Abstract
In this paper, we report the synthesis of novel 1H-pyrano[2,3-d]pyrimidine-2,4(3H,5H)-dione derivatives

Introduction
Organic synthesis is one of the primary methods for the preparation of chemicals with broad pharmaceutical and photophysical properties. Heterocyclic compounds containing one or more functionalized units are of great significance in daily life because of their pronounced medicinal value. 1 Molecular hybridization is a new approach in rational drug designing and the development of novel molecules or prototypes in which chemical modifications made to existing pharmacophores leads to the new hybrid compounds. These can have reduced toxicity, improved physicochemical properties and biological activities, and most importantly, enhanced specificity towards the particular receptor.2,3
Pyran and its derivatives are six-membered oxygen-containing heterocyclic compounds. Among pyran derivatives, 4H-pyrans constitute a vital structural subunit of biologically active natural products as well as synthetic products. A number of naturally occurring products such as carbohydrates, alkaloids, pheromones and polyether antibiotics have a 4H-pyran core unit.4,5 Fused pyrans and substituted pyrans found in both natural and synthetic compounds constitute an important heteroatomic framework with useful pharmacological activities including antibacterial, antiviral, anticoagulant, anti-anaphylactic, anticancer and diuretic activities and activity towards neurodegenerative disorders.6,7
Pyrimidine and its derivatives are the important class of aza-aromatic scaffolds found in biomolecules such as DNA and RNA. The pyrimidine core skeleton is also found in many living organisms. 8 The synthesis of pyrimidine and condensed pyrimidine molecules has been the subject of substantial interest because of their usefulness in biological and clinical applications.
Diabetes mellitus (DM), a multifactorial chronic syndrome, is characterized by the insufficient release of insulin from the pancreas which regulates glucose homeostasis. 9 DM is of three categories, namely, type I, type II and gestational diabetes among which type II contributes to serious medicinal concerns since it is associated with several life-threatening health problems such as metabolic syndrome, cardiovascular disease, foot ulcers, renal function recession and blindness.10,11 One of the most important therapeutic methods to prevent raising chronic hyperglycaemia is to control the glucose level in blood by inhibiting the activity of digestive enzymes which catalyse the hydrolysis of carbohydrates. 12
Pyrano[2,3-d]pyrimidines, pyrido[2,3-d]pyrimidines or pyrimido[4,5-d]pyrimidines are uracil derivatives and play a distinct and significant role in the field of bio-organic chemistry. Pyrano-pyrimidine derivatives are characterized by the fusion of pyran and pyrimidine rings and the resulting pharmacophores are found to possess abundant biological activities. They form a substantial group of pharmacologically active heterocycles and have been given considerable attention by synthetic and medicinal chemists.13,14 In this study, we have synthesized some 1H-pyrano[2,3-d]pyrimidine-2,4(3H,5H)-dione derivatives by three-component reactions involving 4-hydroxycoumarin, barbituric acid and piperidine (Scheme 1). The synthesized molecules were screened for hyperglycaemic activity.
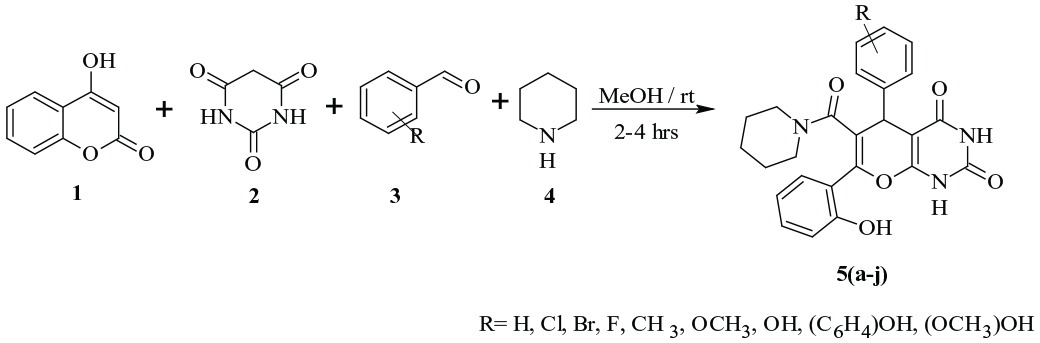
The synthetic strategy used to get the title compounds
Result and discussion
Chemistry
Fused polyheterocycles are invaluable structural motifs in the field of organic chemistry and find application in pharmaceutical and material science. Molecular hybridization facilitates the formation of these fused hybrids and involves the fusion of different pharmacophores in a single framework. The structural modifications stem from the fusion of two or more bioactive scaffolds that impart complexity and more pronounced biological activities to the resulting polycyclic system than that of parent individual heterocycles.
As an initial trial reaction, we performed a multicomponent reaction involving 4-hydroxycoumarin (1 mmol), barbituric acid (1 mmol), piperidine (1 mmol) and benzaldehyde (1 mmol) as a model reaction. The reaction was first carried out by grinding all the substrates in the absence of solvent but the result obtained was not encouraging. The formation of the product was negligible even after prolonged grinding (Table 1, Entry 1). Subsequently, the reaction was carried out using different solvents such as Dichloromethane (DCM), ethyl acetate, acetonitrile, ethanol, methanol, water and a mixture of solvents. When the reaction was carried out using DCM, ethyl acetate, water, acetonitrile, under both reflux and room temperatures, little solid product was formed and thin layer chromatography (TLC) showed the presence of mixtures (Table 1, Entries 2–5).
Optimization of reaction condition.
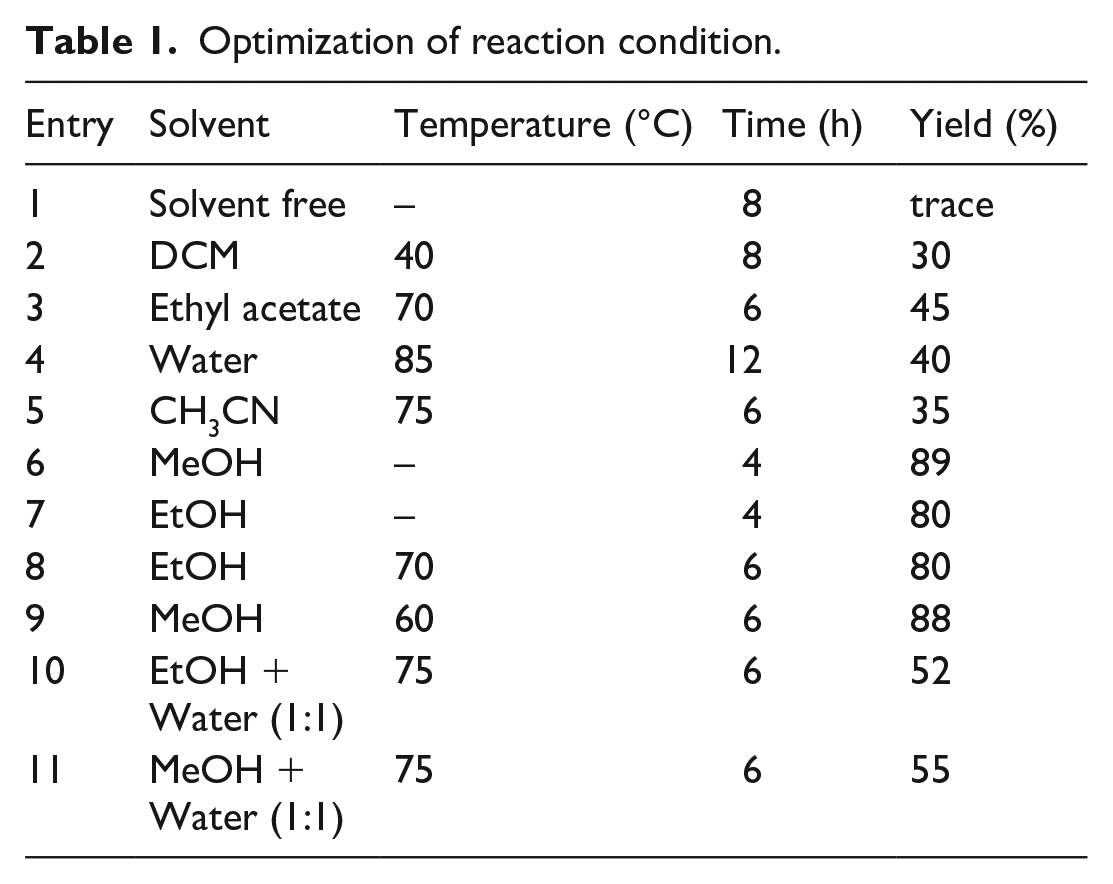
In an endeavour to improve the yield of the product, the same reaction was repeated in ethanol and methanol. Surprisingly, an 89% yield of the product was obtained in methanol at room temperature. This indicated that the nature of the solvent had significant effect on the reaction rate and yield of the product. Among the solvents tested, methanol was found to be the best (Table 1, Entries 6–9). Furthermore, an increase in the temperature did not show any significant improvement in the product yield and reaction rate.
Using this multicomponent reaction, equimolar mixtures of 4-hydroxycoumarin, barbituric acid, piperidine and a substituted aldehyde were used for the construction of coumarin-based pyrano-pyrimidine derivatives. Figure 1 shows a plausible mechanistic pathway for the formation of these novel 1H-pyrano[2,3-d]pyrimidine-2,4(3H,5H)-diones. This may involve a Knoevenagel condensation of the aldehyde and barbituric acid in the presence of piperidine followed by the nucleophilic attack of the 4-hydroxycoumarin with dehydration followed by ring-opening of coumarin nucleus by piperidine leading to the target compound.

Plausible mechanistic pathway for the formation of 1H-pyrano [2,3-d] pyrimidine-2,4(3H,5H)-dione derivatives.
The structures of all the newly synthesized compounds were characterized by spectroscopic techniques. The Fourier transform infrared (FTIR) spectrum of compound
Physicochemical data of synthesized compounds

Pharmacology
Most of the body’s physiological functions are regulated by specific enzymes, but the malfunctioning of these enzymes can be associated with many life-threatening diseases. Thus, to maintain the body equilibrium condition, the control/inhibition of these enzymes is necessary. Recently, effective contributions have been made by researchers in the field of drug discovery by inventing potent enzyme inhibitors from both natural and synthetic sources. α-Amylase and α-glucosidase are the catabolic enzymes that catalyse the hydrolysis of carbohydrates as our intestine can only absorb simple sugars. But the overexpression of these enzymes results in the inadequate production of insulin due to the increased blood glucose level. Thus, inhibitors of these enzymes are useful in the treatment of postprandial hyperglycaemia. Coumarin and its derivatives exhibit many therapeutic applications and most of the reports suggest that the coumarin nucleus is proficient in improving insulin resistance and increasing glucose uptake. The α-amylase and α-glucosidase inhibitory activities of both natural and synthetic coumarin derivatives have been evaluated. In this study, the novel 1H-pyrano[2,3-d]pyrimidine-2,4(3H,5H)-dione derivatives
In vitro α-amylase inhibitory activity
The α-amylase inhibitory potencies of the synthesized compounds
Relative α-amylase enzyme activity values (%) at different concentrations and IC50 values.

Acarbose is standard inhibitor for α-amylase and the IC50 = 0.2137 mM (at five different concentrations).
The variation in the activity exhibited by the compounds in this series may be attributed to the substitution pattern on the phenyl rings even though the core skeleton is same for all the compounds. The highest potency exhibited by the compound
The difference in the activity profile shown by the compounds with the same functional group on the aromatic ring may be due to the position of substituents. If we compare compounds
In vitro α-glucosidase inhibitory activity
The in vitro α-glucosidase assay was performed to study the inhibition efficiency of the synthesized molecules on the α-glucosidase enzyme. The assay is based on the mechanism of hydrolysis of PNPG (p-nitrophenyl-

The increase in the intensity of the yellow colour developed after the reaction indicated the extent of hydrolysis of the complex sugar to the simple sugars. The inhibitory concentration of the title compounds
Overall, we observed that the synthesized targets are more capable of inhibiting the activity of α-glucosidase than the α-amylase enzyme. The hydroxyl substituted analogues showed potent activity in both assays; this may be due to the interaction of phenols with the protein and leading to the inhibition of enzyme activity. Tables 3 and 4 and Figures 2 and 3 showed the in vitro α-amylase and α-glucosidase inhibitory activity of compounds
Relative α-glucosidase enzyme activity values (%) at different concentrations and IC50 values.
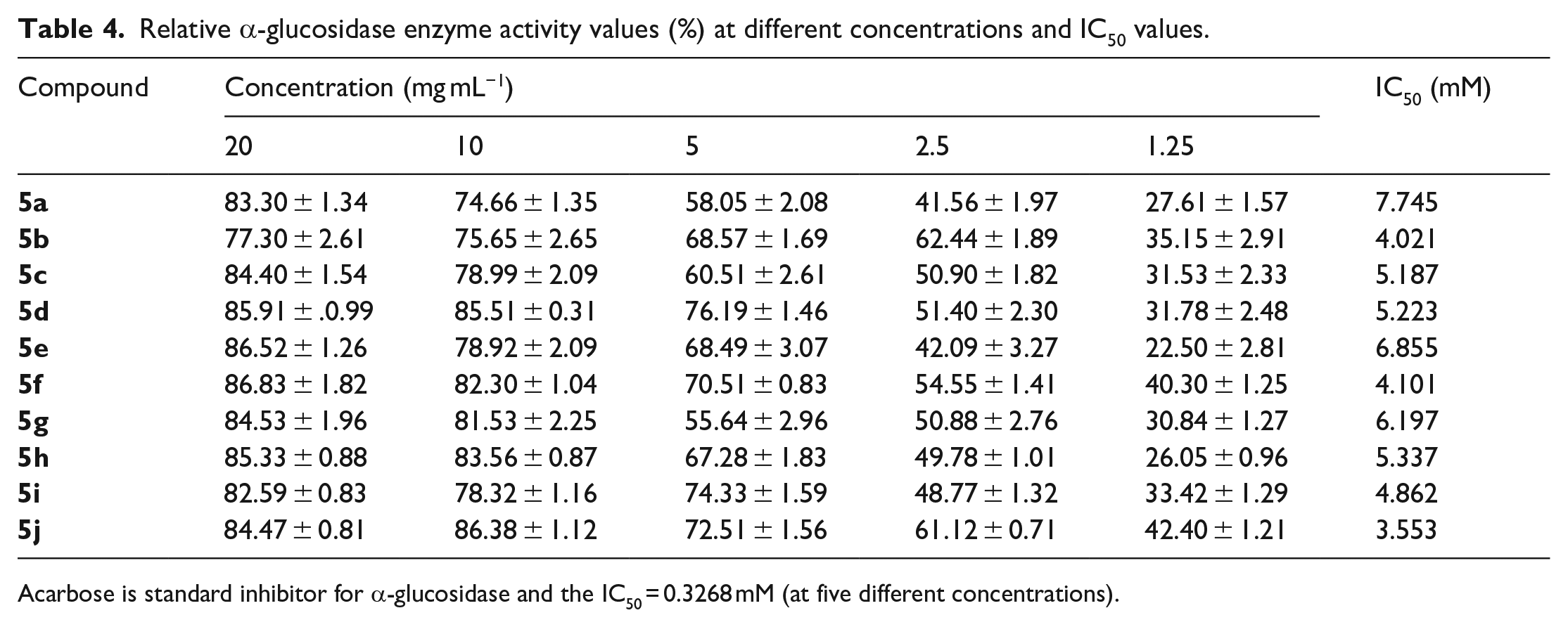
Acarbose is standard inhibitor for α-glucosidase and the IC50 = 0.3268 mM (at five different concentrations).

In vitro α-amylase inhibitory activity of the synthesized compounds

In vitro α-glucosidase inhibitory activity of the synthesized compounds
Conclusion
We have synthesized 10 pyrano-pyrimidine derivatives using multicomponent reaction via a Knoevenagel condensation. The biological evolution of these compounds revealed that all the compounds are active against both digestive enzymes (α-amylase and α-glucosidase). Particularly, the compounds with hydroxyl substitution on the aromatic ring showed better potential with the least IC50 value when compared to other electron-donating groups. On the contrary, the substitution pattern on the phenyl ring also decides the inhibition efficacy of the synthesized targets. Overall, by comparing the IC50 values from both the assays, it was concluded that the synthesized compounds are most potent in inhibiting α-glucosidase enzyme activity than α-amylase.
Experimental section
Materials and methods
The enzymes α-glucosidase from Saccharomyces cerevisiae (Km = 0.55–2.7 µM), Porcine pancreatic α-amylase (Type VI-B, Km = 0.000624–3.05 µM), p-nitrophenyl-α-
General procedure for the synthesis of 1Hpyrano[2,3-d]pyrimidine-2,4(3H,5H)-dione derivatives 5(a–j)
An equimolar mixture of 4-hydroxy coumarin (1 mmol), barbituric acid (1 mmol), piperidine (1 mmol) and the substituted aldehyde (1 mmol) in methanol (10 mL) was charged in 100-mL round-bottomed flask and the resulting homogeneous mixture was magnetically stirred at room temperature for about 2–4 h. The progress of the reaction was monitored by TLC. After completion of the reaction, the precipitated product was collected by filtration, washed thoroughly with methanol and then with water and finally dried to afford pure product
7-(2-Hydroxyphenyl)-5-phenyl-6-(piperidine-1-carbonyl)-1H-pyrano[2,3-d]pyrimidine-2,4(3H,5H)-dione (
5-(4-Chlorophenyl)-7-(2-hydroxyphenyl)-6-(piperidine-1-carbonyl)-1H-pyrano[2,3-d]pyrimidine-2,4(3H,5H)-dione (
5-(4-Bromophenyl)-7-(2-hydroxyphenyl)-6-(piperidine-1-carbonyl)-1H-pyrano[2,3-d]pyrimidine-2,4(3H,5H)-dione (
5-(4-Fluorophenyl)-7-(2-hydroxyphenyl)-6-(piperidine-1-carbonyl)-1H-pyrano[2,3-d]pyrimidine-2,4(3H,5H)-dione (
7-(2-Hydroxyphenyl)-6-(piperidine-1-carbonyl)-5-(p-tolyl)-1H-pyrano[2,3-d]pyrimidine-2,4(3H,5H)-dione (
7-(2-Hydroxyphenyl)-5-(4-methoxyphenyl)-6-(piperidine-1-carbonyl)-1H-pyrano[2,3-d]pyrimidine-2,4(3H,5H)-dione (
5,7-Bis(2-hydroxyphenyl)-6-(piperidine-1-carbonyl)-1H-pyrano[2,3-d]pyrimidine-2,4(3H,5H)-dione (
5-(3-Hydroxynaphthalen-2-yl)-7-(2-hydroxyphenyl)-6-(piperidine-1-carbonyl)-1H-pyrano[2,3-d]pyrimidine-2,4(3H,5H)-dione (
5-(4-Hydroxy-3-methoxyphenyl)-7-(2-hydroxyphenyl)-6-(piperidine-1-carbonyl)-1H-pyrano[2,3-d]pyrimidine-2,4(3H,5H)-dione (
7-(2-Hydroxyphenyl)-5-(4-hydroxyphenyl)-6-(piperidine-1-carbonyl)-1H-pyrano[2,3-d]pyrimidine-2,4(3H,5H)-dione (
Biological studies
In vitro α-amylase inhibitory assay
α-Amylase inhibitory activity was established in accordance with the earlier reported method with slight modification. 15 The test samples were prepared by dissolving synthesized compounds at different concentrations (20, 10, 5, 2.5, 1.25 and 0.625 mg mL−1) in DMSO. A volume of 40 µL of sample and 40 µL of the α-amylase solution in 0.02 M sodium phosphate buffer (pH 6.9 with 0.006 M sodium chloride) was incubated at 25 °C for 10 min. After pre-incubation, 40 µL 1% starch solution in 0.02 M sodium phosphate buffer (pH 6.9 with 0.006 M sodium chloride) was added as substrate. The resultant reaction mixture was incubated at 25 °C for 10 min, then dinitrosalicylic acid as colour reagent was added and the test tubes were incubated in a boiling water bath for about 5 min and then cooled at room temperature. The reaction mixture was diluted by adding 900 µL of distilled water and the contents were mixed properly. 50 µL of reaction mixture was taken from the test tube and loaded to 96-well microplate and the absorbance was measured at 405 nm using multiskan sky (Thermo Scientific, USA). Acarbose was used as a reference standard and all experiments were carried out in triplicates. Percent inhibition was calculated using the following equation

In vitro α-glucosidase inhibitory assay
The α-glucosidase inhibition activity of the synthesized compounds was determined using the assay described by Sancheti et al., with slight modification.
16
Different concentrations of (20, 10, 5, 2.5 and 1.25 mg mL−1) synthesized compounds were prepared by dissolving in DMSO. An aliquot of 20 µL of the test sample was added to the 50 μg mL−1 α-glucosidase solution followed by the addition of 60 µL in phosphate buffer solution (pH 6.8) into the 96-well plate. The contents were mixed and incubated for 5 min and then 10 μL of 10 mM ρ-nitrophenyl-α-

Statistical analysis
The experiments were performed in triplicates and the results of the bioassays were expressed as mean ± SE and the IC50 value was obtained by nonlinear regression analysis using GraphPad Prism software.
Supplemental Material
sj-pdf-1-chl-10.1177_1747519820964048 – Supplemental material for An efficient multicomponent synthesis of 1H-pyrano[2,3-d]pyrimidine-2,4(3H,5H)-dione derivatives and evaluation of their α-amylase and α-glucosidase inhibitory activity
Supplemental material, sj-pdf-1-chl-10.1177_1747519820964048 for An efficient multicomponent synthesis of 1H-pyrano[2,3-d]pyrimidine-2,4(3H,5H)-dione derivatives and evaluation of their α-amylase and α-glucosidase inhibitory activity by Mamata Devendra Naik, Yadav D Bodke, Prashantha J and Jayanth K Naik in Journal of Chemical Research
Footnotes
Acknowledgements
One of the authors would like to acknowledge the Chairman, Department of Industrial Chemistry, Kuvempu University, Shankaraghatta, for providing laboratory facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
