Abstract
A novel chloro- and BF2bdk-substituted dithienylethene derivative, in which a chlorine atom and a difluoroboron β-diketonate (BF2bdk) group are appended at the termini of the dithienylethene core, is developed. The structure was confirmed by 1H NMR, 13C NMR, and high-resolution mass spectrometry (electrospray ionization). It displayed solvent-dependent photophysical properties, and blue/red light-triggered optical switching behavior in nonpolar or less polar solvents.

Introduction
Recently, photochromic materials have attracted increasing attention due to their potential applications in molecular devices and optical memory storage systems.1–3 As a representative of such materials, dithienylethene derivatives, which can undergo reversible photocyclization reactions, have been extensively studied because of their excellent thermal irreversibility, rapid response and robust fatigue resistance.4–10 However, most dithienylethene compounds encounter the serious restriction of having to be triggered by UV light, which shows highly destructive effects and low penetrability for optical and biological materials. Thus, it is necessary to develop visible light–triggered photochromic dithienylethenes for potential applications in optoelectronics and photopharmacology.11–13 In the last few decades, several approaches have been developed for designing visible light–driven dithienylethenes, for example, extension of the π-conjugation, 14 triplet photosensitizers, 15 upconversion nanoparticles, 16 and intramolecular proton transfer 17 . To the best of our knowledge, difluoroboron β-diketonate (BF2bdk) complexes are typical fluorescent organoboron compounds, which have received increasing attention owing to their rich photophysical properties, such as strong emission in the solution and solid state, large extinction coefficients, two-photon–excited emission, tunable fluorescent emission, and sensitivity to the surrounding medium.18–22 In consideration of their intriguing photophysical properties, our group has recently designed and synthesized a range of visible light–responsive difluoroboron β-diketonate (BF2bdk) complexes, based on a diarylethene unit via Knoevenagel condensation reactions.23–28 Most of the obtained complexes demonstrated excellent visible light–controlled photochromic properties and fluorescence switching behavior in solution, polymethyl methacrylate (PMMA) film, and even in living cells. However, these asymmetric BF2bdk–functionalized dithienylethene derivatives normally required non-trivial multistep syntheses. Therefore, it is highly desirable to develop a versatile building block for dithienylethenes modified with a difluoroboron β-diketonate moiety. 29
Herein, we present a novel chloro-substituted dithienylethene derivative containing a BF2bdk moiety (Scheme 1), in which the chlorine atom and difluoroboron β-diketonate (BF2bdk) group are appended at the two termini of the dithienylethene core. In addition, chlorine group can provide a versatile reaction site for the construction of additional novel visible light–controlled BF2bdk–functionalized photochromic materials. As expected, the dithienylethene compound

Synthetic route and photochromic reactions of dithienylethene
Results and discussion
Synthesis and characterization
The synthesis of chloro- and BF2bdk-substituted dithienylethene derivative
Photophysical properties
Before the blue light irradiation, the photophysical properties of dithienylethene

(a) Normalized UV-Vis absorption spectra of dithienylethene
The photophysical data of dithienylethene

THF: tetrahydrofuran; DMSO: dimethyl sulfoxide; (PSS) Photostationary state.
Absorption maximum.
Extinction coefficients calculated at the absorption maxima.
Fluorescence emission maxima.
Fluorescence lifetime excited with a 405 nm light monitored at the emission maximum, and all fluorescence lifetimes are fitted with single exponential decay.
Fluorescence quantum yield of ring-open isomers determined by a standard method with rhodamine 6G in water (ΦF = 0.75 and λex = 488 nm) as the reference.
Fluorescence quantum yield in photostationary state determined by a standard method with rhodamine 6G in water (ΦF = 0.75 and λex = 488 nm) as reference.
Optical switching behavior
Next, the photochromic properties and fluorescent switching behavior of dithienylethene

(a) Absorption spectral changes of dithienylethene

Fatigue resistance of dithienylethene
Absorption characteristics and photochromic quantum yields of
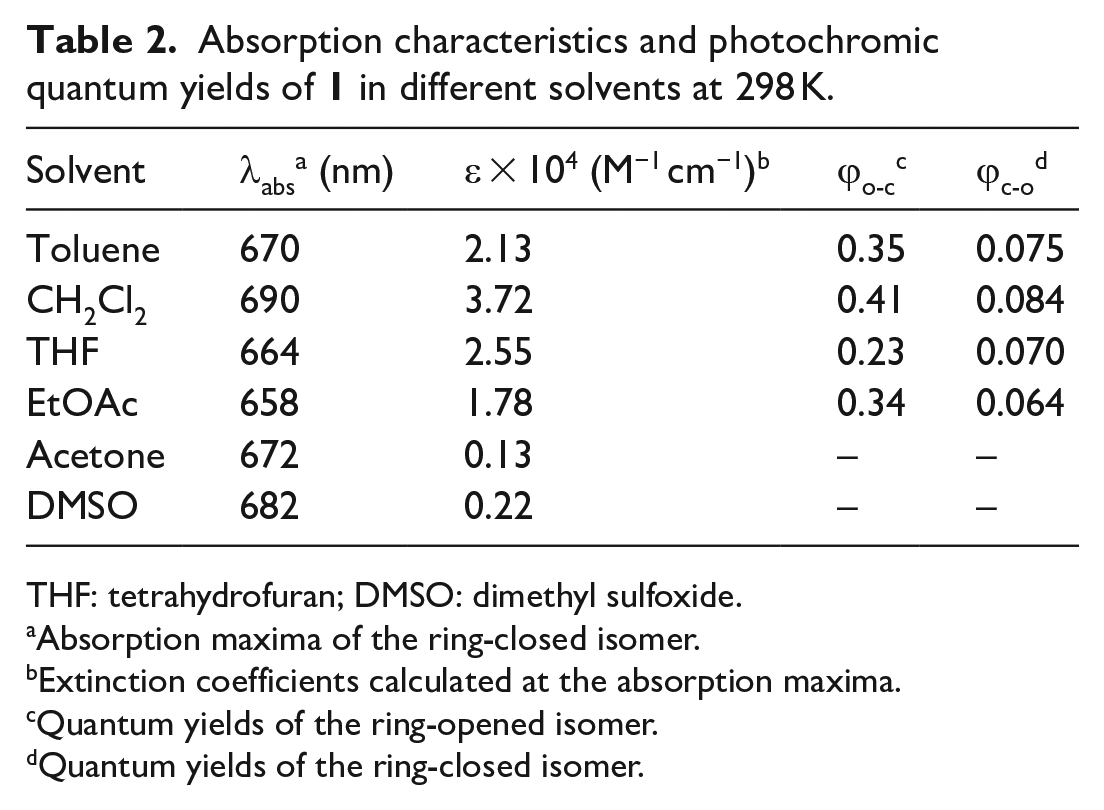
THF: tetrahydrofuran; DMSO: dimethyl sulfoxide.
Absorption maxima of the ring-closed isomer.
Extinction coefficients calculated at the absorption maxima.
Quantum yields of the ring-opened isomer.
Quantum yields of the ring-closed isomer.
Upon alternating irradiation with blue/red light, the fluorescent switching behavior was also investigated in the above solvents at room temperature. As depicted in Figure 4(a), its luminescence intensity at 580 nm in CH2Cl2 gradually decreased along with an obvious fading of the red fluorescence, which was due to efficient energy transfer from the excited difluoroboron β-diketonate moiety to the ring-closed dithienylethene skeleton.
32
On the photostationary state, its luminescence intensity was quenched by ca. 43% with a concomitant decrease of the quantum yield from 0.15 to 0.08 (Table 1). In addition, Figure 4(b) implied decent reversibility based on fluorescence switching cycle. Moreover, upon irradiation with red light at 650–660 nm, the original emission could be restored as a result of the formation of

(a) Emission spectral changes and (b) reversible fluorescence switching (measured at 580 nm) of dithienylethene
Theoretical calculations
To obtain an insight into the relationships between the electronic features and optical properties of dithienylethene

Frontier molecular orbital profiles of the ring-opened isomer
Conclusion
In summary, we have designed and synthesized a novel dithienylethene derivative
Experiments
General methods
All manipulations were carried out under an N2 atmosphere by standard Schlenk techniques unless otherwise stated. THF and toluene were distilled from sodium-benzophenone. 1,2-Bis(5-chloro-2-methylthiophen-3-yl)cyclopent-1-ene
Synthesis of (E)-4-(2-{(4-[(2-(5-chloro-2-methylthiophen-3-yl)cyclopent-1-en-1-yl)]-5-methyl-thiophen-2-yl)}vinyl)-2,2-difluoro-6-phenyl-2H-1,3,2-dioxaborinin-1-ium-2-uide (1 )
To a solution of 1-phenylbutane-1,3-dione
Supplemental Material
sj-pdf-1-chl-10.1177_1747519820951410 – Supplemental material for Chloro- and BF2bdk-substituted dithienylethene: Synthesis, photophysical properties, and optical switching behavior
Supplemental material, sj-pdf-1-chl-10.1177_1747519820951410 for Chloro- and BF2bdk-substituted dithienylethene: Synthesis, photophysical properties, and optical switching behavior by Zhiqiang Lu, Wenbo Yang, Yanhui Bai, Mo Wang, Zixu Li, Yang Yang, Xueling Feng, Yuanxiao Zhu, Xujia Yang and Ziyong Li in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (no. U1304206) and the Innovation and Entrepreneurship Training Program for College students in Henan Province (nos S201910482002, S201910482003, and S201910482021).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
