of the methodology.
Experimental
Infrared spectra were determined on a Nicolet Avatar-370 spectrometer in KBr (ν in cm−1). Melting points were measured on a Büchi B-540 capillary melting point apparatus and were uncorrected. Mass spectra (electrospray ionization mass spectrometry (ESI-MS)) were recorded on a Thermo Finnigan LCQ-Advantage. High-resolution mass spectra (ESI-HRMS) were obtained using an Agilent 6210 TOF instrument. 1H NMR and 13C NMR spectra were recorded on a Varian Mercury Plus-400 spectrometer (400 and 100 MHz), δ in parts per million, J in Hertz, using TMS as the internal standard. Signal multiplicity was assigned as singlet (s), doublet (d), and multiplet (m). All analytical reagents were commercially available and used directly without further purification.
Synthesis of thiosulfonates (2a selected as an example); general procedure
A mixture of p-tolylsulfonyl hydrazide (
S-(p-Tolyl) 4-methylbenzenesulfonothioate
20
(
S-(4-Methoxyphenyl) 4-methoxybenzenesulfonothioate
11
(
S-(4-Ethoxyphenyl) 4-ethoxybenzenesulfonothioate
20
(
S-Phenyl benzenesulfonothioate
12
(
S-(4-Fluorophenyl) 4-fluorobenzenesulfonothioate
12
(
S-(4-Chlorophenyl) 4-chlorobenzenesulfonothioate
12
(
S-(4-Bromophenyl) 4-bromobenzenesulfonothioate
12
(
S-(4-Nitrophenyl) 4-nitrobenzenesulfonothioate
12
(
S-(2-Methylphenyl) 2-methoxybenzenesulfonothioate
11
(
S-(2-Chlorophenyl) 2-chlorobenzenesulfonothioate
33
(
S-(3-Methylphenyl) 3-methylbenzenesulfonothioate
35
(
S-(3-Chlorophenyl) 3-chlorobenzenesulfonothioate
36
(
S-(3-Chloro-2-methylphenyl) 3-chloro-2-methylbenzenesulfonothioate
29
(2
S-(2,4,6-Trimethylphenyl) 2,4,6-trimethylbenzenesulfonothioate
37
(
S-(Naphthalen-1-yl) naphthalene-1-sulfonothioate
38
(
S-(Benzyl) phenylmethanesulfonothioate
12
(
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful to the Traditional Chinese Medicine Research Projects of Health and Family Planning Commission of Jiangxi Province (nos 2018A312 and 2019A228) for the financial support.


