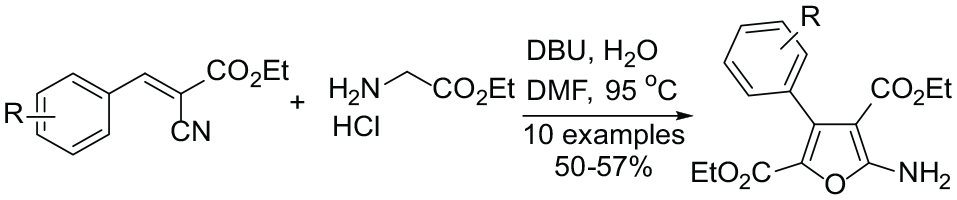
Abstract
An efficient synthetic approach for the preparation of densely substituted furans starting from (E)-ethyl 3-aryl-2-cyanoacrylates and ethyl glycinate hydrochloride mediated by 1,8-diazabicyclo[5.4.0]undec-7-ene in the presence of water is investigated. The reactions are carried out under mild conditions in N,N-dimethylformamide at 95 °C. The products are cleanly obtained in 50%–57% yields, resulting in an appealing alternative for accessing polysubstituted furan-2,4-dicarboxylates. The structure of a typical product, diethyl 5-amino-3-(p-tolyl)furan-2,4-dicarboxylate, is confirmed by X-ray crystallography.
Keywords
Introduction
Polysubstituted furan derivatives are essential oxygen-containing heterocycles that are embedded in a number of biologically active natural products (Figure 1).1–3 A large number of furan derivatives have shown interesting biological activities, such as anti-nociceptive, 4 antidiabetic, 5 anti-allergic and anti-asthamatic, 6 as well as cytotoxic and antitumor properties. 7

Examples of natural compounds containing a furan skeleton.
In addition, many substituted furans are present in dyes, essential oils, cosmetics, flavors, and fragrances.8–11 As valuable building blocks, furans are also employed in the synthesis of natural products, electrochemical polymers, and biomolecular macrocycles.12–14 Accordingly, the construction of substituted furans has attracted significant attention from chemists. Except for the classical methods, numerous efforts have been dedicated to the development of efficient synthetic methods for the preparation of diversely substituted furans.15–17 The development of general and efficient strategies for the synthesis of substituted furans from simple and readily available precursors under mild conditions is of great interest.18,19 In particular, as important furan derivatives, furancarboxylates occur widely as structural units in a variety of synthetic and natural compounds that can be applied as pharmaceuticals and organic materials.20–24 Recently, the utilization of simple ketones or 1,3-dicarbonyl compounds as starting materials to construct furancarboxylates through cyclization has attracted increasing interest. General methods for the preparation of the furancarboxylate framework include ring modification and cyclization of acyclic precursors, the majority of which focus on the cyclization of alkyne- and allene-containing substrates catalyzed by transition metals. 25 For example, Maulide and co-workers developed a method for the synthesis of polysubstituted furancarboxylates from sulfonium ylides and alkynes by gold-catalyzed cross-coupling cyclization. 26 One of the interesting strategies available for the synthesis of polysubstituted furancarboxylates involves the reaction of alkynes with 1,3-dicarbonyls using dl-[Ni(Phen)3]Cl2/Ag2CO3, 27 or SnCl2/CuI as the catalyst. 28 Beyond that, Deng and co-workers reported a strategy for the synthesis of polysubstituted furancarboxylates through potassium iodide (KI)/tert-butyl hydroperoxide (TBHP)-promoted tandem Michael addition/oxidative annulation from 1,3-dicarbonyl compounds and allenes under mild conditions. 29
Although these strategies have been well explored during the past few years, the reactions in many cases suffer from one or more drawbacks such as difficult to obtain substrates, harsh reaction conditions, and the use of noble metal catalysts. In the light of the significance of furancarboxylate systems and their diverse pharmacological properties, there has been a continuous effort to develop new, convenient, and versatile methods for synthesis of this class of compounds. Many studies have shown that glycinate derivatives are highly active and common intermediates in the synthesis of many important heterocycles.30–33 Recently, we reported a metal-free, simple, and practical synthetic approach for the synthesis of 5-amino-1H-pyrrole-2-carboxylates via the iodine-catalyzed [3 + 2] cycloaddition reaction of substituted 2-benzylidene-malononitrile and ethyl glycinate hydrochloride. 34 However, to the best of our knowledge, the cyclization reaction utilizing ethyl glycinate hydrochloride has not been reported to construct furancarboxylates. Herein, we have developed a new method for the synthesis of furancarboxylates starting from 3-aryl-2-cyanoacrylates and ethyl glycinate hydrochloride in the presence of water and 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU).
Results and discussion
To develop optimal reaction conditions, we began our investigation by studying the cyclization reaction of ethyl (E)-2-cyano-3-(p-tolyl)acrylate (
Optimization of the reaction conditions for the synthesis of
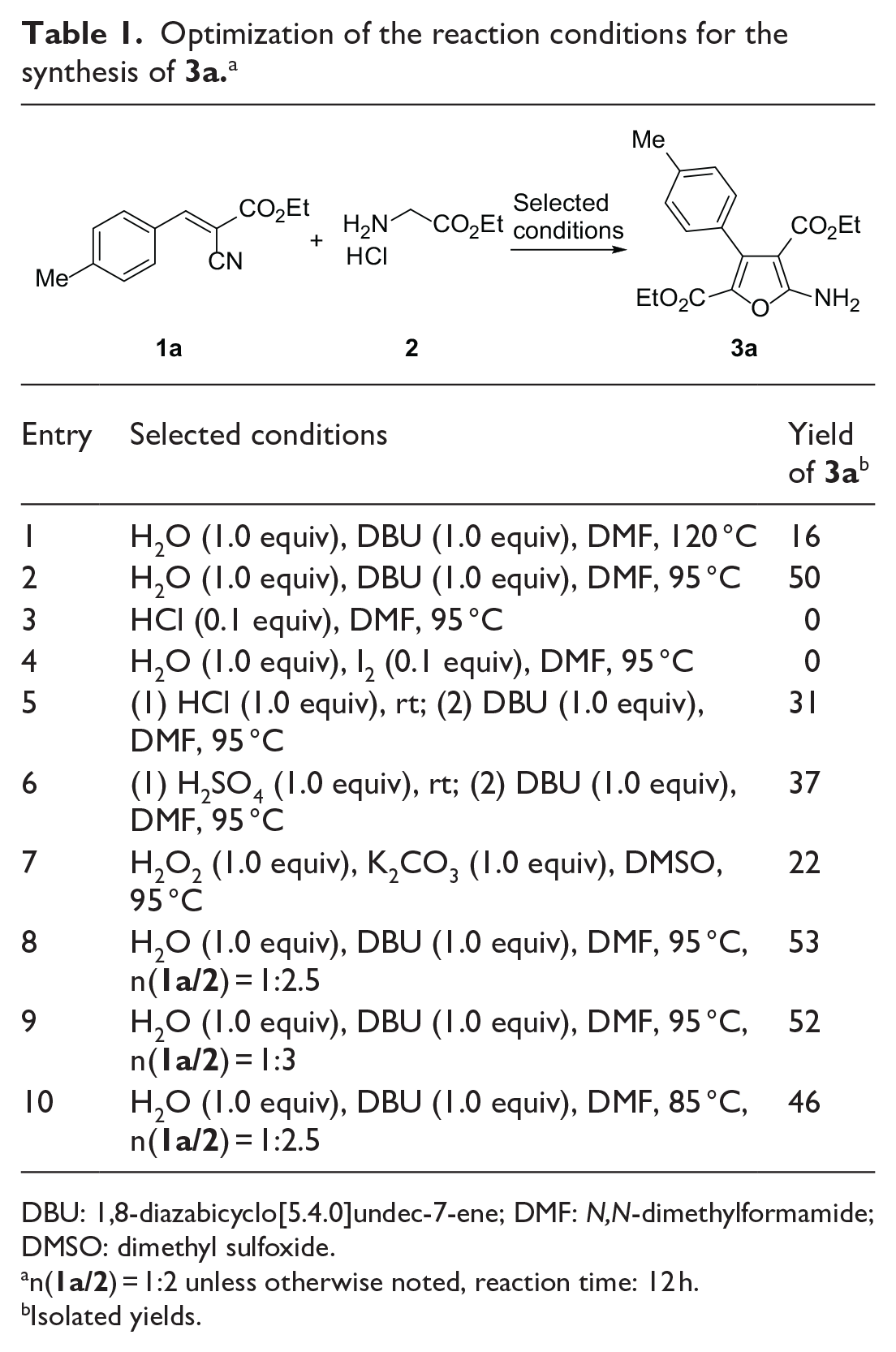
DBU: 1,8-diazabicyclo[5.4.0]undec-7-ene; DMF: N,N-dimethylformamide; DMSO: dimethyl sulfoxide.
n(
Isolated yields.
Further testing revealed that on using 3 mol L−1 HCl (0.1 equiv) instead of DBU at 95 °C, no conversion was observed (entry 3). In the absence of base, using the combination of iodine and water, no reaction took place (entry 4). Next, ethyl (E)-2-cyano-3-(p-tolyl)acrylate (
With optimized conditions in hand, the scope of the reaction was evaluated using different ethyl (E)-3-aryl-2-cyanoacrylates (Table 2). Thus a series of ethyl (E)-3-aryl-2-cyanoacrylates was synthesized from the corresponding aldehydes using ethyl cyanoacetate according to a reported procedure.
35
The (E)-3-aryl-2-cyanoacrylate was then reacted with ethyl glycinate hydrochloride (
Synthesis of diethyl 5-amino-3-arylfuran-2,4-dicarboxylates

Isolated yields.
The molecular structures of diethyl 5-amino-3-arylfuran-2,4-dicarboxylates
The crystal structure of
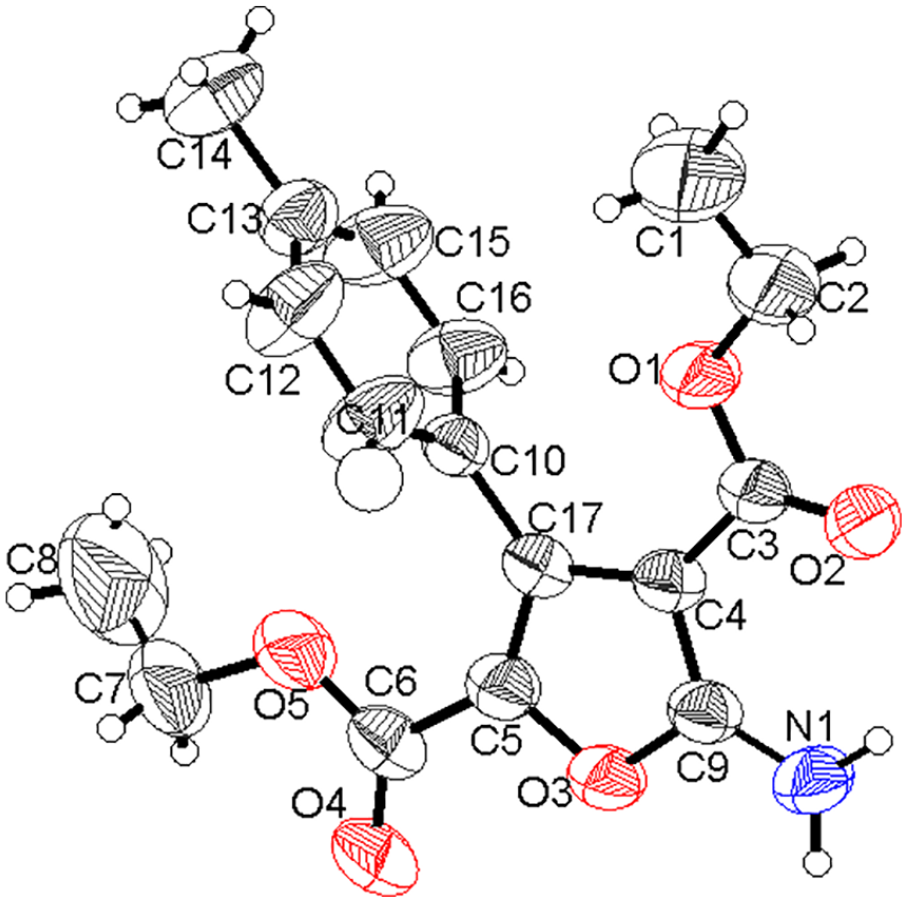
X-ray crystal structure of diethyl 5-amino-3-(p-tolyl)furan-2,4-dicarboxylate (
Crystal data and structure refinements for product

On the basis of these studies, a plausible mechanism is outlined in Scheme 1. The substrate ethyl glycinate hydrochloride undergoes deprotonation in the presence of the base to give ylide intermediate

A plausible mechanism for the reaction of (E)-3-aryl-2-cyanoacrylates with ethyl glycinate hydrochloride.
Conclusion
In summary, this work describes an efficient synthetic approach for the preparation of densely substituted furans starting from (E)-3-aryl-2-cyanoacrylates and ethyl glycinate hydrochloride in the presence of water. The reactions were carried out under mild reaction conditions in DMF medium at 95 °C, and the products were cleanly obtained in moderate yields. Hence, this method represents an appealing alternative for accessing polysubstituted furan-2,4-dicarboxylates.
Experimental
General
All melting points were determined using a Yanaco melting point apparatus and are uncorrected. IR spectra were recorded on a Nicolet FTIR 5DX spectrometer. 1H NMR (400 or 600 MHz) and 13C NMR (100 or 150 MHz) spectra were recorded on Bruker AV-400 and 600 spectrometers with tetramethylsilane (TMS) as the internal reference in CDCl3 solution. The J values are given in hertz. Only discrete or characteristic signals for the 1H NMR spectra are reported. High-resolution electrospray ionization (ESI) mass spectra were obtained on an ultra-high resolution time-of-flight (UHR-TOF) maXis (ESI) mass spectrometer. X-ray crystallographic analysis was performed with a SMART APEX-II diffractometer using monochromatic Mo KR radiation (λ) 0.71073 Å and integrated with the SAINT-Plus program. All calculations were performed with the SHELXTL crystallographic software package. Flash chromatography was performed on Qingdao Ocean silica gel (230–400 mesh) eluting with ethyl acetate hexanes. All reactions were monitored by thin-layer chromatography (TLC). Ethyl (E)-3-aryl-2-cyanoacrylates were synthesized according to a reported method, 35 and other reagents and solvents were purchased from commercial suppliers and purified by standard techniques.
General procedure for the synthesis of diethyl 5-amino-3-arylfuran-2,4-dicarboxylates 3
To a mixture of ethyl (E)-3-aryl-2-cyanoacrylate
Diethyl 5-amino-3-(p-tolyl)furan-2,4-dicarboxylate (3a )
Yellow solid, yield: 53%; m.p. 167.6–168.8 °C (PE/EtOAc); 1H NMR (600 MHz, CDCl3): δ 7.21 (d, J = 7.8 Hz, 2H), 7.15 (d, J = 7.8 Hz, 2H), 6.02 (s, 2H), 4.15 (q, J = 7.2 Hz, 2H), 4.06 (q, J = 7.2 Hz, 2H), 2.38 (s, 3H), 1.12 (t, J = 7.2 Hz, 3H), 1.02 (t, J = 7.2 Hz, 3H); 13C{1H} NMR (150 MHz, CDCl3): δ 163.7, 161.8, 157.9, 136.4, 135.3, 129.5, 128.5, 127.4, 126.6, 91.5, 59.3, 58.7, 20.3, 13.0, 12.8; IR (KBr, cm−1): 3484, 3400, 3302, 2981, 1694, 1668, 1635, 1574, 1547, 1279, 792, 575; HRMS (ESI): m/z [M + Na]+calcd for C17H19NNaO5: 340.1161; found: 340.1165.
Diethyl 5-amino-3-(4-methoxyphenyl)furan-2,4-dicarboxylate (3b )
Yellowish solid, yield: 57%; m.p. 171.5–172.7 °C (PE/EtOAc); 1H NMR (CDCl3, 600 MHz): δ 7.22 (d, J = 8.4 Hz, 2H), 6.88 (d, J = 8.4 Hz, 2H), 5.64 (s, 2H), 4.08 (q, J = 7.2 Hz, 2H), 4.04 (q, J = 7.2 Hz, 2H), 3.83 (s, 3H), 1.06 (t, J = 7.2 Hz, 3H), 1.01 (t, J = 7.2 Hz, 3H); 13C{1H} NMR (100 MHz, DMSO-d6): δ 163.7, 161.8, 158.3, 135.0, 130.0, 127.8, 122.6, 113.2, 111.4, 91.4, 59.3, 58.7, 54.2, 13.0, 12.9; IR (KBr, cm−1): 3479, 3406, 3301, 2965, 1682, 1647, 1556, 1291, 1262, 1098, 1021, 804; HRMS (ESI): m/z [M + Na]+calcd for C17H19NNaO6: 356.1110; found: 356.1119.
Diethyl 5-amino-3-(4-bromophenyl)furan-2,4-dicarboxylate (3c )
Yellowish solid, yield: 55%; m.p. 171.0–172.2 °C (PE/EtOAc); 1H-NMR (CDCl3, 400 MHz): δ 7.48 (d, J = 7.8 Hz, 2H), 7.19 (d, J = 7.8 Hz, 2H), 6.12 (s, 2H), 4.13 (q, J = 6.9 Hz, 2H), 4.06 (q, J = 6.9 Hz, 2H), 1.11 (t, J = 6.9 Hz, 3H), 1.02 (t, J = 6.9 Hz, 3H); 13C{1H} NMR (100 MHz, DMSO-d6): δ 163.4, 161.8, 157.7, 133.7, 130.9, 130.3, 129.1, 120.9, 116.9, 91.2, 59.5, 58.8, 13.0, 12.9; IR (KBr, cm−1): 3445, 3347, 2965, 1682, 1647, 1556, 1291, 1262, 1098, 1021, 804; HRMS (ESI): m/z [M + Na]+calcd for C16H16BrNNaO5: 404.0110; found: 404.0115, 406.0092.
Diethyl 5-amino-3-(m-tolyl)furan-2,4-dicarboxylate (3d )
Yellowish solid, yield: 57%; m.p. 165.0–167.0 °C (PE/EtOAc); 1H-NMR (CDCl3, 400 MHz): δ 7.28–7.23 (m, 1H), 7.19 (d, J = 7.8 Hz, 1H), 7.15 (s, 1H), 7.13 (d, J = 7.8 Hz, 1H), 6.12 (s, 2H), 4.13 (q, J = 6.9 Hz, 2H), 4.06 (q, J = 6.9 Hz, 2H), 2.38 (s, 3H), 1.11 (t, J = 6.9 Hz, 3H), 1.02 (t, J = 6.9 Hz, 3H); 13C{1H} NMR (100 MHz, DMSO-d6): δ 165.2, 162.1, 143.7, 136.7, 130.9, 130.3, 129.6, 129.1, 128.3, 120.9, 116.9, 91.2, 59.5, 58.8, 21.0, 13.0, 12.9; IR (KBr, cm−1): 3432, 3362, 2960, 1680, 1642, 1550, 1295, 1260, 1048, 1022, 796; HRMS (ESI): m/z [M + Na]+calcd for C17H19NNaO5: 340.1161; found: 340.1157.
Diethyl 5-amino-3-(o-tolyl)furan-2,4-dicarboxylate (3e )
Yellowish solid, yield: 53%; m.p. 163.0–164.6 °C (PE/EtOAc); 1H-NMR (CDCl3, 400 MHz): δ 7.29–7.21 (m, 2H), 7.17 (d, J = 7.8 Hz, 1H), 7.15 (d, J = 7.8 Hz, 1H), 6.15 (s, 2H), 4.22 (q, J = 6.9 Hz, 2H), 4.19 (q, J = 6.8 Hz, 2H), 2.30 (s, 3H), 1.16 (t, J = 6.9 Hz, 3H), 1.12 (t, J = 6.9 Hz, 3H); 13C{1H} NMR (100 MHz, DMSO-d6): δ 164.9, 161.2, 145.7, 138.7, 130.5, 130.0, 129.6, 129.0, 128.2, 128.0, 116.9, 91.0, 59.9, 58.6, 20.8, 13.2, 12.6; IR (KBr, cm−1): 3440, 3346, 2950, 1680, 1648, 1552, 1290, 1287, 1262, 1008, 998, 822; HRMS (ESI): m/z [M + Na]+calcd for C17H19NNaO5: 340.1161; found: 340.1158.
Diethyl 5-amino-3-(3-methoxyphenyl)furan-2,4-dicarboxylate (3f )
Yellowish solid, yield: 57%; m.p. 170.5–171.2 °C (PE/EtOAc); 1H-NMR (CDCl3, 600 MHz): δ 7.29–7.22 (m, 3H), 7.15 (s, 1H), 5.60 (s, 2H), 4.20 (q, J = 7.2 Hz, 2H), 4.18 (q, J = 7.2 Hz, 2H), 3.86 (s, 3H), 1.12 (t, J = 7.2 Hz, 3H), 1.08 (t, J = 7.2 Hz, 3H); 13C{1H} NMR (100 MHz, DMSO-d6): δ 165.7, 162.6, 158.2, 146.7, 135.2, 130.6, 129.2, 127.8, 120.6, 114.2, 112.4, 93.4, 59.6, 58.6, 54.3, 13.4, 12.6; IR (KBr, cm−1): 3482, 3402, 3302, 2970, 1680, 1640, 1562, 1290, 1258, 1088, 1001, 996, 836; HRMS (ESI): m/z [M + Na]+calcd for C17H19NNaO6: 356.1110; found: 356.1114.
Diethyl 5-amino-3-(2,4-dibromophenyl)furan-2,4-dicarboxylate (3g )
Yellowish solid, yield: 52%; m.p. 185.0–186.2 °C (PE/EtOAc); 1H NMR (CDCl3, 400 MHz): δ 7.68 (s, 1H), 7.52 (d, J = 7.8 Hz, 1H), 7.19 (d, J = 7.8 Hz, 1H), 6.10 (s, 2H), 4.25 (q, J = 6.9 Hz, 2H), 4.12 (q, J = 6.9 Hz, 2H), 1.12 (t, J = 6.9 Hz, 3H), 1.08 (t, J = 6.9 Hz, 3H); 13C{1H} NMR (100 MHz, DMSO-d6): δ 166.4, 161.8, 157.0, 155.2, 133.8, 130.8, 130.0, 129.1, 120.9, 118.8, 116.9, 91.8, 59.8, 58.0, 13.6, 12.2; IR (KBr, cm−1): 3440, 3331, 2952, 1680, 1642, 1550, 1321, 1230, 1098, 1004, 992, 804; HRMS (ESI): m/z [M + Na]+calcd for C16H15Br2NNaO5: 481.9215; found: 481.9212, 483.9192, 485.9170.
Diethyl 5-amino-3-(2-bromophenyl)furan-2,4-dicarboxylate (3h )
Yellowish solid, yield: 50%; m.p. 168.5–169.5 °C (PE/EtOAc); 1H NMR (CDCl3, 400 MHz): δ 7.56 (d, J = 7.8 Hz, 1H), 7.30–7.18 (m, 3H), 6.32 (s, 2H), 4.18 (q, J = 7.2 Hz, 2H), 4.12 (q, J = 6.9 Hz, 2H), 0.96 (t, J = 6.9 Hz, 3H), 0.88 (t, J = 6.9 Hz, 3H); 13C{1H} NMR (100 MHz, DMSO-d6): δ 163.5, 162.0, 157.7, 133.3, 133.1, 130.7, 129.6, 128.0, 125.3, 122.3, 116.2, 91.3, 59.4, 58.6, 12.8, 12.6; IR (KBr, cm−1): 3452, 3320, 2988, 1686, 1652, 1560, 1268, 1202, 1092, 992, 834; HRMS (ESI): m/z [M + Na]+calcd for C16H16BrNNaO5: 404.0110; found: 404.0112, 406.0094.
Diethyl 5-amino-3-(3-chlorophenyl)furan-2,4-dicarboxylate (3i )
Yellowish solid, yield: 55%; m.p. 165.5–166.4 °C (PE/EtOAc); 1H NMR (DMSO-d6, 600 MHz): δ 7.42 (s, 1H), 7.38 (d, J = 7.8 Hz, 1H), 7.30 (dd, J = 8.0, 7.8 Hz, 1H), 7.22 (d, J = 7.8 Hz, 1H), 6.05 (s, 2H), 4.19 (q, J = 7.2 Hz, 2H), 4.08 (q, J = 7.2 Hz, 2H), 1.04 (t, J = 7.2 Hz, 3H), 0.96 (t, J = 7.2 Hz, 3H); 13C{1H} NMR (100 MHz, DMSO-d6): δ 163.9, 161.6, 157.8, 145.2, 135.2, 130.2, 129.2, 127.0, 126.6, 126.2, 113.2, 91.3, 59.6, 58.9, 12.9, 12.7; IR (KBr, cm−1): 3466, 3392, 3292, 2930, 1685, 1648, 1562, 1278, 1260, 1082, 1041, 956, 796; HRMS (ESI): m/z [M + Na]+calcd for C16H16ClNNaO5: 360.0615; found: 360.0612.
Diethyl 5-amino-3-(thiophen-3-yl)furan-2,4-dicarboxylate (3j)
Yellowish solid, yield: 51%; m.p. 171.0–171.2 °C (PE/EtOAc); 1H NMR (CDCl3, 600 MHz): δ 7.37 (dd, J = 5.0, 1.0 Hz, 1H), 7.05 (dd, J = 3.6, 1.0 Hz, 1H), 7.02 (dd, J = 5.0, 3.6 Hz, 1H), 6.17 (s, 2H), 4.15 (q, J = 7.2 Hz, 2H), 4.08 (q, J = 7.2 Hz, 2H), 1.13 (t, J = 7.2 Hz, 3H), 1.06 (t, J = 7.2 Hz, 3H); 13C{1H} NMR (100 MHz, DMSO-d6): δ 163.5, 161.8, 157.6, 130.5, 129.9, 127.7, 127.4, 125.1, 124.9, 91.8, 59.5, 58.8, 12.9, 12.8; IR (KBr, cm−1): 3423, 3320, 2950, 1682, 1652, 1556, 1320, 1280, 1202, 1086, 1012, 995, 782; HRMS (ESI): m/z [M + Na]+calcd for C14H15NNaO5S: 332.0569; found: 332.0572.
Supplemental Material
checkcif_of_3a – Supplemental material for Novel synthesis of furancarboxylate derivatives via cyclization reactions of (E)-ethyl 3-aryl-2-cyanoacrylates and ethyl glycinate hydrochloride
Supplemental material, checkcif_of_3a for Novel synthesis of furancarboxylate derivatives via cyclization reactions of (E)-ethyl 3-aryl-2-cyanoacrylates and ethyl glycinate hydrochloride by Naili Luo, Shan Wang, Zhenjie Su and Cunde Wang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions.
Supplemental Material
Full details of the X-ray structure and crystal refinement of compound
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
