Abstract
A synthesis of benzo[e][1,2,4]triazines and 1,2,4-triazolospiro[4,5]deca-2,6,9-trien-8-ones has been developed from reactions of amidrazones with 2-chloro-1,4-benzoquinone in EtOAc containing 0.5 mL of piperidine. This highly regioselective and one-pot process provided rapid access to 1,2,4-triazolospiro[4,5]deca-2,6,9-trien-8-ones (60%–70%) and benzo[e][1,2,4]triazines (11%–18%). On reacting amidrazones with 5-hydroxy-1,4-naphthoquinone in an EtOAc/piperidine mixture, the reaction proceeded to give 5-hydroxy-2-(piperidin-1-yl)naphthalene-1,4-dione. The structures of the isolated products were proved by infrared, NMR (2D-NMR), mass spectra, and elemental analyses in addition to X-ray structure analysis. The reaction mechanisms are discussed. The anticancer screening of selected compounds showed broad-spectrum anticancer activity against most melanoma cancer cell lines, ovarian cancer OVCAR-3, central nervous system cancer SF-295 and U251, non-small cell lung cancer NCI-H23, renal cancer SN12C, and colon cancer HCT-15 and HCT-116. The selected compounds exhibited moderate to weak anticancer activity to other cell lines.
Keywords

Introduction
Amidrazones display fungistatic, bacteriostatic, and antimycotic activity. 1 In addition, they have herbicidal 1 and lipoxygenase-1 inhibitory activity. 2 Aly et al. 3 reported that amidrazones reacted with 1,4-benzoquinone or 1,4-naphthoquinone to give, in a few minutes, benzo- and naphtho-1,2,4-triazin-6(4H)-ones. The reactions of amidrazones with 2,3,5,6-tetrachloro-1,4-benzoquinone and 2,3-dichloro-1,4-naphthoquinone in dry DMF proceed to give indazole derivatives. 3 The synthesis of various 4-aryl-5-imino-3-phenyl-1H-naphtho[2,3-f]-1,2,4-triazepine-6,11-diones has also been reported 4 by the reaction between amidrazones and 1,4-dioxo-1,4-dihydronaphthalene-2,3-dicarbonitrile, while 1,4-diphenyl-2-arylamino-2-{[1-phenylmeth-(Z)-ylidene]-hydrazino}butane-1,4-diones were obtained from the reaction of amidrazones with 1,4-diphenyl-2-butyne-1,4-dione. 5
Quinones are widely distributed in the natural world, 6 being found in bacteria, plants, and arthropods and are ubiquitous to living systems. Quinones play pivotal roles in biological functions including oxidative phosphorylation and electron transfer. 7 They also have important roles as electron transfer agents in primary metabolic processes like photosynthesis and respiration which is vital to human life. 7 A large number of chemicals with 1,4-benzoquinone as the basic subunit exhibit prominent pharmacological applications such as antibiotics,8,9 antitumor,10–14 antimalarial 13 and anticoagulant compounds. 15 Juglone itself shows effective anticancer activity.16,17 However, 2-chlorocyclohexa-2,5-diene-1,4-dione (CBQ) and 2-chloro-1,4-benzoquinone are common metabolites of polycyclic aromatic hydrocarbons generated through industrial processes. Chlorobenzoquinones have remarkable effects on DNA, and a few studies are available regarding chlorobenzoquinone-induced protein modifications.18–20 Drugs containing quinone moieties are represented by anthracyclines, anthraquinones, mitomycin C, and streptonigrin. 20 Doxorubicin has been used clinically to treat solid tumors 21 and acute lymphoblastic and myeloblastic leukemia.22–24 Anti-neoplastic agents such as actinomycin and streptonigrin as well as antibiotics such as mitamycin 25 (Figure 1) and rifamycin 26 have aminoquinone moieties in their structures. 1,4-Dioxo-3-(phenylamino)-1,4-dihydronaphthalene-2-carboxylic acid (Figure 1) shows potent cytostatic effects against both renal and melanoma cancer cell lines. Against renal cell lines, the activity (GI50 = 8.38 μM) was nearly as good as that of the anticancer agent, etoposide (GI50 = 7.19 μM).

Representative quinone-based anticancer agents.
A series of naphthoquinone derivatives showed proteasome inhibitor activity against PI-083 (Figure 1).27–30 In addition, they also exhibited high selectivity (twofold to fourfold more selective) for cancer cells over normal cells (L-929, IC50 = 2.85 μM). The activity of 3-(4-(1-hydroxycyclohexyl)-1H-1,2,3-triazol-1-yl)-2,2-dimethyl-2,3-dihydronaphtho[1,2-b]furan-4,5-dione against melanoma cells was comparable to that of the initial quinones, nor-ß-lapachone and β-lapachone, and better than that of doxorubicin 31 (Figure 1).
Moreover, 3-amino-1,2,4-benzotriazine-1,4-dioxides have received considerable attention as a class of antitumor agent. 32 Triazoles can easily bind with a variety of enzymes and receptors in biological systems via diverse non-covalent interactions. 33 Therefore, many triazole derivatives serve as medicinal drugs. 34 More specifically, compounds having the 1,2,4-triazole moiety have shown various biological activities, such as antifungal, 35 antimicrobial, 36 antitubercular, 37 anticancer, 38 anticonvulsant, 39 hypoglycemic, 40 anti-inflammatory, and analgesic activities. 41 In this paper, we report a new straightforward reaction of amidrazones with electron-deficient naturally occurring quinones, and the new compounds with a quinone nucleus were screened for their anticancer activity.
Results and discussion
Equimolar amounts of an amidrazone

Reaction between amidrazones
Methods and corresponding yields of compounds

Method
Elemental analysis and mass spectroscopy of compound
The regioselectivity of the formation of compounds

Plausible mechanism showing the formation of compound
The structure of
Detailed NMR spectral data of compound
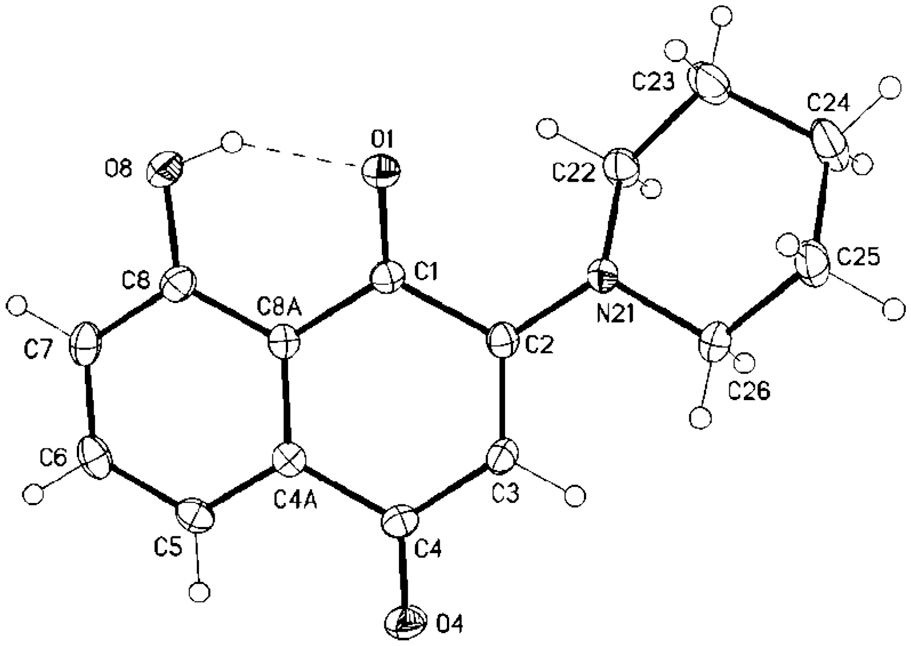
Molecular structure of compound
Biological evaluation
Screening of the in vitro anticancer activity
Among the synthesized compounds, NCI selected compounds
Anticancer activity of compounds

CNS: central nervous system.
In vitro five-dose full NCI 60 cell panel assay
Compounds
Results of in vitro five-dose testing of nine human cancer types and selectivity for compound

MIDa = 2.905; selectivity ratio = MIDa/MIDb.

Five-dose diagram of compound
Conclusion
Herein, we report reactions of amidrazones with two naturally occurring quinones, namely, 2-chloro-1,4-benzoquinone and 5-hydroxy-1,4-napthalene-1,4-dione (Juglone). Spiro-triazoles and 1,2,4-triazines were obtained from the reactions of amidrazones with the aforementioned quinones. The screening results were encouraging and promising, but further studies of their anticancer activities should be continued.
Experimental
Melting points were determined using open glass capillaries on a Gallenkamp melting point apparatus (Weiss Gallenkamp, Loughborough, UK), and they are uncorrected. The IR spectra were recorded from potassium bromide disks with an FT device, and Minia University NMR spectra were measured on a Bruker AV-400 spectrometer (400 MHz for 1H, 100 MHz for 13C, and 40.55 MHz for 15N); chemical shifts are expressed in δ (ppm) versus internal tetramethylsilane (TMS) = 0 for 1H and 13C, and external liquid ammonia = 0 for 15N. Coupling constants are stated in Hz. Correlations were established using 1H–1H COSY, and 1H–13C and 1H–15N HSQC and HMBC experiments. Mass spectra were recorded on a Finnigan Fab 70 eV, Institute of Organic Chemistry, Karlsruhe University, Karlsruhe, Germany. TLC was performed on analytical Merck 9385 silica aluminum sheets (Kieselgel 60) with Pf254 indicator; TLCs were viewed at λmax = 254 nm. Elemental analyses were carried out at National Research Center, Al Doki, Egypt.
Starting materials
Amidrazones
Reactions of amidrazones 1a–d with 2-chloro-1,4-benzoquinone (2 )
Equimolar mixtures of amidrazones
6-Chloro-3,4-diphenyl-1,2,4-triazaspiro[4.5]deca-2,6,9-trien-8-one (3a )
Red solid (EtOH), yield: 0.218 g (65%), m.p. 115–117 °C. IR (KBr): ν = 3260 (NH), 3136 (Ar–CH), 1702 (C=O), 1614 (C=N), 1599 cm−1 (Ar–C=C). 1H NMR (400 MHz, DMSO-d6): δH = 8.83 (s, 1H, NH), 8.20-8.15 (m, 2H), 7.80-7.75 (m, 2H, Ar–CH), 7.60-6.72 (m, 8H, Ar–CH), 6.60 (d, 1H, J = 0.7 Hz, Ar–CH). 13C NMR (100 MHz, DMSO-d6): δC = 169.0 (C=O), 163.0 (C=N), 141.8, 134.0, 130.6 (Ar–C), 130.3, 130.0 (Ar–CH), 129.7, 129.4, 127.9, 126.4 (Ar–2CH), 126.3, 125.5, 122.6 (Ar–CH), 78.1 (Spiro-C). MS: m/z (%) = 337 (6), 335.8 (27), 256.0 (42), 225.3 (47), 185 (30), 183.6 (100), 154.0 (58), 149.2 (47), 114.1 (15), 81.0 (39), 77.1 (93). Anal. calcd for C19H14ClN3O (335.79): C, 67.96; H, 4.20; Cl, 10.56; N, 12.51; found: C, 68.05; H, 4.08; Cl, 10.70; N, 12.60.
6-Chloro-4-(4ʹ-chlorophenyl)-3-phenyl-1,2,4-triazaspiro[4.5]deca-2,6,9-trien-8-one (3b )
Red solid (EtOH), yield: 0.222 g (60%), m.p. 204–206 °C. IR (KBr): ν = 3262 (NH), 3135 (Ar–CH), 1701 (C=O), 1613 (C=N), 1548 cm−1 (Ar–C=C). 1H NMR (400 MHz, DMSO-d6): δH = 8.82 (s, 1H, NH), 8.40-8.35 (m, 2H, Ar–CH), 8.25-8.22 (m, 2H, Ar–CH), 7.62-7.52 (m, 7H, Ar–CH), 7.42 (d, 1H, J = 0.8 Hz, Ar–CH). 13C NMR (100 MHz, DMSO-d6): δC = 169.2 (C=O), 161.0 (C=N), 141.17, 138.49, 132.48, 130.49 (Ar–C), 130.22 (Ar–CH), 129.57, 129.46, 129.12, 128.89 (Ar–2CH), 125.72, 125.65, 125.19 (Ar–CH), 78.12 (Spiro-C). MS: m/z (%) = 370.6 (63), 372.6 (42), 336.0 (14), 296.1 (18), 294.1 (59), 261.1 (6), 259.1 (26), 236.0 (36), 225.3 (47), 185.6 (29), 154.0 (59), 149.2 (47), 114.2 (4), 112.2 (14), 81.1 (39), 77.1 (93). Anal. calcd for C19H13Cl2N3O (370.23): C, 61.64; H, 3.54; N, 11.35; Cl, 19.15; found: C, 61.52; H, 3.43; Cl, 19.03; N, 11.13.
6-Chloro-4-(3ʹ-chlorophenyl)-3-phenyl-1,2,4-triazaspiro[4.5]deca-2,6,9-trien-8-one (3c )
Red solid (CH3OH), yield: 0226 g (61%), m.p. 174–176 °C. IR (KBr): ν = 3269 (NH), 3130 (Ar–CH), 1705 (C=O), 1616 (C=N), 1548 cm−1 (Ar–C=C). 1H NMR (400 MHz, DMSO-d6): δH = 8.73 (s, 1H, NH), 8.35 (dd, 2H, J = 1.2, 0.8 Hz, Ar–CH), 8.13-7.95 (m, 2H, Ar–CH), 7.53-7.35 (m, 7H, Ar–CH), 7.33 (d, 1H, J = 0.7 Hz, Ar–CH). 13C NMR (100 MHz, DMSO-d6): δC = 169.7 (C=O), 161.8 (C=N), 141.14, 140.7, 135.8, 132.59 (Ar–C), 131.4 (Ar–CH), 131.17, 130.96, 130.45, 129.58 (Ar–2CH), 126.28, 125.22, 124.34 (Ar–CH), 78.23 (Spiro-C). MS: m/z (%) = 372.2 (16), 370.2 (53), 336.0 (15), 296.1 (20). 294.1 (62), 261.1 (9), 259.1 (27), 236.0 (37), 225.3 (47), 185.0 (100), 154.0 (60), 149.2 (47), 114.0 (7), 112.0 (26), 81.1 (39), 77.1 (70). Anal. calcd for C19H13Cl2N3O (370.23): C, 61.64; H, 3.54; Cl, 19.15; N, 11.35; found: C, 61.72; H, 3.60; Cl, 19.25; N, 11.20.
6-Chloro-3-phenyl-4(4ʹ-methoxyphenyl)-1,2,4-triazaspiro[4.5]deca-2,6,9-trien-8-one (3d )
Red solid (CH3CN) 245 mg (67%), m.p. 254–256 C. IR (KBr): ν = 3260 (NH), 3140 (Ar–CH), 1700 (C=O), 1610 (C=N), 1550 (C=C), 1110 cm−1 (OCH3). 1H NMR (400 MHz, DMSO-d6): δH = 8.90 (s, 1H, NH), 8.40 (dd, 2H, J = 1.2, 0.7 Hz, Ar–CH), 8.25-8.20 (m, 3H, Ar–CH), 7.52-7.45 (m, 5H, Ar–CH), 6.60 (dd, 2H, J = 0.8 Hz, Ar–CH), 3.90 (s, 3H, OCH3). 13C NMR (100 MHz, DMSO-d6): δC = 169.2 (C=O), 161.0 (C=N), 150.1 (Ar–C–OCH3), 138.2, 132.0, 130.3 (Ar–C), 129.2 (Ar–CH), 128.1, 127.5, 127.1, 126.8 (Ar–2CH), 125.7, 123.6, 122.2 (Ar–CH), 78.1 (Spiro-C), 57.6 (OCH3). MS: m/z (%) = 367 (13), 365.0 (40), 2960 (20), 184.6 (19), 154.0 (60), 121.0 (100), 77.0 (60). Anal. calcd for C20H16ClN3O2 (365.81): C, 65.67; H, 4.41; N, 11.49; Cl, 9.69; found: C, 65.72; H, 4.43; N, 11.30; Cl, 9.60.
3,4-Diphenyl-1,4-dihydrobenzo[e][1,2,4]triazin-6-ol (4a )
White solid (CH3CN), yield: 0.045 g (15%), m.p. 302–304 °C. IR (KBr): ν = 3424 (OH), 3264 (NH), 1631 (C=N), 1590 cm−1 (Ar–C=C). 1H NMR (400 MHz, DMSO-d6): δH = 10.12 (s, 1H, OH), 8.54 (s, 1H, NH), 7.48-7.13 (m, 13H, Ar–CH). 13C NMR (100 MHz, DMSO-d6): δC = 158.2 (Ar–C–OH), 154.0 (C=N), 132.0, 130.8, 130.0, 129.0 (Ar–C), 128.6, 128.4, 127.8, 127.4, 127.0 (Ar–2CH), 126.9, 126.5, 126.0 (Ar–CH). MS: m/z (%) = 301.34 (60), 225.25 (33), 156.26 (59), 133.15 (100), 77.05 (58). Anal. calcd for C19H15N3O (301.34): C, 75.73; H, 5.02; N, 13.94; found: C, 75.61; H, 5.11; N, 14.02.
4-(4ʹ-Chlorophenyl)-3-phenyl-1,4-dihydrobenzo[e][1,2,4]triazin-6-ol (4b )
White solid (CH3CN), yield: 0.044 g (13%), m.p. 264–266 °C. IR (KBr): ν = 3411 (OH), 3270 (NH), 1629 (C=N), 1593 cm−1 (C=C). 1H NMR (400 MHz, DMSO-d6): δH = 10.05 (s, 1H, OH), 8.70 (s, 1H, NH), 7.55-7.40 (m, 12H, Ar–CH). 13C NMR (100 MHz, DMSO-d6): δC = 156.6 (Ar–C–OH), 153.5 (C=N), 134.2, 133.7, 130.2, 129.7, 128.5 (Ar–C), 128.3, 128.1, 127.8, 127.2, 127.0 (Ar–2CH), 126.9, 126.8 (Ar–CH). MS: m/z (%) = 337.1 (15), 335.1 (45), 261.7 (19), 259.1 (61), 225.3 (100), 149.2 (58), 133.2 (33), 114.6 (19), 112.6 (53), 77.1 (92). Anal. calcd for C19H14ClN3O (335.79): C, 67.96; H, 4.20; Cl, 10.56; N, 12.51; found: C, 68.05; H, 4.08; Cl, 10.70; N, 12.77.
4-(3ʹ-Chlorophenyl)-3-phenyl-1,4-dihydrobenzo[e][1,2,4]triazin-6-ol (4c )
White solid (CH3CN), yield: 0.037 g (11%), m.p. 262–264 °C. IR (KBr): ν = 3424 (OH), 3264 (NH), 1631 (C=N), 1584 cm−1 (C=C). 1H NMR (400 MHz, CDCl3): δH = 10.22 (s, 1H, OH), 7.94 (s, 1H, NH), 7.48-7.30 (m, 12H, Ar–CH). 13C NMR (100 MHz, DMSO-d6): δC = 158.2 (Ar–C–OH), 154.2 (C=N), 134.3, 133.9, 132.1, 130.3, 129.8 (Ar–C), 128.7, 128.2, 127.9, 127.3, 127.1 (Ar–2CH), 126.8, 126.5 (Ar–CH). MS: m/z (%) = 337.1 (15), 335.1 (45), 261.7 (28), 259.1 (60), 225.3 (100), 190.0 (8), 149.2 (59), 133.2 (22), 114.6 (15), 112.6 (44), 77.1 (80). Anal. calcd for C19H14ClN3O (335.79): C, 67.96; H, 4.20; Cl, 10.56; N, 12.51; found: C, 68.05; H, Cl, 10.65; 4.08; N, 12.72.
4-(4ʹ-Methoxyphenyl)-3-phenyl-1,4-dihydrobenzo[e][1,2,4]triazin-6-ol (4d )
White solid (CH3CN), yield: 0.060 g (18%), m.p. 210–212 °C. IR (KBr): ν = 3410 (OH), 3250 (NH), 1620 (C=N), 1590 cm−1 (C=C). 1H NMR (400 MHz, DMSO-d6): δH = 10.00 (s, 1H, OH), 8.80 (s, 1H, NH), 7.60 (dd, 2H, J = 1.2, 0.7 Hz), 7.30-7.10 (m, 8H, Ar–CH), 6.60 (dd, 2H, J = 1.2, 0.7 Hz), 3.90 (s, 3H, OCH3). 13C NMR (100 MHz, DMSO-d6): δC = 157.8 (Ar–C–OH), 152.0 (Ar–OCH3), 151.2 (C=N), 135.0, 130.2, 129.4, 128.6 (Ar–C), 127.6, 126.4, 125.8, 125.2, 122.0 (Ar–2CH), 121.0, 120.8 (Ar–CH), 52.0 (OCH3). MS: m/z (%) = 331.1 (30), 225.0 (100), 149.2 (40), 133.2 (30), 77.1 (90). Anal. calcd for C20H17N3O2 (331.37): C, 72.49; H, 5.17; N, 12.68; found: C, 73.00; H, 5.00; N, 12.82.
Reaction of piperidine with 5-hydroxy-1,4-naphthaquinone (5 )
An equimolar mixture of
8-Hydroxy-2-(piperidin-1-yl)naphthalene-1,4-dione (6 ) [42 and 43]
Green solid (EtOH), yield: 0.206–0.219 g (80%–85%), m.p. 159–160 °C (rep. 158 °C). IR (KBr): ν = 3440 (OH), 1700 (C=O), 1590 cm−1 (C=C). NMR (see Table 2). MS: m/z (%) = 257 (M+, 100). Anal. calcd for C15H15NO3 (257.28): C, 70.02; H, 5.88; N, 5.44; found: C, 70.20; H, 5.98; N, 5.60.
Crystal structure determinations of 6
The single-crystal X-ray diffraction study was carried out on a Bruker D8 Venture diffractometer with Photon100 detector at 123(2) K using Mo-Kα radiation (λ = 0.71073 Å). Dual space methods (SHELXT)
45
were used for structure solution, and refinement was carried out using SHELXL-2014 (full-matrix least squares on F2).
46
Hydrogen atoms were localized by difference electron density determination and refined using a riding model (H(O) free). A semi-empirical absorption correction and an extinction correction were applied.
CCDC 1968210 (
NCI screening assay
The methodology of the NCI procedure for primary anticancer assay is detailed on their site (http://www.dtp.nci.nih.gov), but briefly, the protocol was performed using the 60 human tumor cell lines panel derived from nine different neoplastic diseases. NCI-60 testing is performed in two parts: first, a single concentration is tested in all 60 cell lines at a single dose of 10−5 M or 15 μg/mL in accordance with the protocol of the Drug Evaluation Branch, National Cancer Institute, Bethesda, USA. If the results obtained meet selection criteria, then the compound is tested again in all 60 cell lines in 5 × 10 folds of dilution.
Supplemental Material
supporting_file – Supplemental material for Synthesis of quinone-based heterocycles of broad-spectrum anticancer activity
Supplemental material, supporting_file for Synthesis of quinone-based heterocycles of broad-spectrum anticancer activity by Ashraf A Aly, Alaa A Hassan, Nasr K Mohamed, Mohamed Ramadan, Amal S Abd El-Aal, Stefan Bräse and Martin Nieger in Journal of Chemical Research
Footnotes
Acknowledgements
The authors thank DFG Collaborative Center “3MET,” Karlsruhe Institute of Technology, Karlsruhe, Germany, for financial support to Professor Aly enabling him to carry out analyses in the aforesaid Institute. Purchase of the NMR spectrometer at the Florida Institute of Technology was assisted by the US National Science Foundation (CHE 03-42251).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
