Abstract
A novel aldehyde-substituted acceptor-DTE-acceptor-type dithienylethene derivative, in which the difluoroboron β-diketonate and aldehyde groups at the termini of the dithienylethene function as electron acceptors, has been developed. The structure was confirmed by 1H NMR, 13C NMR, 19F NMR, high-resolution mass spectrometry (electrospray ionization) and infrared. The derivative displays solvent-dependent photophysical properties, visible light–triggered near-infrared photochromism, and fluorescent switching behavior in solvents of different polarity. Moreover, it can be utilized as a versatile building block to construct novel near-infrared photochromic materials.

Introduction
In recent years, photochromic materials have drawn tremendous attention due to their application in photoswitchable molecular devices and optical memory storage systems.1,2 Photochromic compounds can undergo reversible photochemical reactions by irradiation with light.3–5 Among them, dithienylethene has been extensively studied by virtue of its irreversible thermal stability, high sensitivity, and remarkable fatigue resistance.6–11 However, one serious restriction for dithienylethene compounds in future applications is the necessity to trigger photochromism by ultraviolet (UV) light, which is often damaging to cells and penetrates only partially through most media in vivo. 12 Thus, there is a need to develop near-infrared (NIR) photochromic materials which are triggered by visible light for future applications in optoelectronics and photopharmacology.13–16 In the past few decades, significant progress has been made in visible-light-controlled photochromic compounds.17,18 The most commonly used method to achieve visible light switches is to extend the π-conjugation system,19–21 which can reduce the highest occupied molecular orbital and the lowest unoccupied molecular orbital (HOMO-LUMO) energy gap of the ring-opened isomer and hence achieve NIR photochromic activity. 22 Recently, several alternative approaches have also been explored, such as metal complex systems,23,24 energy up-conversion, 25 electron transfer, 26 multiphoton absorption, 27 and donor-DTE-acceptor systems. 28 Furthermore, the introduction of electron-withdrawing groups on both sides of the dithienylethene provides a versatile route to fabricate acceptor-DTE-acceptor (A-DTE-A)-type photochromic materials, in which the acceptor group can reduce the energy of the ring-opened isomer and make bathochromical shift absorbance. 29 However, we still know very little about these photochromic compounds, thus searching for new scaffolds with visible light–triggered dithienylethenes with A-DTE-A structures is highly desirable.
Difluoroboron β-diketonate (BF2bdk) complexes are a class of highly fluorescent organoboron compounds which have received increasing attention due to their intriguing photophysical properties, such as strong emission in solution and solid state, two-photon-excited fluorescence, large extinction coefficients, intramolecular charge transfer (ICT) character, and environment-sensitive emission.30–34 In addition, the aldehyde group as an electron acceptor (A) unit is widely used in photoelectric materials. Furthermore, it can also function as a classical functional group for further transformations, such as reduction, oxidation, aldol condensation, and Knoevenagel condensation. Herein, we present a novel aldehyde-substituted A-DTE-A-type dithienylethene

Synthetic route and design rationale of dithienylethene
Result and discussion
Synthesis and characterization
The stepwise synthesis of aldehyde- and BF2bdk-substituted A-DTE-A-type dithienylethene derivative
Photophysical properties
The photophysical properties of dithienylethene

(a) Normalized UV-Vis absorption spectra of
The photophysical data of

THF: tetrahydrofuran; DMSO: dimethyl sulfoxide.
Absorption maximum.
Extinction coefficients calculated at the absorption maxima.
Fluorescence emission maxima.
Fluorescence quantum yield determined by a standard method with rhodamine 6G in water (Φf = 0.75, λex = 488 nm) as reference.
Subsequently, the photoisomerization behaviors of dithienylethene
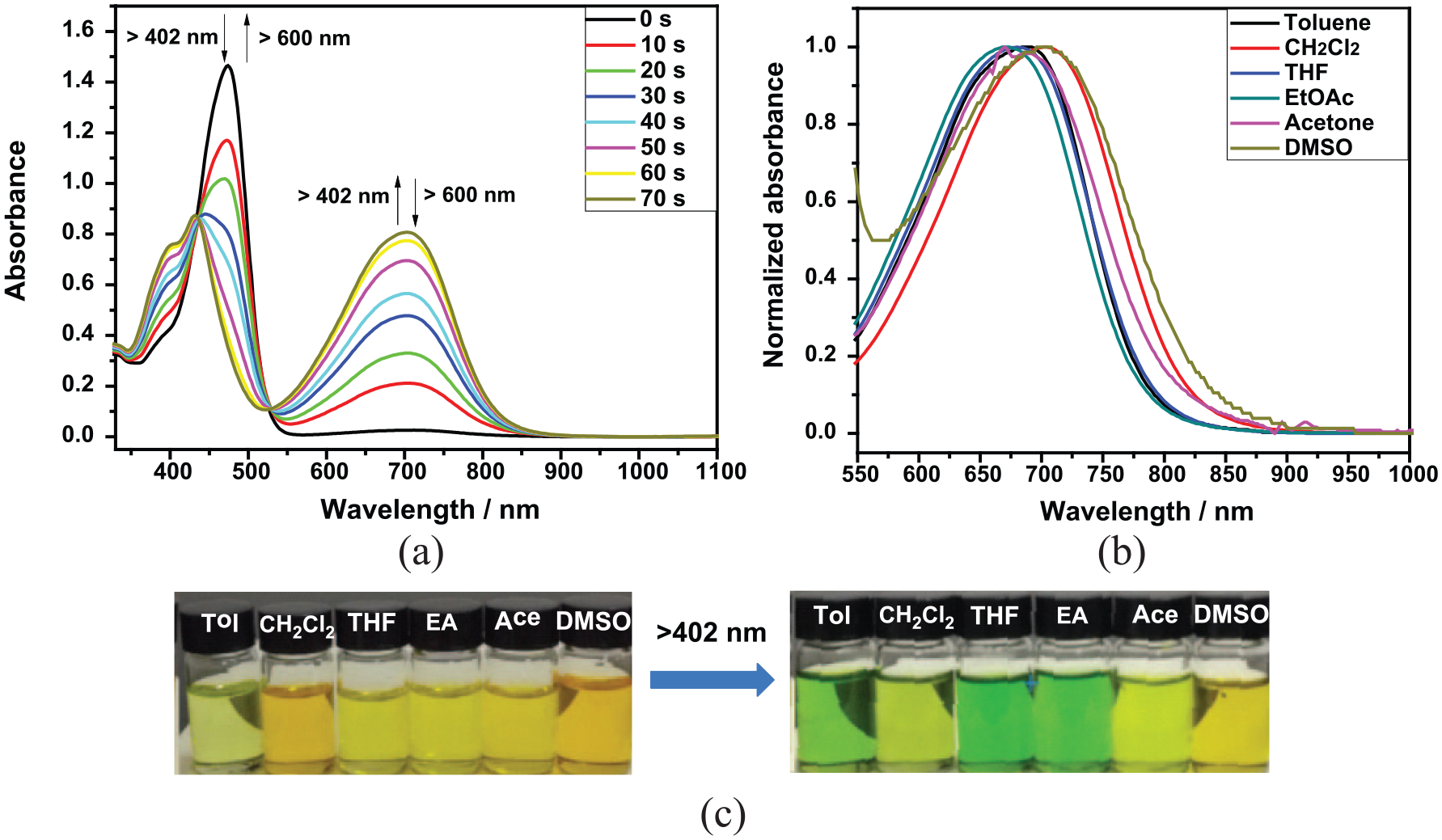
(a) Absorption spectral changes of dithienylethene
Absorption characteristics and photochromic quantum yields of

THF: tetrahydrofuran; DMSO: dimethyl sulfoxide.
Absorption maximum of ring-closed isomers.
Extinction coefficients calculated at the absorption maxima.
Quantum yields of ring-opened isomers.
Quantum yields of ring-closed isomers.
The photochromic behavior of dithienylethene
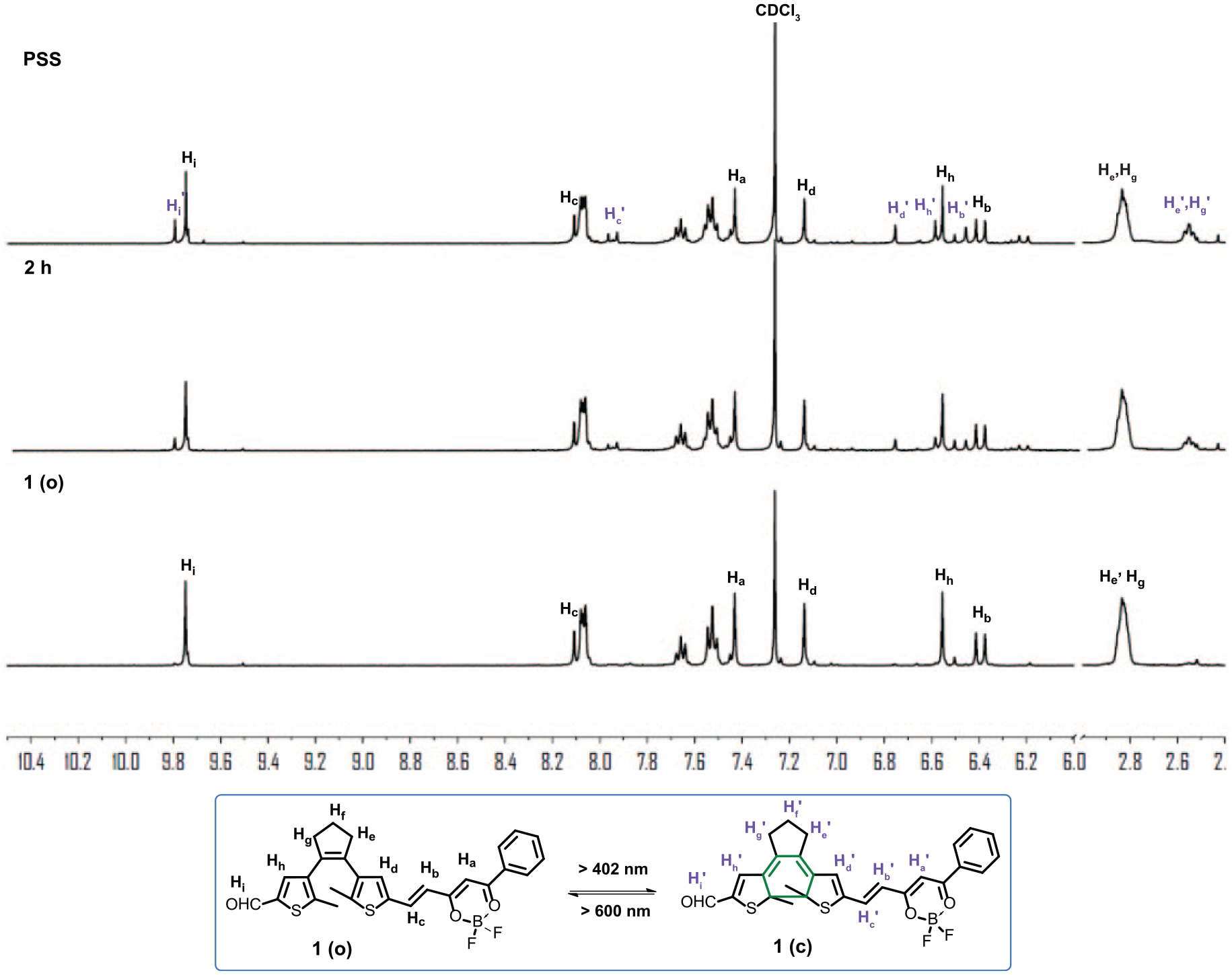
Partial 1H NMR spectral changes of
The fluorescent switching behavior of dithienylethene

(a) Emission spectral changes of of dithienylethene
Conclusion
In summary, a novel aldehyde- and BF2bdk-substituted dithienylethene derivative
Experiments
General methods
All manipulations were carried out under a nitrogen atmosphere by standard Schlenk techniques unless otherwise stated. THF and toluene were distilled from sodium-benzophenone. DMF was dried with magnesium sulfate and then distilled under vacuum. 1,2-Bis(5-chloro-2-methylthiophen-3-yl)cyclopent-1-ene (
Synthesis of (E)-(2-{5-[2-(2,2-difluoro-6-phenyl -2H-1λ, 3, 2λ-dioxaborinin-4-yl)vinyl}-2- methyl-thiophen-3-yl]cyclopent-1-en-1-yl)-5-methylthiophene-2-carbaldehyde (1)
A solution of 1-phenylbutane-1, 3-dione (
Supplemental Material
Suppl_file – Supplemental material for Aldehyde-substituted acceptor-DTE-acceptor-type dithienylethene as a versatile building block for near-infrared photochromic materials
Supplemental material, Suppl_file for Aldehyde-substituted acceptor-DTE-acceptor-type dithienylethene as a versatile building block for near-infrared photochromic materials by Yangyang Wang, Ming Qin, Yufei Song, Yingying Pei, Yijie Dai, Yongjie Ma and Ziyong Li in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge financial support from the Key Scientific Research Project of Higher Education of Henan Province (grant nos. 18A150012 and 18A550012) and the Science and Technology Project of Henan Province (grant no. 182102310668).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
