Abstract
An efficient copper-catalyzed strategy for the synthesis of α-ketoamides via cross-coupling of methyl ketones and pyridin-2-amines is described. This transformation has provided a simple process for the formation of C−N and C=O bonds to prepare α-ketoamides, which are important substrates and intermediates for the preparation of fine chemicals. The reaction mechanism is investigated, which suggests that the reaction proceeds via a radical pathway.

Introduction
Amides are very common in nature and technology as structural materials, and exhibit a wide range of biological functionalities. 1 Many drugs contain amide moieties, including paracetamol, amoxicillin, penicillin, zolpidem, and cefpimizole (Figure 1). Such compounds have attracted the attention of scientists because of their important applications in pharmaceuticals, natural products, agrochemicals, and biologically active molecules. 2 Therefore, it is not surprising that significant effort has been directed toward developing synthetic transformations3–5 for the preparation of amides. Several classic and successful synthetic approaches, such as the Beckmann, Ritter, Ugi, and Staudinger reactions, have been developed for the synthesis of amide derivatives. Recently, transition-metal-catalyzed reactions have become powerful tools for the formation of carbon-nitrogen bonds to prepare amides.6–25 Ahmed and colleagues 26 reported a unique dimethyl sulfoxide (DMSO)-promoted oxidative amidation approach for synthesis of α-ketoamides from 2-oxoaldehydes and aliphatic amines (Scheme 1(a)); Zhang and Wang 27 and Wan and colleagues 28 independently developed a facile TBHP/I2-promoted oxidative coupling reaction of acetophenones with aliphatic amines for the synthesis of α-ketoamides (Scheme 1(b)); Kaliappan and colleagues 29 has described a one-pot copper-catalyzed biomimetic route to N-heterocyclic amides from methyl ketones and pyridin-2-amines (Scheme 1(c)). Although numerous investigations in this field have been conducted, the development of a new strategy is still highly desirable for the construction of α-ketoamides, which are an important class of amide compounds with the general structure (R1COCONR2).

Important amides.

Methods for the synthesis of amides.
Very recently, we have developed approaches for the formation of C–C, C–N, and C–O bonds to synthesize heterocycles.30–38 Our current interest is focused on the formation of C–N and C–O bonds in order to synthesize N-(2-pyridyl)-α-ketoamides from methyl ketones and pyridin-2-amines (Scheme 1(d)).
Results and discussion
In our initial study, pyridin-2-amine (
Optimization of the reaction conditions. a .

Reaction conditions:
Determined by gas chromatography (GC) analysis.
Based on the optimized reaction conditions, the substrate scope of the oxidative coupling reaction for the synthesis of α-ketoamides was then studied. The results are described in Table 2. The oxidative coupling reaction of
Cu-catalyzed synthesis of amides. a
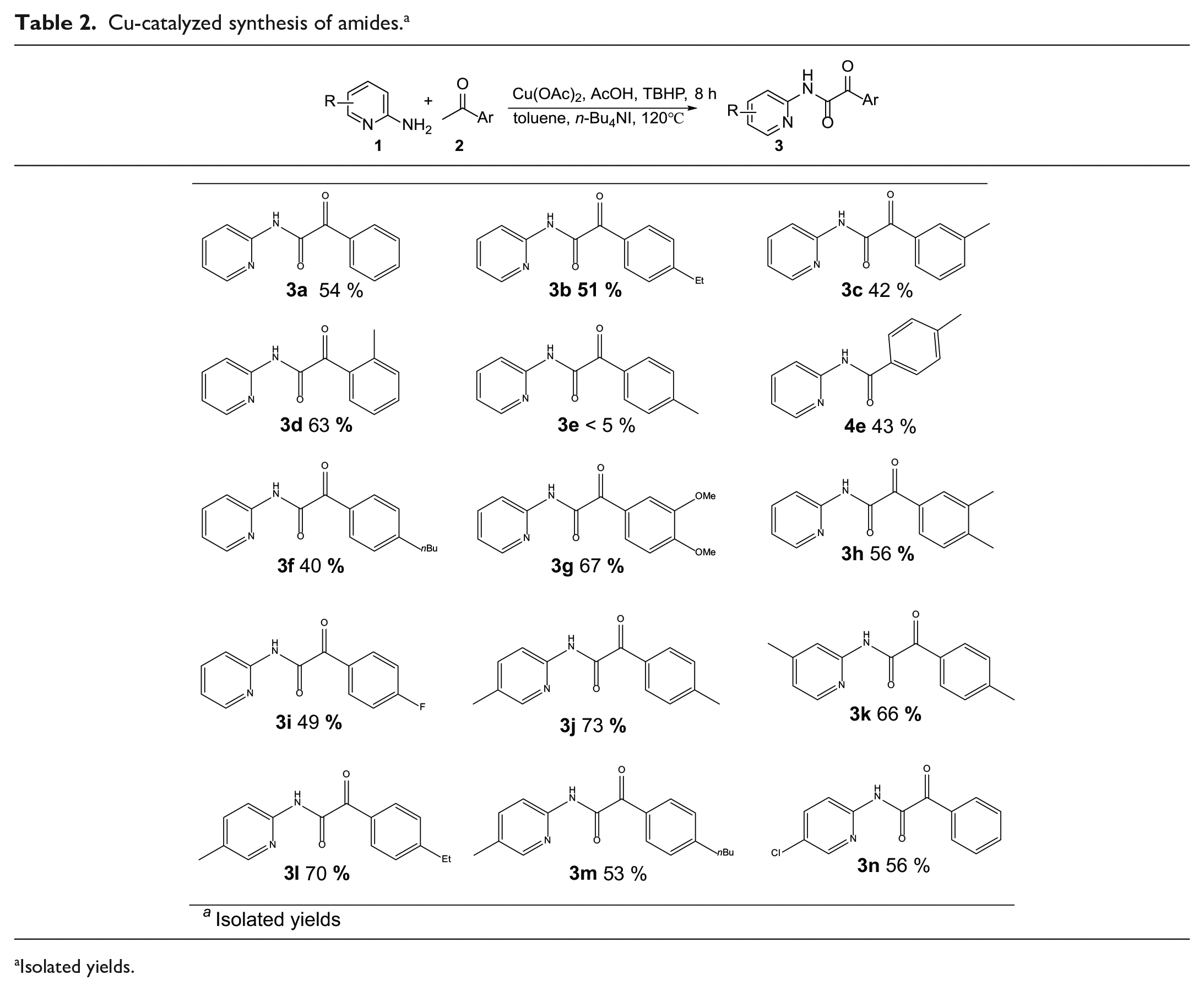
Isolated yields.
Mechanism
To gain insight into the mechanism of the Cu-catalyzed transformation, control experiments were performed. To prove that an organic radical species was involved in the reaction, we carried out the radical trapping reactions by adding a radical-trapping reagent (TEMPO) (Scheme 2(a)). The result indicated that the reaction had been inhibited and that a radical process was involved in this Cu-catalyzed strategy. In addition, the reaction of

Control experiments.
On the basis of the above experiment results, a plausible mechanism is described in Scheme 3. Initially, radical intermediate

Proposed mechanism.
Conclusions
In conclusion, we have developed a novel and straightforward Cu-catalyzed reaction to prepare amides via oxidative coupling of methyl ketones and pyridin-2-amines. This strategy represents a simple process for the formation of C−N and C=O bonds and provides a new route for the synthesis of α-ketoamides which are common structural motifs in natural products and pharmaceuticals. The mechanism was investigated, which suggested that the reaction occurs via a radical pathway. Further studies on the applications and development of amides are underway in our laboratory.
Experimental section
Commercially available chemicals were purchased from commercial sources and used without further purification. Fourier transform infrared spectra (FTIR) were recorded on a Perkin-Elmer Spectrum 100 Series with pressed KBr pellets. The 1H and 13C NMR spectra were recorded with a Bruker Avance 400 MHz spectrometer (100 MHz for carbon). Mass spectra recorded were obtained on an electrospray ionization mass spectrometry (ESIMS). Elemental analyses were performed with an elemental analyzer. GC-MS was obtained using electron ionization. Thin-layer chromatography (TLC) was performed using commercially prepared 100–400 mesh silica gel plates.
Synthesis of
Supplemental Material
sj-pdf-1-chl-10.1177_1747519820950222 – Supplemental material for Cu-catalyzed cross-coupling of methyl ketones and pyridin-2-amines for the synthesis of N-(2-pyridyl)-α-ketoamides
Supplemental material, sj-pdf-1-chl-10.1177_1747519820950222 for Cu-catalyzed cross-coupling of methyl ketones and pyridin-2-amines for the synthesis of N-(2-pyridyl)-α-ketoamides by Ali Chen, Daji Yang, Yue Yu, Xiang Liu, Chuixiong Rao, Haoming Lin and Pengfeng Guo in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was financially supported by the National Natural Science Foundation of China (21302023), the Innovation and Strong School Project of Guangdong Pharmaceutical University (2014KTSCX120, 2015cxqx212), the Special Fund for Science and Technology Innovation Strategy of Guangdong Province (pdjh2020a0299), the Project of Innovation for Enhancing Guangdong Pharmaceutical University, Provincial Experimental Teaching Demonstration Center of Chemistry & Chemical Engineering, and Special Funds of Key Disciplines Construction from Guangdong and Zhongshan Cooperating.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



