Abstract
A facile and efficient method was developed for the preparation of a variety of aryl, heteroaryl, and alkyl N-sulfinyl imines using 1,8-diazabicyclo[5.4.0]undec-7-ene. In addition to tert-butanesulfinamide, the condensation is also effective with p-toluenesulfinamide. The reaction was performed at room temperature and produces the corresponding N-sulfinyl imines in excellent yields in the absence of acids, metals, and additives. This methodology is also useful for the preparation of N-sulfinyl imines on gram scale. A one-pot synthesis was developed using aryl and heteroaryl alcohols with both tert-butanesulfinamide and p-toluenesulfinamide at room temperature, resulting in the corresponding N-sulfinyl imines with good yields.

Introduction
Imine bond formation by the condensation of carbonyl compounds and primary amine groups is important in organic synthesis due to the significance of imines in the fields of chemistry and biology. 1 Chiral sulfinamides function as amine counterparts and have been employed as useful chiral auxiliaries in asymmetric synthesis.2–4 They represent key building blocks for the synthesis of a number of biologically active molecules, chiral amines,4–8 and aziridines9–12 and also are constituents of pharmaceuticals, agro-chemicals, and compounds with industrial interest. 13 There are several reports for the synthesis of aldimines, and these are mainly based on the condensation of aldehydes with sulfinamides in the presence of reagents containing metals. Earlier reports were focused on the application of either activating or dehydrating agents such as copper(II) sulfate, 14 magnesium sulfate–pyridinium p-toluenesulfonate, 14 titanium(IV) alkoxides,14–16 cesium carbonate, 17 ytterbium(III) triflate, 18 potassium hydrogen sulfate, 19 and so on, to accomplish this conversion. These methods suffer from certain disadvantages like long reaction times, heating, low yields, formation of by-products, and usage of large excess reagents. Nevertheless, several reagents can achieve this transformation; indeed, there is a scope for the development of mild and efficient methods to address the drawbacks of the existing methodologies.
1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) is a sterically hindered amidine, which is used as a non-nucleophilic base in organic synthesis to achieve various transformations. 20 It is cheap, readily available, and easy to remove from reaction mixtures. To the best of our knowledge, methods for the synthesis of N-sulfinyl imines using DBU have not been reported yet. Herein, we aimed to develop a simple, mild, highly efficient, economic, and metal-free protocol for the formation of sulfinyl imines from sulfinamides and aldehydes in the presence of DBU. This method offers various advantages such as high yields and simple work-up, is scalable, and employs mild reaction condition.
Results and discussion
We initially optimized the method by using benzaldehyde (1; Table 1) and (S)-tert-butanesulfinamide (
Optimization of the reaction conditions.

NMM: N-methylmorpholine; THF: tetrahydrofuran; DABCO: 1,4-diazabicyclo[2.2.2]octane; DBU: 1,8-diazabicyclo[5.4.0]undec-7-ene; DME: dimethoxyethane; DMF: dimethyl formamide; RT: room temperature.
Isolated yield after chromatographic purification.
np: No product formation was observed.
The bold values in Table 1, indicate the best condition among screened conditions this is to attract readers attention to find the best among screened conditions.
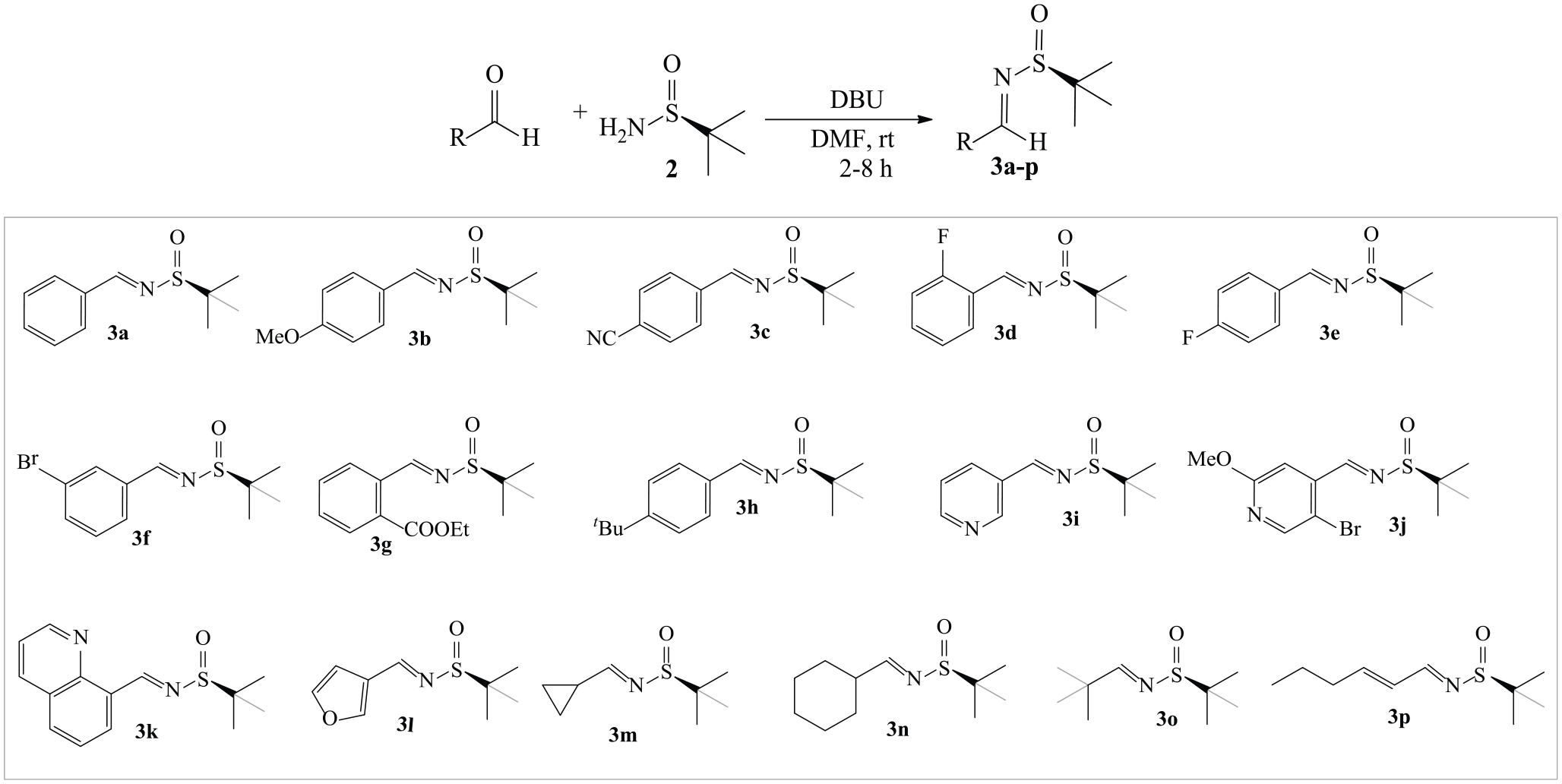
With optimized reaction conditions in hand, the scope of the reaction was subsequently examined with respect to various aldehydes (Table 2). The parent benzaldehyde (Table 2, entry 1) was smoothly reacted with
Synthesis of N-(tert-butylsulfinyl)imines

DBU: 1,8-diazabicyclo[5.4.0]undec-7-ene; DMF: dimethyl formamide; RT: room temperature.
Reaction conditions: aldehyde (1.0 mmol) and
Isolated yield after chromatographic purification.
Similarly, furan-3-carbaldehyde (Table 2, entry 12) gave the desired product
To illustrate the scalability of the standardized conditions, a scale-up batch was performed by using
In addition to N-(tert-butylsulfinyl)imine (
Synthesis of N-p-toluenesulfinyl imines
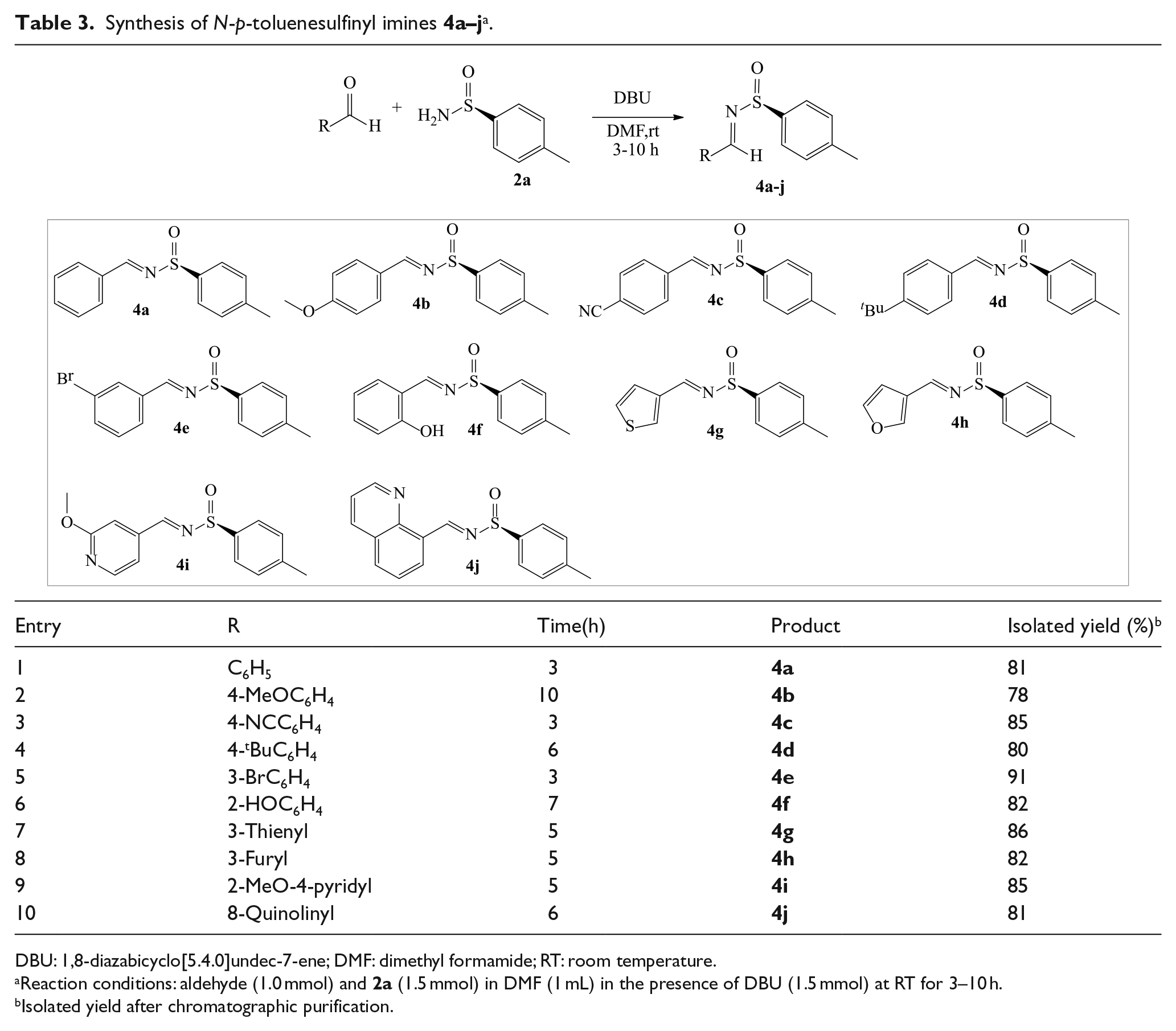
DBU: 1,8-diazabicyclo[5.4.0]undec-7-ene; DMF: dimethyl formamide; RT: room temperature.
Reaction conditions: aldehyde (1.0 mmol) and
Isolated yield after chromatographic purification.
Subsequently, we conducted the one-pot synthesis of N-sulfinyl imine
21
using benzyl alcohol (Table 4, entry 1), (S)-tert-butanesulfinamide
One-pot synthesis of N-sulfinyl imines a .

DBU: 1,8-diazabicyclo[5.4.0]undec-7-ene; DMF: dimethyl formamide; RT: room temperature.
Reaction conditions: alcohol (1.0 mmol);
Isolated yield after chromatographic purification.
np: No product formation was observed.
Our efforts toward the formation of (S)-(tert-butane-sulfinyl) imine under standardized conditions using acetophenone (

N-Sulfinyl ketimine formation.
Conclusion
In conclusion, we have established DBU as an efficient reagent for the condensation of sulfinamides with aldehydes to provide N-sulfinyl imines at room temperature. The reactions proceed under mild and homogeneous conditions with clean reaction profiles and simple work-up. The method was shown to be appropriate for a broad range of aldehydes, including sterically hindered, electron-rich, and electron-deficient substrates. This methodology is also useful for the preparation of N-sulfinyl imines on gram scale. Remarkably, the one-pot reaction of aromatic and heteroaromatic alcohols with sulfinamides for the preparation of the corresponding N-sulfinyl imines resulted in good yields. Unfortunately, this one-pot protocol for the formation of N-sulfinyl imines was unsuccessful with aliphatic alcohols.
Experimental
All reagents and solvents used in this work were obtained from commercial sources and were used without further purification. TLC was performed on Merck silica gel 60F254 (0.25-mm thickness) plates and visualized under UV light. Column chromatography was performed using brand silica gel of 100–200 mesh. Melting points were determined in open capillary tubes using paraffin oil bath and are uncorrected. NMR data were collected for 1H at 400 MHz and for 13C at 100 MHz on an Agilent NMR spectrometer. Chemical shifts are reported in parts per million relative to tetramethylsilane (TMS) as an internal standard (δ 0), and the values are reported as follows: chemical shift, integration, multiplicity (s = singlet, d = doublet, t = triplet, q = quartet, m = multiplet, br s = broad singlet), and the coupling constants are in hertz. For 13C NMR, CDCl3 (δ 77.27) was used as the internal standard, and spectra were obtained with complete proton decoupling. Mass spectra were recorded on an Agilent mass spectrometer in electrospray ionization (ESI) mode. High-resolution mass spectra were obtained on a WATERS Q-TOF Premier-HAB213 spectrometer in ESI mode.
General experimental procedures for the synthesis of N-sulfinyl imines 3 and 4
In a round-bottom flask, to aldehyde (1.0 mmol) in DMF (1 mL) was added sulfinamide (
General experimental procedures for the one-pot synthesis of N-sulfinyl imines 3 and 4
In a round-bottom flask, to alcohol (1.0 mmol) and manganese dioxide (10 mmol) in DMF (1.5 mL), was added sulfinamide (
(S)-N-Benzylidene-2-methylpropane-2-sulfinamide (
(S)-N-(4-Methoxybenzylidene)-2-methylpropane-2-sulfinamide (
(S)-N-(4-Cyanobenzylidene)-2-methylpropane-2-sulfinamide (
(S)-N-(2-Fluorobenzylidene)-2-methylpropane-2-sulfinamide (
(S)-N-(4-Fluorobenzylidene)-2-methylpropane-2-sulfinamide (
(S)-N-(3-Bromobenzylidene)-2-methylpropane-2-sulfinamide (
(S)-Ethyl 2-(((tert-butylsulfinyl)imino)methyl) benzoate (
(S)-N-(4-(tert-Butyl)benzylidene)-2-methylpropane-2-sulfinamide (
(S)-2-Methyl-N-(pyridin-3-ylmethylene)propane-2-sulfinamide (
(S)-N-((5-Bromo-2-methoxypyridin-4-yl)methylene)-2-methylpropane-2-sulfinamide (
(S)-2-Methyl-N-(quinolin-8-ylmethylene)propane-2-sulfinamide (
(S)-N-(Furan-3-ylmethylene)-2-methylpropane-2-sulfinamide (
(S)-N-(Cyclopropylmethylene)-2-methylpropane-2-sulfinamide (
(S)-N-(Cyclohexylmethylene)-2-methylpropane-2-sulfinamide (
(S)-N-(2,2-Dimethylpropylidene)-2-methylpropane-2-sulfinamide (
(S)-N-((E)-Hex-2-en-1-ylidene)-2-methylpropane-2-sulfinamide (
(S)-N-Benzylidene-4-methylbenzenesulfinamide (
(S)-N-(4-Methoxybenzylidene)-4-methylbenzene-sulfinamide (
(S)-N-(4-Cyanobenzylidene)-4-methylbenzene-sulfinamide (
(S)-N-(4-(tert-Butyl)benzylidene)-4-methylbenzene sulfinamide (
(S)-N-(3-Bromobenzylidene)-4-methylbenzene-sulfinamide (
(S)-N-(2-Hydroxybenzylidene)-4-methylbenzene-sulfinamide (
(S)-4-Methyl-N-(thiophen-3-ylmethylene)benzene sulfinamide (
(S)-N-(Furan-3-ylmethylene)-4-methylbenzene-sulfinamide (
(S)-N-((2-Methoxypyridin-4-yl)methylene)-4-methyl benzenesulfinamide (
(S)-4-Methyl-N-(quinolin-8-ylmethylene)benzenesulfinamide (
Supplemental Material
Supplemental_file.docx_R1 – Supplemental material for 1,8-Diazabicyclo[5.4.0]undec-7-ene-mediated formation of N-sulfinyl imines
Supplemental material, Supplemental_file.docx_R1 for 1,8-Diazabicyclo[5.4.0]undec-7-ene-mediated formation of N-sulfinyl imines by Manjunatha M Ramaiah, Priya Babu Shubha, Pavan Kumar Prabhala and Nanjunda Swamy Shivananju in Journal of Chemical Research
Footnotes
Acknowledgements
The authors wish to thank Srinivasaiah and Krishna Koushik for their support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.