Abstract
Four 2-phenylthiazole derivatives are synthesized, characterized, and evaluated as cholinesterase inhibitors. The structures of the 2-phenylthiazole derivatives are confirmed by 1H and 13C nuclear magnetic resonance spectroscopy, single-crystal X-ray diffraction studies, and Hirshfeld surfaces analysis. Hirshfeld surface analysis of the prepared compounds showed C–H···O intermolecular interactions. The cholinesterase inhibition activities of the synthesized compounds are tested by Ellman’s method. [2-(4-Benzyloxyphenyl)-thiazol-4-yl]-(3,5-dimethylpiperidin-1-yl)-methanone showed the best acetylcholinesterase inhibition activity with an IC50 value of 8.86 µM and the best butyrylcholinesterase inhibition activity with an IC50 value of 1.03 µM. A docking study demonstrates that the same compound interacts with the catalytic anionic site and peripheral anionic site of acetylcholinesterase and the catalytic anionic site of butyrylcholinesterase.

Introduction
Alzheimer’s disease (AD) is one of the major diseases threatening the health of the elderly in modern society. AD is a chronic progressive neurodegenerative disease that worsens over time. 1 The common early symptoms are forgetfulness and cognitive dysfunction. As the disease progresses, it can lead to serious language problems, disorientation, loss of self-management, and even gradual loss of physical functions, eventually leading to death. 2 The incidence of AD is progressively increasing across the world due to an aging population. 3 Therefore, it is of great significance to develop effective anti-AD drugs.
Five drugs are currently used to treat AD cognitive dysfunction, four of which are acetylcholinesterase inhibitors (AChEIs): tacrine, 4 donepezil,5,6 galantamine, 7 and rivastimine. 8 Acetylcholinesterase (AChE) is considered as the main factor responsible for acetylcholine (ACh) metabolism, and AChEIs can effectively prevent the hydrolysis of ACh and provide a promising therapeutic effect. 9 In recent decades, the role of butyrylcholinesterase (BuChE) in the development of AD has been clarified. Evidence indicates that BuChE is related to some important AD hallmarks such as the extracellular deposition of the amyloid •-protein(A•) and the aggregation of hyperphosphorylated tau protein. 10 The activity of AChE is decreased during the course of AD progression, while the activity of BuChE is stable or even increased, which is a compensatory feedback of ACh metabolism and cannot be overcome clinically at present. 11 Consequently, the simultaneous inhibition of both enzymes may be a more attractive therapeutic strategy in AD.
Thiazole containing compounds have a variety of biological activities such as antioxidant, anti-inflammatory, antitumor, antiviral, and cholinesterase inhibition activity.
12
Thiazole and its derivatives have attracted continuing interest for the design of various novel central nervous system (CNS) active agents. In the past few decades, thiazoles have been widely used to develop a variety of therapeutic agents against numerous CNS targets.
13
Many thiazole derivatives have been reported to possess cholinesterase inhibitory activities.14–16 For instance, Rahim et al.
17
studied the potential inhibitory activity of 30 thiazole analogues against AChE and BuChE, among which (E)-4-({2-[4-(4-chlorophenyl)thiazol-2-yl]hydrazono}methyl)benzene-1,2-diol (compound

Chemical structures of (E)-4-({2-[4-(4-chlorophenyl)thiazol-2-yl]hydrazono}methyl)benzene-1,2-diol (compound
Previously, several studies with thiazole-derived AChEIs have demonstrated that the thiazole ring primarily interacts with peripheral anionic site (PAS) of AChE and the amine functional moiety interacts with catalytic anionic site (CAS) of AChE.18,19 To find novel dual binding site inhibitors of AChE, we attempted to connect the 2-phenylthiazole moiety (binding to PAS) with amine functional moieties (binding to CAS) to design a series of new 2-phenylthiazole derivatives as dual binding site inhibitors of AChE (Figure 2). In this study, four novel 2-phenylthiazole derivatives have been synthesized and characterized. The intermolecular interactions of these compounds were analyzed through single-crystal X-ray structure studies and Hirshfeld surface analysis. Furthermore, these derivatives were subjected to biological evaluation as potential inhibitors of AChE and BuChE. Finally, molecular docking studies of the most potent inhibitors were carried out to determine the binding interactions with AChE and BuChE, respectively.

Design strategy of the target compounds.
Results and discussion
Spectral characterization
The synthetic route to compounds

Synthesis of 2-phenylthiazole derivatives
The FTIR spectra of the prepared compounds are characterized by a strong broad absorption in the range of 1603–1629 cm–1, which can be assigned to the υ (C=O) stretches. The absorption bands falling in the range of 2810–2971 cm–1 correspond to υ (C–H) of the alkane chain in compounds
Crystal structure of compounds 6a and 6d
The structures of compounds
In vitro inhibition of AChE and BuChE and the selectivity indices of compounds

50% inhibitory concentration (mean ± standard deviation (SD) of three experiments) of AChE from electric eel.
50% inhibitory concentration (mean ± SD of three experiments) of BuChE from equine serum.
Selectivity index for AChE; IC50 BuChE/IC50 AChE

Oak Ridge Thermal Ellipsoid Plot (ORTEP) representation of compound

Packing diagram of compound

ORTEP representation compound

Packing diagram of compound
Hirshfeld surface analysis
Hirshfeld surfaces and fingerprint plots of compounds

Hirshfeld surfaces mapped with dnorm for compounds
The 2D fingerprint plots of the main intermolecular contacts for compounds
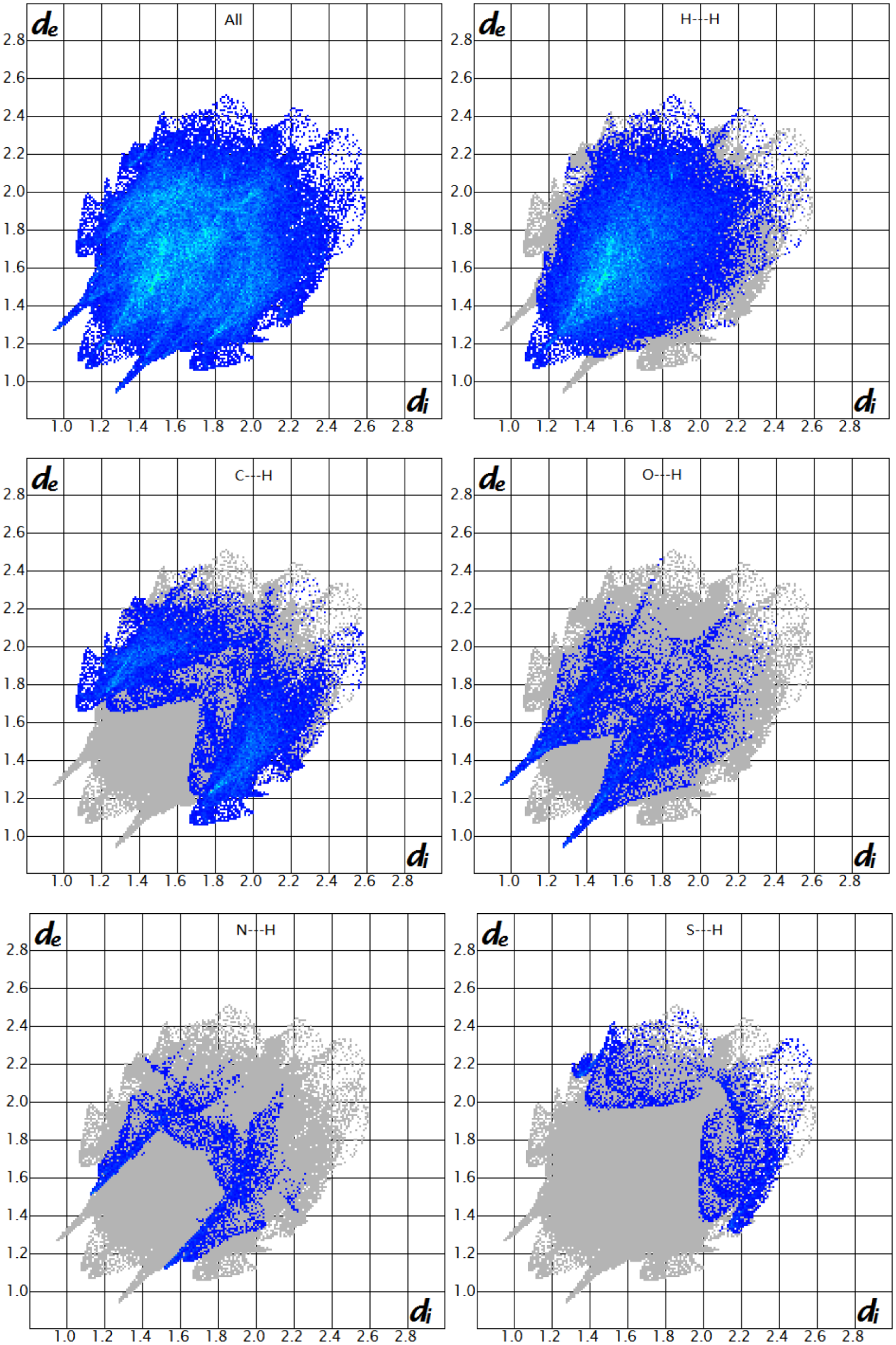
The 2D fingerprint plots of compound

The 2D fingerprint plots of compound
In vitro inhibition studies of cholinesterase
The target compounds
Compound

Dose-dependent inhibition of
Molecular modeling studies
To understand the binding mode of compound

A view of the potential interactions between compound
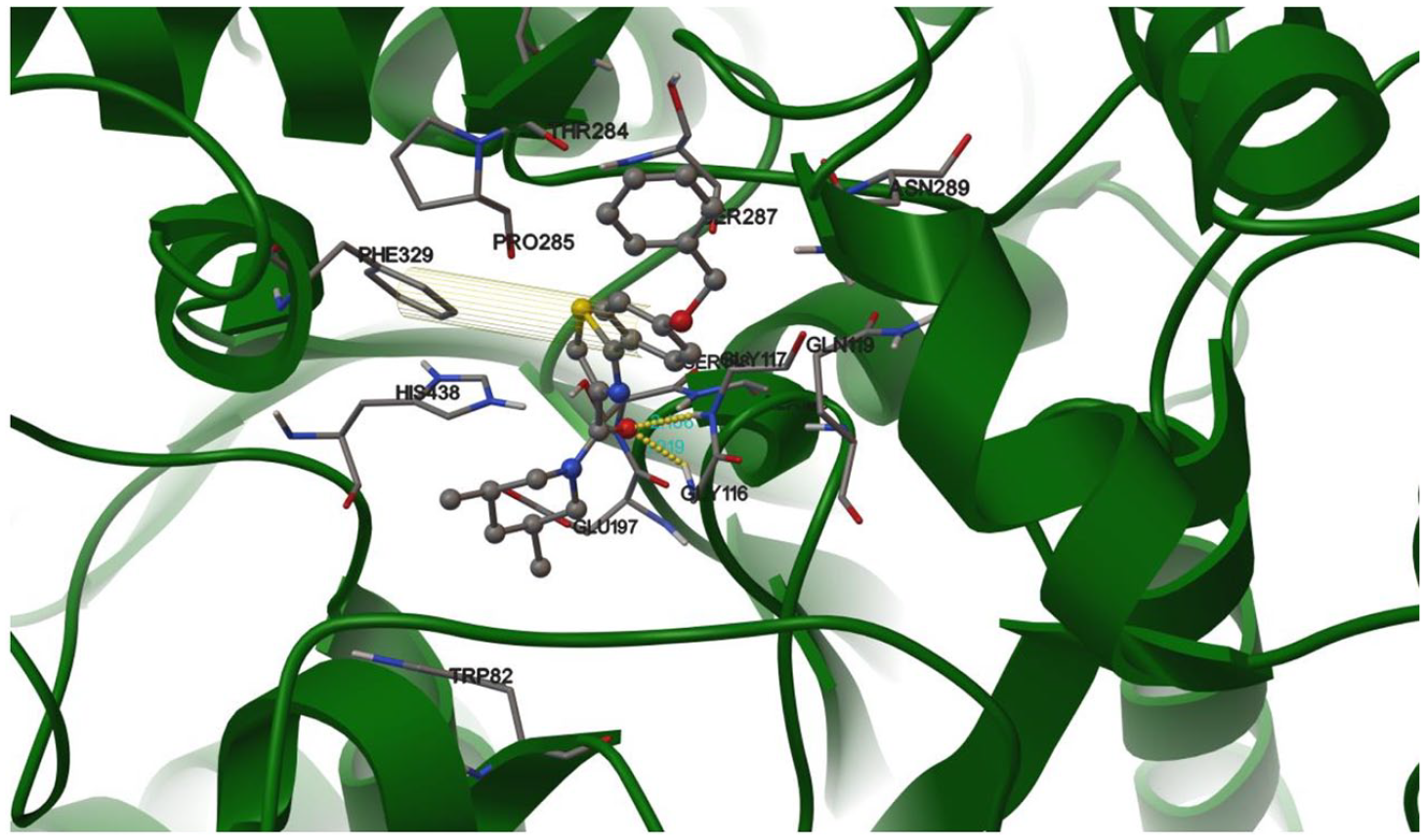
A view of the potential interactions between compound
Conclusion
In this study, four novel 2-phenylthiazole derivatives were designed, synthesized, and evaluated as cholinesterase inhibitors for the treatment of AD. The structures of the four compounds were determined by IR, NMR, HRMS, and X-ray crystallography. Hirshfeld surface analysis and associated fingerprint plots showed that H···H intermolecular contacts are the major contributor to the Hirshfeld surface. The O···H intermolecular contacts were one of the strong contacts due to the presence of C–H···O intermolecular interactions in compounds
Experimental
Materials and methods
All synthetic reagents were purchased from Aladdin Industrial Corporation. All reagents were AR grade and were used without further purification. AChE from electric eel, BuChE from equine serum, 5,5′-dithiobis-(2-nitrobenzoic acid) (DTNB), and butyrylcholine iodide (BTCI) were purchased from Sigma-Aldrich. TLC was performed on glass-backed silica gel sheets (silica gel 60 GF254) and spots were visualized under UV light (254 nm). Melting points were recorded using a Kohler melting point apparatus. 1H NMR and 13C NMR spectra were recorded at 500 MHz using a Bruker Avance III spectrometer. The X-ray single-crystal diffraction data were recorded on a Bruker SMART APEX-II CCD diffractometer. HRMS were performed using an Agilent Technologies 6230 TOF LC/MS instrument.
Synthesis of compounds 6a–d
The target compounds
Yield: 90%. m.p. 78–80 °C. 1H NMR (500 MHz, DMSO-d6): δ 10.11 (s, 1H), 8.44 (s, 1H), 7.80 (m, 2H), 6.90 (m, 2H), 4.33 (q, J = 7.1 Hz, 2H), 1.33 (t, J = 7.1 Hz, 3H). 13C NMR (126 MHz, DMSO-d6): δ 168.0, 160.7, 159.9, 146.5, 128.1, 127.7, 123.6, 115.9, 60.6, 14.1.
A mixture of compound
Yield: 91%. m.p. 111–113 °C. 1H NMR (500 MHz, CDCl3): δ 8.09 (s, 1H), 8.01–7.90 (m, 2H), 7.50–7.29 (m, 5H), 7.08–6.98 (m, 2H), 5.12 (s, 2H), 4.44 (q, J = 7.1 Hz, 2H), 1.43 (t, J = 7.1 Hz, 3H). 13C NMR (126 MHz, CDCl3): δ 168.7, 161.6, 160.8, 147.9, 136.4, 128.5, 128.7, 128.2, 127.5, 126.3, 126.0, 115.2, 70.2, 61.5, 14.4.
Potassium hydroxide (KOH, 0.83 g) was added to a solution of compound
Yield: 89%. m.p. 184–186 °C. 1H NMR (500 MHz, dimethyl sulfoxide (DMSO)-d6): δ 8.41 (s, 1H), 7.91 (d, J = 8.8 Hz, 2H), 7.47 (d, J = 7.2 Hz, 2H), 7.41 (t, J = 7.4 Hz, 2H), 7.34 (t, J = 7.3 Hz, 1H), 7.16 (d, J = 8.9 Hz, 2H), 5.19 (s, 2H). 13C NMR (126 MHz, DMSO-d6): δ 167.7, 162.6, 160.8, 148.4, 137.1, 129.0, 128.5, 128.4, 128.3, 128.2, 126.0, 116.0, 69.94. HRMS (electrospray ionization (ESI)): m/z [M − H]– calcd for C17H12NO3S: 310.0543; found: 310.0533.
Thionyl chloride (0.45 mL) was added to a solution of compound
[2-(4-Benzyloxy-phenyl)-thiazol-4-yl]-(4-methyl-piperazin-1-yl)-methanone (
2-(4-Benzyloxy-phenyl)-thiazol-4-carboxylic acid (2-morpholin-4-yl-ethyl)-amide (
1-[2-(4-Benzyloxy-phenyl)-thiazol-4-carbonyl]-piperidine-4-carboxylic acid ethyl ester (
[2-(4-benzyloxy-phenyl)-thiazol-4-yl]-(3,5-dimethyl-piperidin-1-yl)-methanone (
Single-crystal XRD studies
Crystals of compounds
Supplemental Material
sj-pdf-1-chl-10.1177_1747519820976543 – Supplemental material for Synthesis, characterization, crystal structures, and the biological evaluation of 2-phenylthiazole derivatives as cholinesterase inhibitors
Supplemental material, sj-pdf-1-chl-10.1177_1747519820976543 for Synthesis, characterization, crystal structures, and the biological evaluation of 2-phenylthiazole derivatives as cholinesterase inhibitors by Da-Hua Shi, Meng-qiu Song, Xiao-Dong Ma, Jia-Bin Su, Jing Wang, Xiu-Jun Wang, Yu-Wei Liu, Wei-Wei Liu and Xin-Xin Si in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the financial support from the Natural Science Foundation of Jiangsu Province (No. BK20191470), the National Natural Science Foundation of China (No. 81703557), and a project funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions.
Supplemental material
Supplemental material for this paper is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
