Abstract
Blue fluorescent carbon dots are synthesized via a one-step hydrothermal method using rosemary leaves as a carbon source. The obtained carbon dots are characterized by X-ray diffraction, transmission electron microscopy, X-ray photoelectron spectroscopy, UV-Vis, FTIR, and fluorescence spectrometry. The results show that carbon dots gave a maximum emission peak at 422 nm when excited at 332 nm. The average particle diameter of the synthesized spherical carbon dots is found to be 16.13 nm with a 4.6-nm standard deviation. The diameters of the produced carbon dots vary, as is evident from standard deviation heights, resulting in multicolor emission spectra. The fruit storage experiment is carried out by investigating the qualitative antioxidant ability of the produced carbon dots. The results obtained for polyvinyl alcohol–carbon dot–coated samples differ remarkably compared to polyvinyl alcohol–coated samples only. The polyvinyl alcohol–carbon dot composition is used in latent fingerprint detection applications. In analyses made on a glass surface, fingerprints cannot be monitored on the glass surface with the polyvinyl alcohol layer under ultraviolet light, while they are observed on the surface covered with the polyvinyl alcohol–carbon dot composition. Moreover, biological activity against Gram-positive, Gram-negative, and yeast strains are investigated.
Introduction
Nanomaterials have found wide applications in many fields leading to significant interest in the scientific world; in particular, carbon nanomaterials have been at the forefront of this growing interest. Among carbon nanomaterials, carbon dots (CDs) have attracted considerable interest since they were described in the early 2000s because of their unique properties. 1 CDs are very interesting because of their low toxicity, water solubility, photostability, high electrical conductivity, and biocompatibility properties, as well as their low-cost production methods, chemical inertness, and photoluminescence (PL) properties.2–7
After their accidental discovery in 2004, numerous synthesis techniques are used to produce CDs: laser ablation, arc discharge, hydrothermal/solvothermal combustion/heating, microwave irradiation are some of the methods used to synthesize water-soluble CDs.8–10 Energy consumption, the complexity of CD production, and synthetic conditions are the basic limiting factors preventing enlarging the production and applications of CDs. 11 However, hydrothermal methods with biomass carbon sources to synthesize CDs have generated great interest because they are compatible with green chemistry processes. 12 There is a growing interest in the hydrothermal synthesis of CDs from different biomass as a carbon source. Ginkgo leaves, 11 Ocimum sanctum, 13 Prunus mume, 14 sweet potato,15,16 sugarcane bagasse, 17 durian shell waste, 18 radish,19,20 date kernel, 21 mustard seeds, 22 aloe vera, 23 onion waste, 24 starch, 25 milk,26,27 coffee bean shell, 28 black tea, 29 yogurt, 30 finger millet ragi, 31 Plectranthus amboinicus, 32 Borassus flabellifer, 33 and carob 34 are some used recent examples of carbon sources to synthesis CDs.
Rosemary (Rosmarinus officinalis L.), which we use as a carbon source in this study, is an aromatic herb that has been used for medicinal, culinary, fragrance, and ornamental purposes since ancient times, and it is generally grown in the Mediterranean region. 35 Numerous studies on the antimicrobial, antioxidant, and anticancer effects of this herb have been reported in the literature.36,37
Polyvinyl alcohol (PVA) is a synthetic polymer that is biodegradable, completely water-soluble, harmless, non-carcinogenic, and has high biocompatible properties. PVA can be easily combined with different synthetic and natural materials due to its good chemical-forming, high chemical stability, and highly hydrophilic properties. PVA has many uses in applications such as polymer recycling, controlled drug delivery systems, film formation, and packaging. 38 Due to the good biological activities of materials such as CDs and PVA, a chemically blended combination of CDs and PVA may have positive effects, especially on the biological properties of food packaging-storing and protection films. 39 According to recent reports, results have been published showing that CDs can be immobilized as matrix films by dissolving them in polymeric solutions such as poly(ethylene glycol) and PVA.40–42 In this study, we have examined such features especially on the food-coating qualities of CDs and PVA. The acceleration of biological events that cause deterioration in post-harvest vegetables and fruits causes changes in the nutritional value, quality, aroma, texture, and appearance of foods. Edible films and coatings supply additional protection in storing fresh food and extending the shelf life. These coating materials, by providing a semi-permeable structure between the food product and the environment, control the respiratory rate, dehydration, oxidative reactions, and transfer of active compounds. 43 Hence, many materials such as polysaccharides, proteins, essential oils, or combinations thereof can serve as edible coatings. Edible polymeric materials, essential oils, or blends of these can be used as edible coatings to improve the stability of fresh food products.39,44
Bananas are the second most abundant fruit produced after citrus and are grown in more than 130 countries worldwide representing about 16% of the world’s total fruit production. Bananas, which are extremely rich and nutritious in terms of potassium and calcium, and more easily digestible than many other fruits, are one of the essential fruits consumed in daily life, both in terms of aroma-flavor and easy peeling and eating. 45
CDs have also been utilized for fingerprint visualization and detection. Latent fingerprint detection research is an important topic in forensic science because it is an essential tool used in identifying offenders. Therefore, the identification of clear and invisible hidden fingerprint images is crucial in the resolution of forensic events. Today, latent fingerprint detection using nanomaterials is a very important application. CDs exhibit fluorescent properties and have attracted the attention of researchers. 46 Moreover, since CDs are non-toxic and can be synthesized easily and cheaply, their potential for use in many different biological applications such as bio-imaging, bio-sensing, and drug delivery is being explored. 47 Widely used antibacterial chemicals create potential toxicity for environmental and ecological systems, especially when a high dosage is required, while CDs are non-toxic and pollution-free. 48
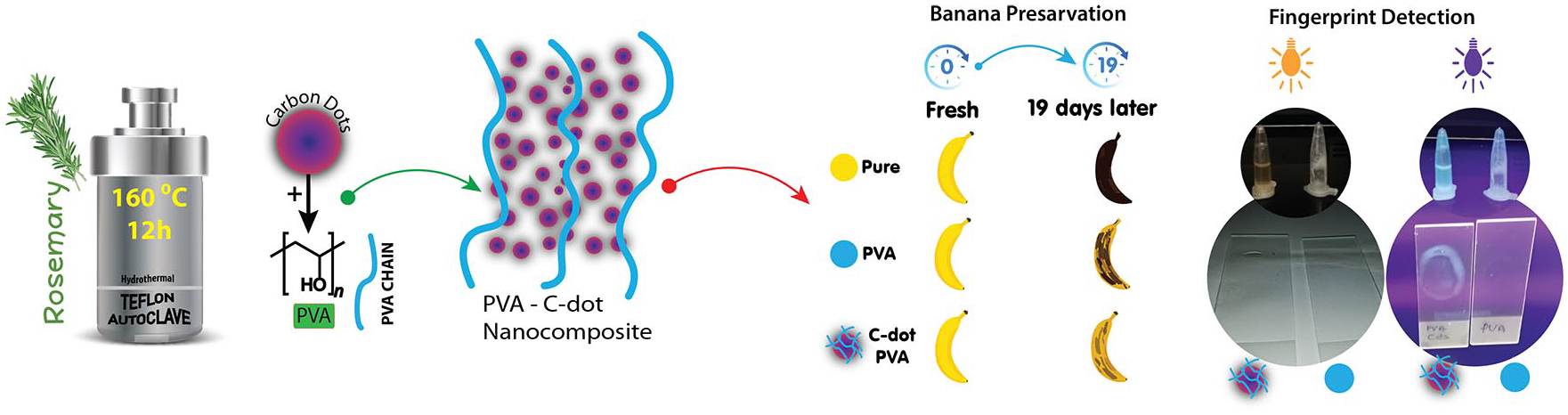
Herein, considering these points, we wanted to investigate the food storage, fingerprint examination, and antimicrobial properties of the new material obtained by the combination of CDs and PVA. As a result, this study presents an original method that may extend the shelf life of bananas and to develop applications in the food industry and fingerprint identification using a combination of nanomaterials containing CDs and polymeric materials.
Results and discussion
In this study, the CDs were synthesized from rosemary leaves using hydrothermal methods. During the synthesis of the water-soluble CDs, eight experiments were carried out to investigate the effect of the hydrothermal reaction time and temperature. The fluorescence properties of stable colloidal suspensions obtained after removing large particles from the produced sample were investigated. The maximum emission intensities of the synthesized dispersions under the appropriate excitations were measured. It is known that emission intensity is usually considered as a semi-quantitative index for the number of CDs suspension in the produced sample. A high fluorescence intensity represents a high CD amount and vice versa. 49
For the hydrothermal synthesis of CDs, the reaction temperature and reaction time are the two essential parameters related to CD formation levels, carbonization degrees, and PL performance. To investigate the reaction temperature–dependent PL behavior of the CDs, the four commonly used temperatures for CD synthesis via hydrothermal methods were selected. These temperatures are 140, 160, 180, and 200 °C and the reaction time was chosen as 6 h. The obtained results are given in Figure S2 (see the Supplementary Information). The PL peak of the CDs is gradually increased as the reaction temperature moves from 160 to 200 °C. To investigate the influence of the reaction time, 6, 8, 10, and 12 h were selected according to the literature3,50,51 and are demonstrated in Figure S3. The results indicated that the PL intensity decreased on increasing the reaction time from 6 to 8 h and continued to increase on increasing the reaction time. In hydrothermal synthesis, a longer reaction time can lead to further conversion of the components into carbonization products. 52 The CDs produced with the longest reaction time (12 h) were used for other analyses. The phase structure of the synthesized CDs (12 h at 160 °C) was characterized by the help of X-ray diffraction (XRD). The produced CDs gave a low-intensity broad diffraction peak centered at θ = 21.6° indicating an amorphous carbon phase (see Figure S4 in the Supplementary Information).
The size and morphological properties of the produced carbon dots were examined by high-resolution transmission electron microscopy (HR-TEM) (Figure 1(a)). It is clearly shown that the produced CDs have non-uniform size distribution. The average diameter of the CDs is 16.13 nm with a 4.6-nm standard deviation by counting 68 nanoparticles (Figure 1(c)). Figure 1(b) shows the highly resolved lattice fringe with an average interplanar spacing (d value) of 0.39 nm, and this lattice fringe has a higher than graphite (0.33 nm) interlayer spacing. The magnitude of the d value related to the amorphous nature of the material with the decrease in d value results in a reduction of the amorphous nature and an increase in the d value of the synthesized CDs stream from oxygen-containing groups.53,54 The selected area electron diffraction (SAED) pattern of the CDs also supports the imperfect crystalline nature of the CDs (Figure 1(d)). The interlayer distance found from XRD at 21.6° is 0.41 nm and is remarkably close to the measured value from the HR-TEM results.

(a) High-resolution TEM image of the CDs derived from rosemary leaves, (b) lattice plane distances of the CDs, (c) particle size distribution histogram, and (d) SAED pattern of the CDs.
The UV-Vis absorption spectrum and fluorescence spectrum were used to investigate the optical properties of the CDs. Figure 2 shows that the UV-absorption spectrum of the synthesized CDs gives a peak at 280 nm which is attributed to the π–π* transition of the C=C bonds, which is consistent with the literature.54,55 The excitation-dependent PL emission properties of the produced CDs are given in Figure 3. The PL emission peak shifted to a longer wavelength (red shift) when the excitation wavelength changed from 332 to 422 nm. The fluorescence intensity, however, decreases and becomes weaker as the excitation wavelength increases. 55 The maximum emission intensity was observed at 332 nm excitation. The multicolor fluorescence is related to the wavelength of excitation, and this excitation-dependent fluorescence is due to the non-uniform size of the produced CDs. 56 The inequality in the size of the CDs might be the reason for obtaining tunable fluorescence.57,58 The fluorescence emission–excitation three-dimensional (3D) map of the CDs and the corresponding contour plot of the CDs are demonstrated in Figure 4. In this graph, the maximum emission intensity is shown at 424 nm when the CDs are excited at 332 nm. Thereafter, the maximum emission value decreased with increasing or decreasing excitation wavelength.

UV-Vis absorption spectra of the CDs extracted from rosemary leaves.
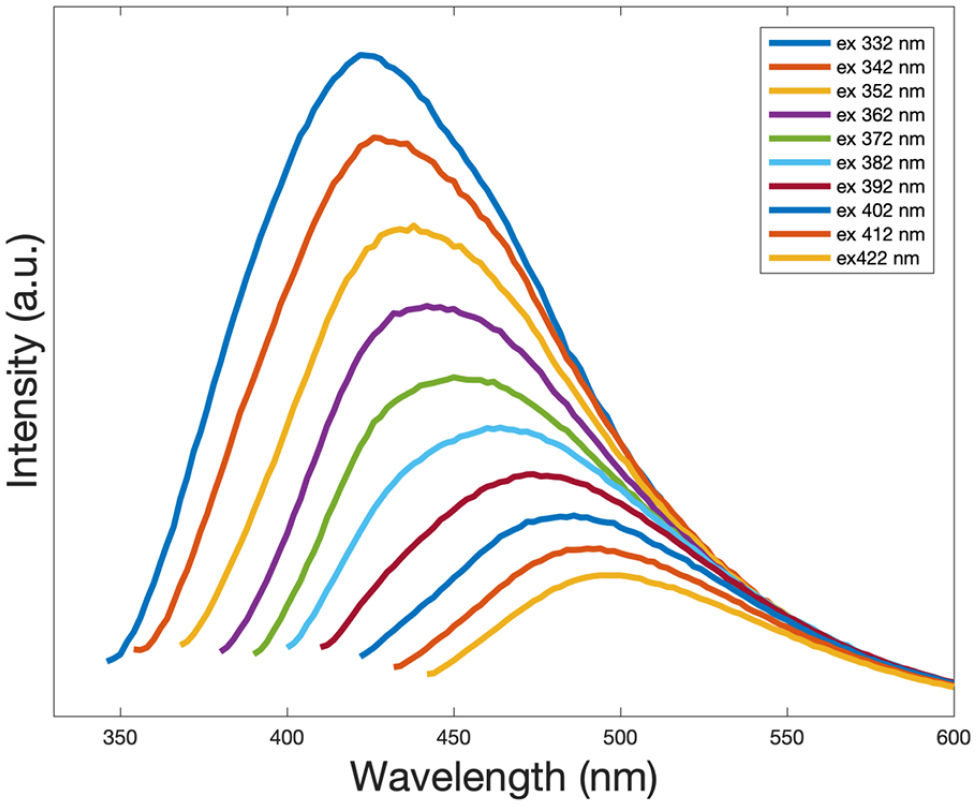
Excitation-dependent PL emission spectra of the CDs extracted from rosemary leaves.

The fluorescence emission–excitation 3D map of the CDs and the corresponding contour plot of the CDs.
The FTIR spectrum gives information about the chemical and structural nature of the synthesized CDs. The FTIR spectrum of the CDs obtained from rosemary leaves is shown in Figure 5. The peak at 3271 cm−1 corresponds to the stretching vibration of O–H/N–H, which indicate hydrophilic functional groups, and the peak at 1636 cm−1 corresponds to the vibrational absorption of the C=O group.59,60 The surface composition and elemental analysis of the produced CDs were further characterized by the X-ray photoelectron spectroscopy (XPS). The XPS of the CDs presents four peaks at 530, 398, 345, and 283 eV corresponding to O1s, N1s, Ca2p, and C1s, respectively, (Figure 6) with the corresponding content of each element being consistent with the literature.61,62 The International Commission on Illumination generated the CIE diagram that helps to plot PL data results seen by the human eye with the established standard. 63 The CIE diagram is plotted for different excitation wavelengths and is given in Figure S5 (see the Supplementary Information). As the excitation wavelength increased, the CIE color changes from violet to green. The banana storage experiment was conducted for the qualitative evaluation of the antioxidant ability. Three bananas were used as pure (no additive), PVA coated, and CDs + PVA coated. All the bananas were unripe, and the greenish-yellow color can be seen in Figure 7. The difference between the CDs + PVA and PVA additive became apparent day after day. Although the PVA-coated banana surface became rough, the CDs + PVA-coated banana surface became negatively affected on the ninth day of the experiment. On the 19th day of the experiment, the untreated banana had turned black in color, the PVA-coated banana was also mostly black in color, while the CDs + PVA-covered banana was in the best condition compared to the others. Similar results were also found in Zhang et al. 28 supporting that CDs have functional antioxidant capacity for use in fruit storage. The photographs of PVA and CDs + PVA used in the latent fingerprint experiment under room light and under 365 nm UV light irradiation are shown in Figure 8(a) and (b), respectively. The PVA + CDs fingerprint under 365 nm irradiation is given in Figure 8(c).

FTIR spectrum of the as-prepared CDs.
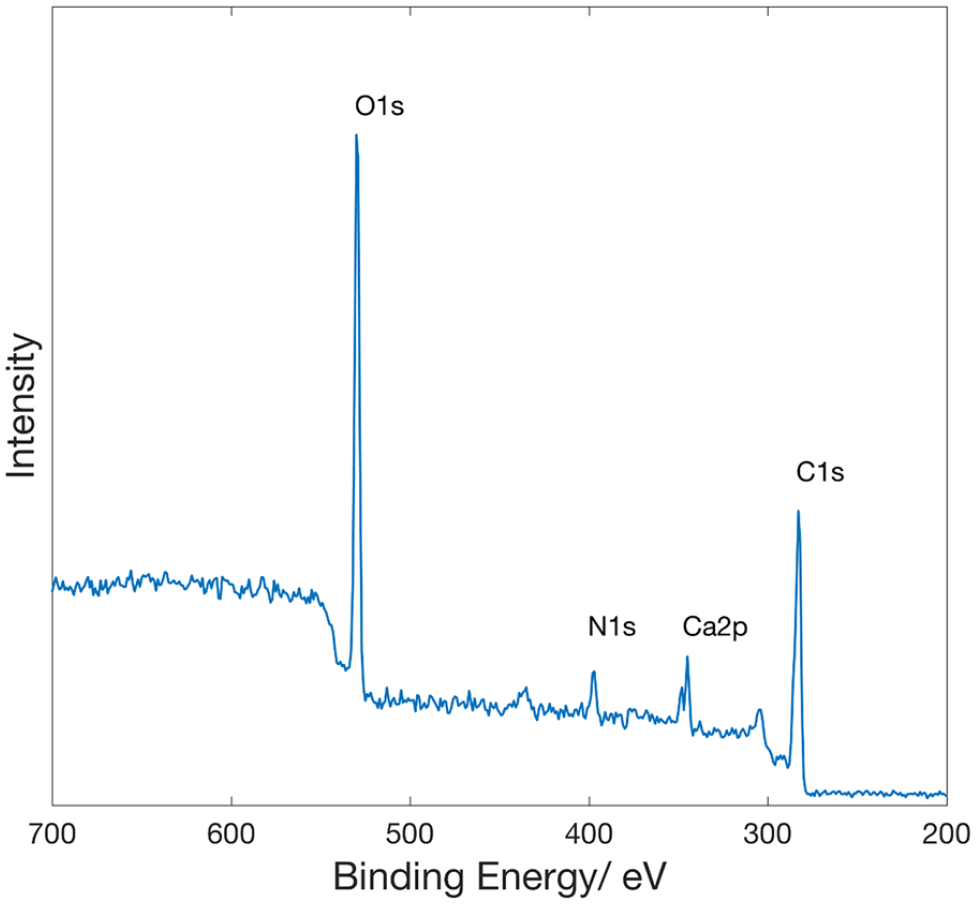
X-ray photoelectron spectrum (XPS) of the as-prepared CDs.

Digital images of untreated (bottom), PVA (middle), and PVA + CDs (top) coated bananas as a function of days.

(a) Daylight image of PVA and PVA + CDs, (b) image of PVA and PVA + CDs under UV light, and (c) fingerprint image of PVA and PVA + CDs under UV light.
Biological activity findings
The biological activities of the CDs were studied with reference to the active ingredients Amikacin and Gentamicin and are shown in Table S1. In this study, it was determined that samples (1 mL) showed 8–10 mm diameter inhibition zones against Gram-positive, Gram-negative, and yeast strains. As can be seen from Table S1, there is no biological activity observed on the strains of Escherichia coli, Salmonella typhimurium, Staphylococcus aureus, Bacillus subtilis, and Candida albicans at other concentrations.
Minimum inhibition concentration findings
The minimum inhibition concentrations (MICs) of the CDs on microorganism strains are given in Table S2. It has been determined that the CDs have different antimicrobial activities on microorganism strains. Gram-positive, Gram-negative, and yeast strains were determined to have an MIC. MICs of CDs were 12 µg/mL on yeast strains. It is thought that these different findings may be caused by the presence of specific cell wall structure in each strain.
Conclusion
In summary, water-soluble CDs have been successfully synthesized from rosemary leaves for the first time by a simple hydrothermal synthesis method. The morphological, structural, and optical properties are characterized by TEM, XRD, XPS, UV-Vis, FTIR, and fluorescence spectrometry. The average diameter of the produced CDs is found to be 16.13 nm. Biological activities against Gram-positive, Gram-negative, and yeast strains have been explored. A banana storage experiment was conducted for a qualitative investigation of the antioxidant ability and the usability of CDs for fruit storage. The obtained results are promising for the use of CDs in this field. Because of the suitable biological activities of CDs and PVA, a combination of CDs and PVA may have beneficial effects on the biological characteristics of food storage. Also, a PVA–CDs composite has been used for latent fingerprint detection under UV light. However, further improvements and modifications are needed to detect fingerprints under visible light. These findings may extend the field of CD applications by contributing to the development of new perspectives on composite materials with CDs.
Experimental
Materials
The rosemary leaves (R. officinalis L.) were collected from Kahramanmaraş Sütçü İmam University (KSÜ), Avşar Campus (Kahramanmaraş, Turkey). PVA was obtained from Alfa Aesar. All reagents were analytic grade and used without further purification. Ultrapure water was used throughout the experimental process.
Synthesis of CDs
Rosemary leaves were first washed under tap water and then distilled water. They were then dried in an oven for 24 h at 50 °C. The dry rosemary leaves were ground with a pestle in a mortar, and 2 g of ground rosemary leaves were dispersed into 35 mL of pure water. After being sonicated for 20 min, suspension was transferred to a 50-mL Teflon-lined stainless steel autoclave. The autoclave was sealed and heated at different reaction temperatures (140, 160, 180, and 200 °C) for different reaction times (6, 8, 10, and 12 h). The obtained dark brown solution of the CDs was centrifuged and filtered to remove insoluble and large black particles. The obtained CD suspension was stored at 4 °C for further characterization and application. The fluorescent stability/photostability of the synthesized CDs was investigated under UV light (365 nm) irradiation for 40 min and is shown in Figure S1 (see the Supplementary Information). The fluorescent intensity of the CDs changed slightly after 40 min of UV light irradiation. Very small photobleaching was observed after 40 min of UV light irradiation, demonstrating that the synthesized CDs have excellent stability.
Characterization
The morphology and microstructure of the CDs were analyzed using HR-TEM (JEOL JEM-2100 (UHR)). Fluorescence spectra were measured with a Varian Cary Eclipse fluorescence spectrometer. UV-Vis absorption spectra were obtained using a Shimadzu 1800 UV-Vis spectrometer. FTIR spectra were obtained with a PerkinElmer Spectrum 400 system in the range 4000–400 cm−1. XRD of samples was recorded using a Philips X’Pert PRO XRD with Cu Kα radiation (λ = 0.154056 nm, set at 40 kV and 30 mA). XPS measurements were performed on a Specs-Flex Mod electron spectrometer using a monochromatized Al Kα excitation source.
Plant material preparation
The bananas used in the shelf life extension experiment were sourced in the freshest possible form from a local convenience store around campus. We made sure that the bananas were physiologically mature. The bananas, which had a uniform shape, size, and color, and were not subject to any crushing or spoilage during selection, were selected and quickly moved to the laboratory for experimentation. Prior to the experiment, the weight and volume of the bananas were chosen to be the most homogeneous and closest to each other.
Fabrication of CDs–PVA composites
The banana experiment was conducted using process similar to that in the literature. 64 PVA (25 g) and pure water (130 mL) were mixed and heated to 98 °C and magnetically stirred. Next, about 10 g of a CD aqueous solution was added. The surface of the banana was coated with the obtained PVA–CDs solution with the help of a brush. Furthermore, latent fingerprint detection was carried out with PVA and the obtained PVA–CDs solution using a thumb trace on a glass substrate. The latent fingerprints were photographed under UV light.
Antimicrobial activity
The test microorganisms used in the study included three Gram-positive bacteria (S. aureus, Rosenbach ATCC-6538; B. subtilis, Ehrenberg ATCC-14028; and Bacillus cereus ATCC-7064), two Gram-negative bacteria (E. coli ATCC-8739, S. typhimurium), and one yeast (C. albicans ATCC-90028). KSÜ, Kahramanmaraş, Turkey, provided the cultures. The biological materials used in the study were activated by incubation at 37 ± 1 °C for 18 h. In the antimicrobial activity studies, Müller-Hilton Agar (MHA) medium was used for bacteria, and Malt Extract Agar (MEA) was used for the yeast strain. 65 The strains in sterile Petri dishes were inoculated with a 0.5 McFarland standard, and the strains were incubated at 37 ± 1 °C for 1 h. For control groups, a DMSO-containing disk and Amikacin (AK: 30 μg) and Gentamicin (CN: 10 μg) were used.
Disk diffusion assay
The antimicrobial activity of the synthesized complexes was determined using the Kirby–Bauer disk diffusion method. 65 The synthesized compounds were dissolved in 10% DMSO and impregnated in disks at a concentration of 25 μL to disks made of blank sterile Whatman papers (Schleicher & Schül no. 2668, Germany) with a diameter of 6 mm. The prepared disks were placed on the cultivated samples of bacteria and yeast strains and incubated at 37 ± 1 °C for 18–24 ± 2 h.65,66 The study was performed in plicate, and the mean values are given. To determine the minimum inhibition values of the samples, the minimum effect values against Gram-positive and Gram-negative bacteria and the yeast strain were determined by dissolving in 10% DMSO at the following three different concentrations: 1, 0.1, and 0.01 mL.
Supplemental Material
Supp – Supplemental material for Green synthesis of water-soluble fluorescent carbon dots from rosemary leaves: Applications in food storage capacity, fingerprint detection, and antibacterial activity
Supplemental material, Supp for Green synthesis of water-soluble fluorescent carbon dots from rosemary leaves: Applications in food storage capacity, fingerprint detection, and antibacterial activity by Hasan Eskalen, Mustafa Çeşme, Süleyman Kerli and Şükrü Özğan in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially sponsored by the Kahramanmaraş Sütçü İmam University, Scientific Research Projects Coordination Unit (project no. 2019/1-26 M).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
