Abstract
A new approach for preparation of phenols from benzaldehydes using Na2S2O8 has been established. This reaction is a Na2S2O8-promoted Baeyer–Villiger oxidation. It provides a highly efficient and practical approach for the preparation of phenols. The protocol is highlighted by its operational simplicity and mild conditions.

Introduction
Phenols and their derivatives are bioactive compounds that are found in natural products, food and medicine. 1 In pursuit of synthetic methods for phenols, two synthetic routes are often involved. One is direct hydroxylation of aryl compounds (Scheme 1(a)).2–5 This route generally suffers from harsh conditions or multi-step reactions for synthesis of the hydroxylation reagent. The Baeyer–Villiger (BV) oxidation and the Dakin reaction are oxygen insertion processes which involve nucleophilic attack on the carbonyl group of aldehydes/ketones by peroxide anion and substituent migration leading to formation of an ester (Scheme 1(b)).6,7 Therefore, phenols can be obtained through hydrolysis of the phenolic ester when employing aromatic aldehydes. Baeyer–Villiger monooxygenases (BVMOs) have shown potential application in biotechnology. 8 However, in the field of chemical synthesis, the BV reaction is still dependent on the use of peracid and even metal catalysts. Although BV reactions using peracid formed in situ from an aldehyde and O2 have been reported, excess aldehyde is necessary. 9

Synthetic methods for phenols: (a) hydroxylation of aryl compounds, (b) Baeyer–Villiger or Dakin oxidation of benzaldehydes and (c) this work.
It is well known that persulfates may decompose to sulfate radical anions and not monopersulfates under heating. 10 So, persulfates would not be expected to perform the BV reaction. However, an unintentional reaction between 2-methoxybenzaldehyde and Na2S2O8 in CH3CN at 55 °C produced 2-methoxyphenol. 11 More importantly, gas chromatography–mass spectrometry (GC-MS) analysis revealed formation of the intermediate 2-methoxyphenyl formate similar to the BV reaction pathway (Scheme 1(c)). To our knowledge, no literature concerning peroxydisulfate-promoted BV reactions has been reported.
Results and discussion
The following investigation continued with 2-methoxybenzaldehyde
Optimization of reaction conditions. a

DMF: dimethylformamide; DCE: 1,2-dichloroethane; THF: tetrahydrofuran.
Reaction conditions: 2-methoxybenzaldehyde (0.2 mmol), oxidant (1.5 equiv., 0.3 mmol), solvent, stirred at 55 °C.
Isolated yields.
CH3CN/H2O (v/v=50:1; 2.0 mL).
Oxone = potassium monopersulfate triple salt.
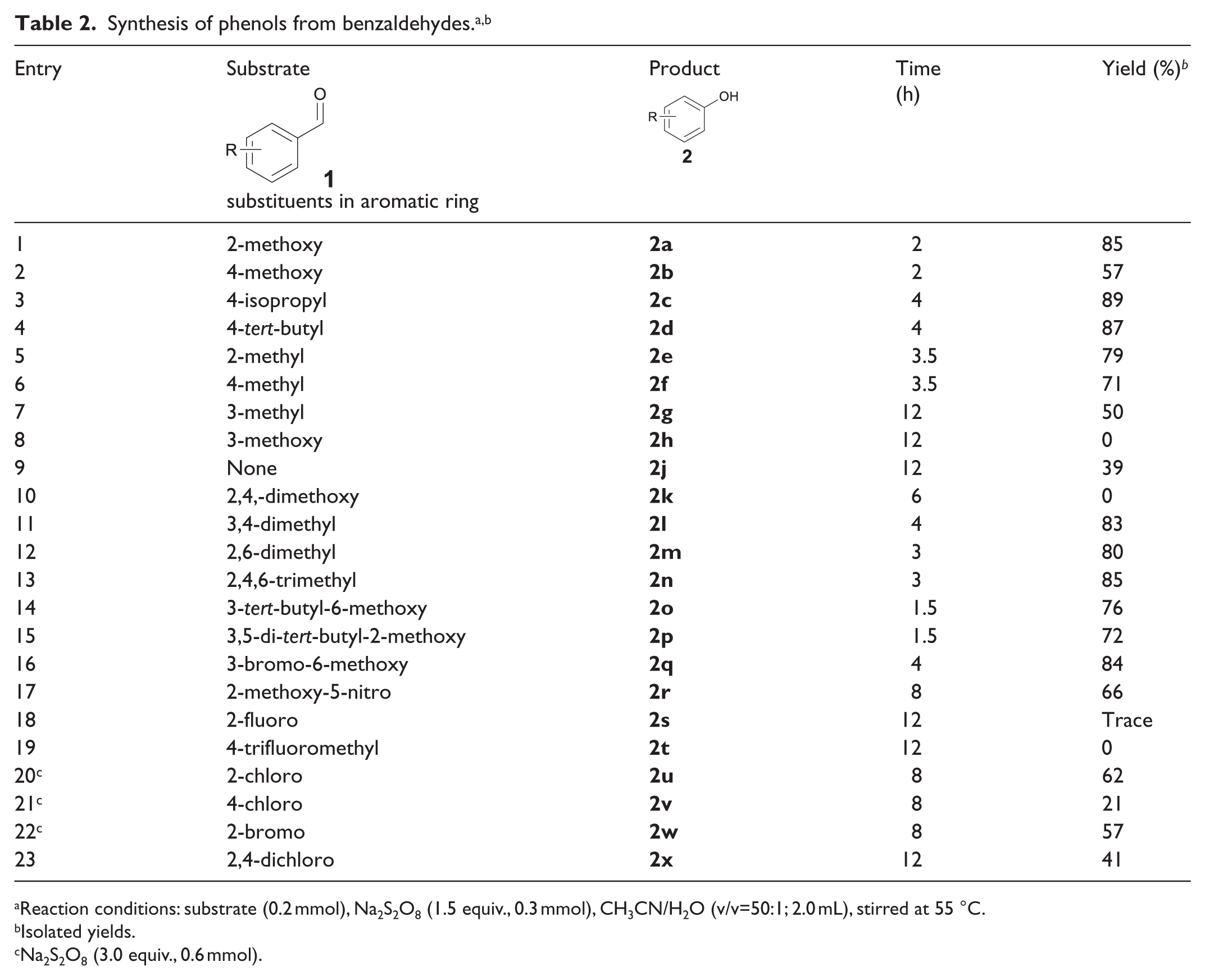
With the optimized conditions in hand, we set out to evaluate the scope of the reaction. The results are listed in Table 2. Substrates bearing electron-donating groups, such as methoxy and alkyl at the para and ortho position of the aryl ring, were well tolerated under standard conditions, providing the corresponding product in 71%–89% yields (Table 2, Entries 1–6). Meta-substituted and unsubstituted benzaldehydes disfavoured this reaction and no or low yields were observed (Table 2, Entries 7–9). Substituents with stronger electron-donating or electron-withdrawing substituents, such as two methoxy, F or CF3, were unfavourable for this reaction (Table 2, Entries 10, 18, 19). More than one alkyl or a combination of alkyl and one methoxy group were compatible with the reaction, forming the products in good yield (Table 2, Entries 11–15). Even a nitro group could be tolerated when there was also a methoxy group on the aryl ring (Table 2, Entry 17). Benzaldehydes substituted by Br and Cl could be converted into phenols by addition of 3.0 equiv. Na2S2O8 (Table 2, Entries 20–23).
Reaction conditions: substrate (0.2 mmol), Na2S2O8 (1.5 equiv., 0.3 mmol), CH3CN/H2O (v/v=50:1; 2.0 mL), stirred at 55 °C.
Isolated yields.
Na2S2O8 (3.0 equiv., 0.6 mmol).
To gain more information about the reaction mechanism, two control experiments were conducted using H218O and under 18O2, respectively. The GC-MS analysis showed that the phenolic oxygen originated from neither H2O nor O2, indicating that Na2S2O8 must be its source (see details in the Supplemental material).
On the basis of the above results and the literature,
12
a plausible mechanism has been proposed in Scheme 2. This is a BV oxidation reaction with peroxysulfate instead of the peracid. It involves nucleophilic attack on the carbonyl group of benzaldehydes

Proposed reaction mechanism.
In conclusion, we have developed a Na2S2O8-promoted BV type oxidation of benzaldehydes to phenols. It provides a highly efficient and practical approach for the preparation of phenols. The method described in this letter is highlighted by its operational simplicity, mild and metal-free conditions without the involvement of acid or base.
Experimental analysis
All reagents were purchased from commercial sources unless otherwise noted. All reactions were monitored by thin-layer chromatography (TLC) and visualized by ultraviolet (UV) lamp (254 nm) or by treatment with a solution of phosphomolybdic acid (10 g) in EtOH (100 mL) followed by heating. Flash column chromatography was performed using 230-400 mesh silica gel. 1H NMR (400/600 MHz) was obtained on Bruker AV-400/600 instrument. GC-MS analysis was performed on a 6890N-5973N/Agilent (electron ionization (EI) mode) instrument.
Oxidation of benzaldehydes to phenols using Na2S2O8 – general procedure
The benzaldehyde
Footnotes
Acknowledgements
The authors are grateful for the financial support from Harbin University of Commerce Youth Innovative Project, Harbin Applied Technology Research and Development Project and the Scientific Research Project in School-level of Harbin University of Commerce.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The study was supported by the Harbin University of Commerce Youth Innovative Project (grant no. 17XN021), Harbin Applied Technology Research and Development Project (grant no. 2014RFQXJ115) and the Scientific Research Project in School-level of Harbin University of Commerce (grant no. 18XN079).
Supplemental material
Supplemental material (Formation of 2-methoxyphenyl formate, 18O experiments and 1H NMR spectra of phenol products) for this article is available online.