Abstract
2,2’-Bipyridine with strong coordination ability for metal ions was used to prepare N-doped porous carbon materials by a facile method of solution agitation and annealing, with KOH as the activated solvent. Applied to supercapacitors, these porous carbon materials exhibit high specific capacitance and outstanding rate performance, which are attributed to their larger specific surface area, richer pore structure, and N-doping after KOH activation, leading to fast electron transport and increased ion storage in the unique architecture.
Keywords
A nitrogen-containing heterocyclic ligand called 2,2’-bipyridine, possessing strong coordination ability with metal ion, was used to prepare nitrogen-doped porous carbon by a facile method of solution agitation and KOH activation at various annealing temperatures. Applied to supercapacitors, this porous carbon exhibits a good cycle life.

Introduction
Supercapacitors (SCs) are energy storage devices and the electrode material affects their performance immensely. Porous carbon materials have been researched extensively due to their good electrical conductivity, chemical inertness, environmental friendly nature, and so on.1,2 They also serve important roles as electrode materials for SCs. There are numerous carbon sources such as biomass,3,4 polymers,5,6 and metal organic frameworks (MOFs),7,8 but they usually have small specific surface areas (SSAs) and poor electrochemical performance, and they are employed as electrode materials after activation, such as via physical and chemical activation. Vijayakumar et al. 9 reported an activated porous carbon fiber, made from waste cotton, using the KOH-activation method, which exhibited excellent capacitance and long charge–discharge cycles. Wang et al. 10 prepared a nitrogen-doped carbon-based material derived from a covalent triazine framework and KOH acted as the pore former. Yu et al. 11 synthesized three-dimensional (3D) interconnected porous carbons derived from MOF-5 and KOH as the activator. Furthermore, heteroatoms, especially nitrogen-containing carbon materials, have been widely used due to their distinct electrochemical and mechanical properties, but sometimes additional nitrogen-doping steps are required to synthesize nitrogen-containing carbon materials.
Dipyridine and its derivatives have good σ-electron-donating ability and π-electron-accepting ability. They can form stable complexes with various metal ions due to their strong coordination ability, and they are excellent chelating ligands in modern coordination chemistry. They are widely used in many fields such as photomolecular devices, antineoplastic drugs, and supramolecular self-assembly, but there are fewer applications as electrode materials in SCs. 12
In this work, we describe the preparation of activated dipyridyl compound–derived carbons (ADCDCs) via a facile approach. Compared with the solvothermal method which usually needs a long reaction period and strict reaction conditions, the Zn-based dipyridine coordination compound is easily produced under simple stirring owing to the strong coordination ability between the N atoms in dipyridine with Zn2+. Moreover, an additional nitrogen-doping step is not required owing to the intrinsic nitrogen atom in dipyridyl. In addition, KOH is employed to activate the resulting product without any extra step. Finally, nitrogen-doped porous carbon materials are prepared by heat treatment. The resulting carbon materials possess stable structures and abundant pore size distributions. Applied as an SC, this porous carbon electrode exhibits a good specific capacitance and a stable long-term cycling performance.
Results and discussion
Scheme 1 shows a representation of the fabrication process for ADCDCs. Here, 2,2’-dipyridine is an important bidentate chelating ligand, and three of them can ordinate with Zn2+. Subsequently, nitrogen-doped porous carbon materials (ADCDCs) can be prepared after KOH activation and annealing. The X-ray diffraction (XRD) analysis is shown in Figure 1. ADCDC-n650 (non-activated dipyridine carbon compound prepared at 650 °C) and ADCDC-650 (activated dipyridine carbon compound prepared at 650 °C) are amorphous owing to existence of weak and broad peaks (002) at 25°–30° and 40°–45°. 11 Obviously, K2CO3 (PDF#16-0820) and ZnO (PDF#36-1451) are present in the unwashed material, and ZnO exists in the ADCDCs only washed with deionized (DI) water, while ADCDC-650 was prepared after washed with HCl and DI water. These phenomena can be explained according to equation (1) 13
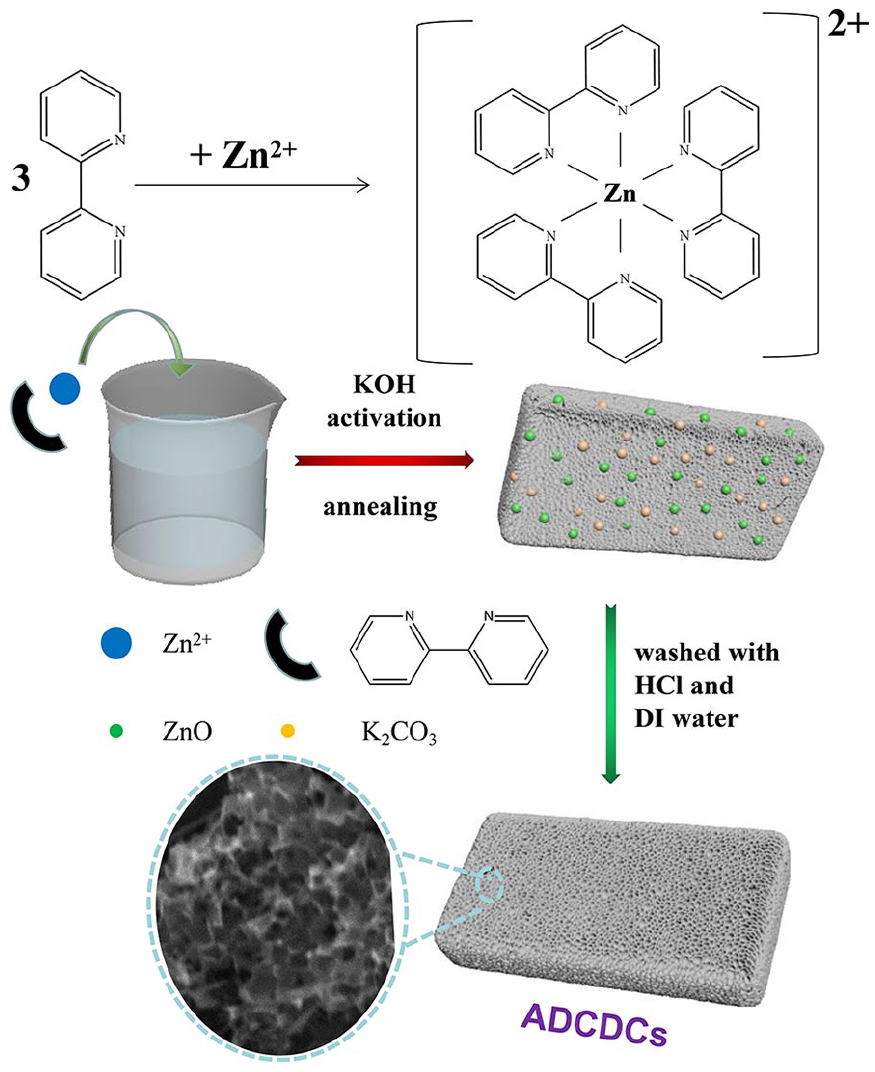
Fabrication of the ADCDCs.
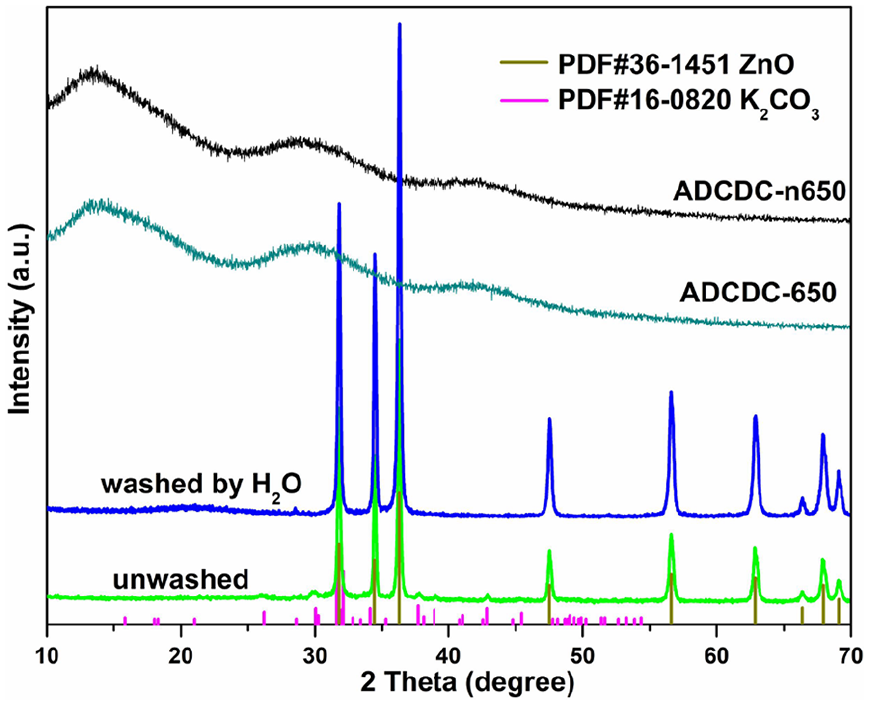
XRD patterns in the preparation process of ADCDC-650.
The elemental composition of ADCDC-650 was determined from the X-ray photoelectron spectroscopy (XPS) spectrum in Figure 2(f); the C, N, and O contents of the ADCDC-650 are 87.09%, 1.15%, 11.76%, respectively. The morphologies of these carbon materials were observed by scanning electron microscope (SEM) and transmission electron microscope (TEM). Figure 2(a) shows that ADCDC-n650 exhibits a relatively smooth structure. Obviously, the surface of ADCDCs displays different pore structures based on the different processing temperatures. ADCDC-600 shows a porous structure (Figure 2(b)), and ADCDC-650 displays a more abundant porous structure (Figure 2(c)). Unfortunately, ADCDC-700 has a degraded porous structure (Figure 2(d)), which is most likely attributed to the collapse in part of the structure at a higher annealing temperature of 700 °C. Figure 2(e) demonstrates the rich porous structure of ADCDC-650. 14 Simultaneously, N-doping is beneficial for faster ion transport. 15

SEM images of (a) ADCDC-n650, (b) ADCDC-600, (c) ADCDC-650, (d) ADCDC-700, (e) TEM image, and (f) XPS spectra of ADCDC-650.
The N2 adsorption–desorption isotherm pore size distribution of ADCDC-650 and ADCDC-n650 is shown in Figure 3. The SSA of ADCDC-650 is 962 m2 g−1, which is far larger than that of ADCDC-n650. The pore size distribution (inset) demonstrates that ADCDC-650 is a mesoporous carbon, the larger SSA, total pore volume (V), and more appropriate pore structure (Da) of which lead to its better performance as an SC (Table 1).

N2 adsorption–desorption isotherm and pore size distribution (inset) of ADCDC-650.
Porosity parameters of three ADCDCs.
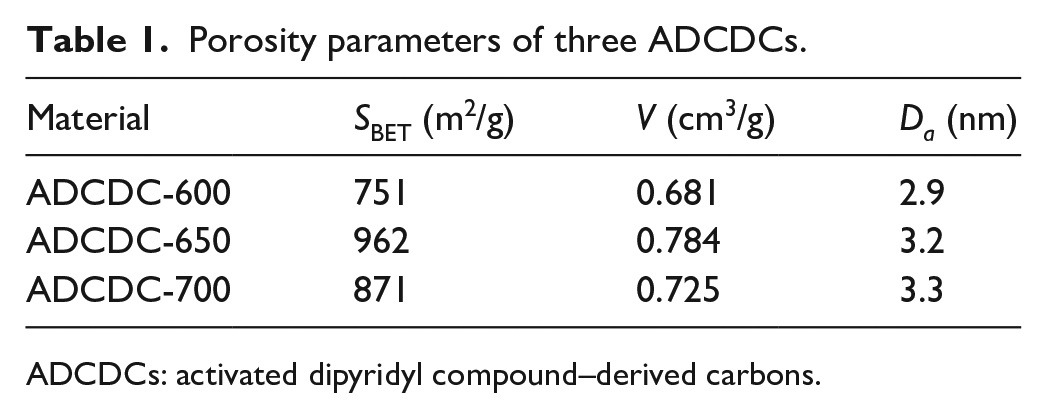
ADCDCs: activated dipyridyl compound–derived carbons.
Figure 4(a) shows cyclic voltammogram (CV) curves of ADCDCs with various annealing temperatures at 50 mV s−1. The areas surrounded by the curves represent the specific capacitance of electrode materials. Obviously, ADCDC-650 possesses a higher specific capacitance than the other materials due to its more appropriate pore structure after activation. Figure 4(b) shows the galvanostatic charge–discharge (GCD) curves of ADCDCs with different annealing temperatures measured at 1 A g−1. All four electrode materials exhibit almost inverted triangular GCD curves in the potential range of −1 to 0 V, further exposing the majority of electric double layer capacitor (EDLC) characteristic of all the electrode materials. 16 The specific capacitance of electrode materials can be calculated according to equation (2) 17
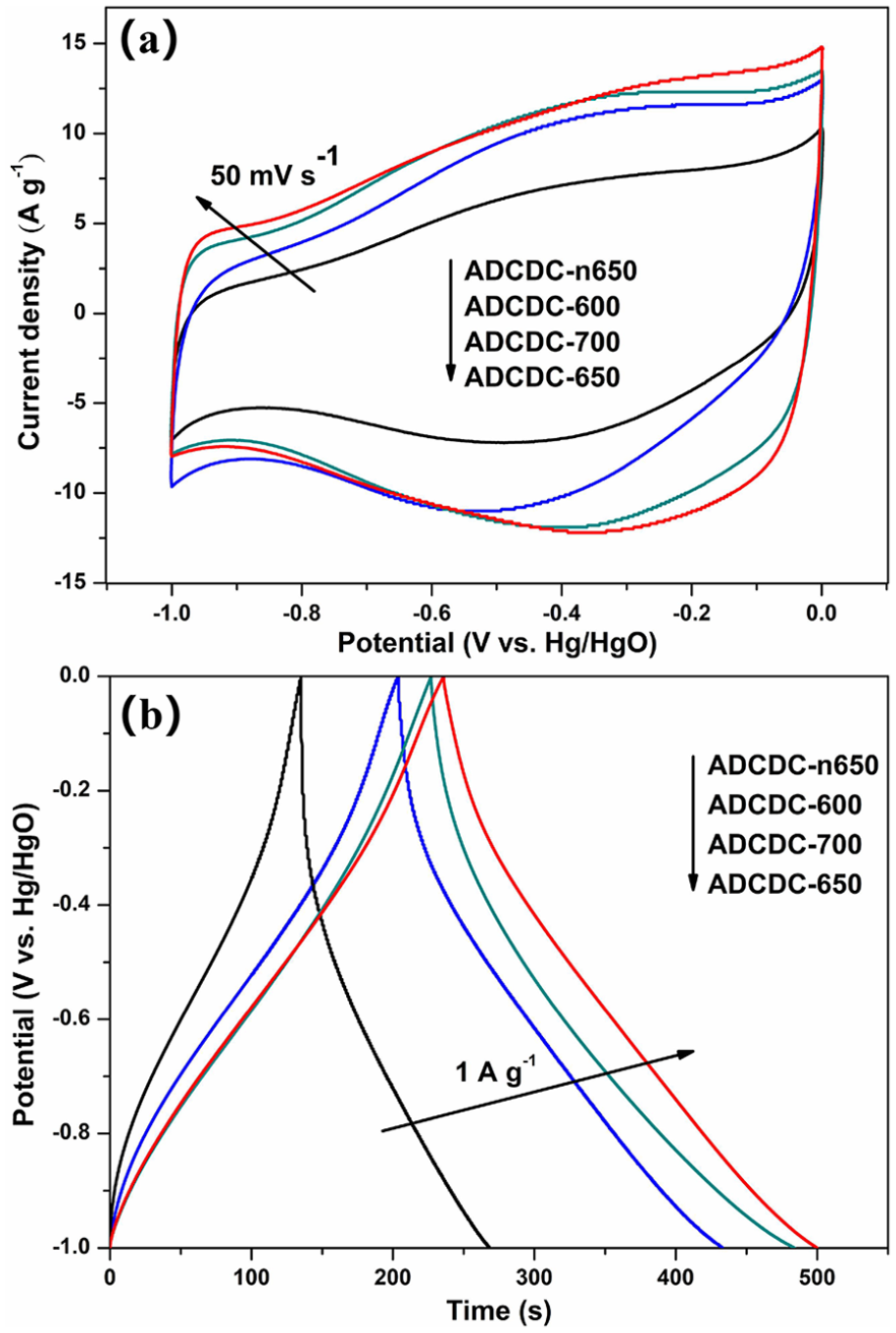
(a) CV curves and (b) galvanostatic charge–discharge curves of carbon materials with various annealing temperatures.

where Cm is the mass specific capacitance, I is the discharge current, Δt is the discharge time from 0 to −1 V, m is the mass of active electrode materials, and ΔV is the voltage difference within discharge time. As can be seen from Figure 5, not only ADCDC-650 exhibits highest specific capacitance at 3 A g−1 but also possesses excellent capacitance retention with increasing current density. Figure 6 shows electrochemical impedance spectroscopy (EIS) of ADCDCs in the frequency range from 100 kHz to 10 MHz at open-circuit potential. The inset expresses the similar low x-intercept of ADCDCs and the small diameter of the semicircle. In particular, ADCDC-650 has a higher slope, demonstrating typical capacitor behavior compared to the other three electrode materials. These results reveal low internal resistance, fast electron/ion transport, and excellent electrical conductivity for ADCDC-650. 18

The values of the specific capacitances of carbon materials with various annealing temperatures.

Electrochemical impedance spectroscopy (EIS) of ADCDCs with various annealing temperatures.
In view of the excellent electrochemical performance of the ADCDC-650 electrode, further measurements on this material were recorded. Figure 7(a) shows the CV curves of ADCDC-650 at 5, 10, 20, 50, and 100 mV s−1, respectively. All the CV curves exhibit superior mirror symmetry, certifying the excellent cyclic performance of the material. With the improvement of scan rate, the shapes of the CV curves remain nearly the same, which is favorable for an electrode to charge and discharge at large current. Figure 7(b) reveals the GCD curves of ADCDC-650 at different current densities. Typical triangular shapes illustrate significant coulombic efficiencies and excellent capacitive performance.

(a) CV curves at different scan rates and (b) specific capacitance at different current densities for ADCDC-650.
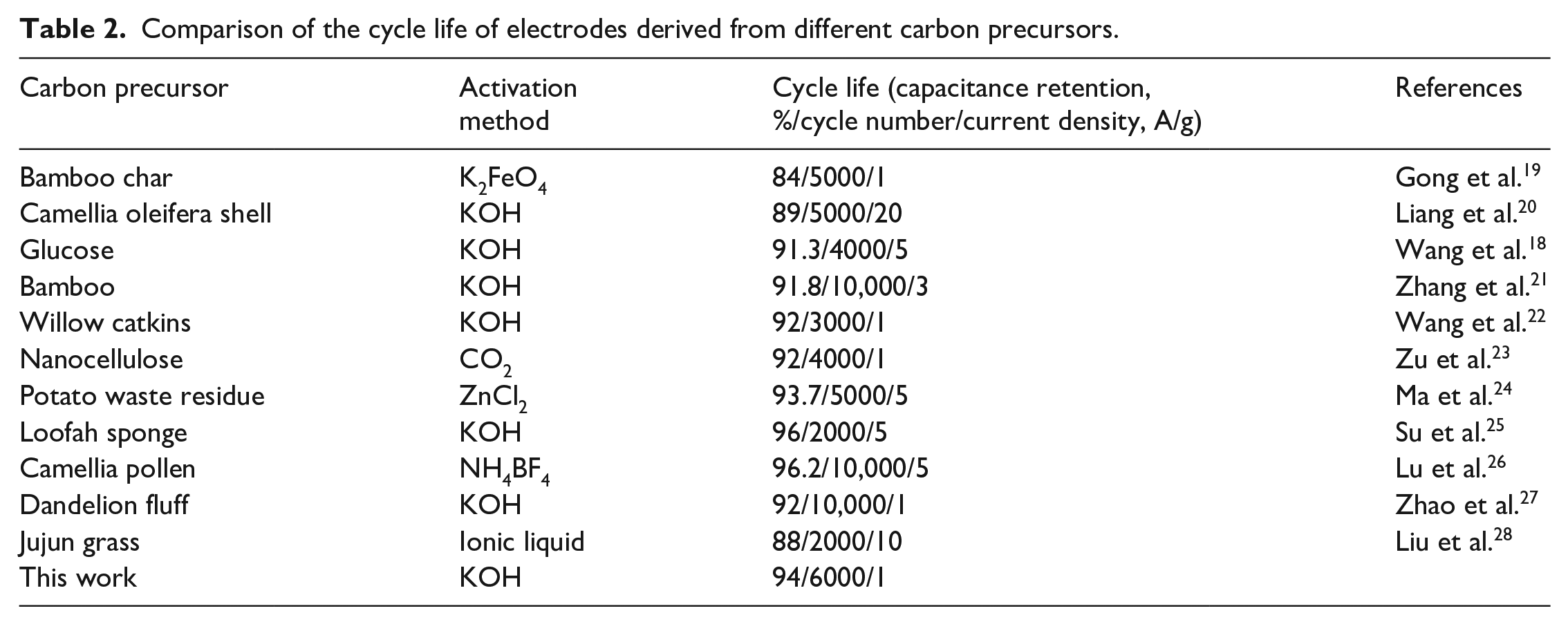
The specific capacitance of ADCDC-650 as an electrode can reach an excellent value of 305 F g−1 at 0.5 A g−1, 265 F g−1 at 1 A g−1, 219 F g−1 at 3 A g−1, 200 F g−1 at 5 A g−1, and even 170 F g−1 at 10 A g−1 (Figure 7(b)), which proves a good capacitance retention. Figure 8 shows cycle life of the ADCDC-650 electrode at 1 A g−1; the capacitance retention remains 94% after 6000 cycles. Compared to some other carbon precursors listed in Table 2, the ADCDC-650 electrode possesses a good electrochemical cycling performance, but there are still some porous carbon materials with better cycling lifetimes, which demonstrates that ADCDC-650 may have some disadvantages in structure and electrochemical performance.29,30
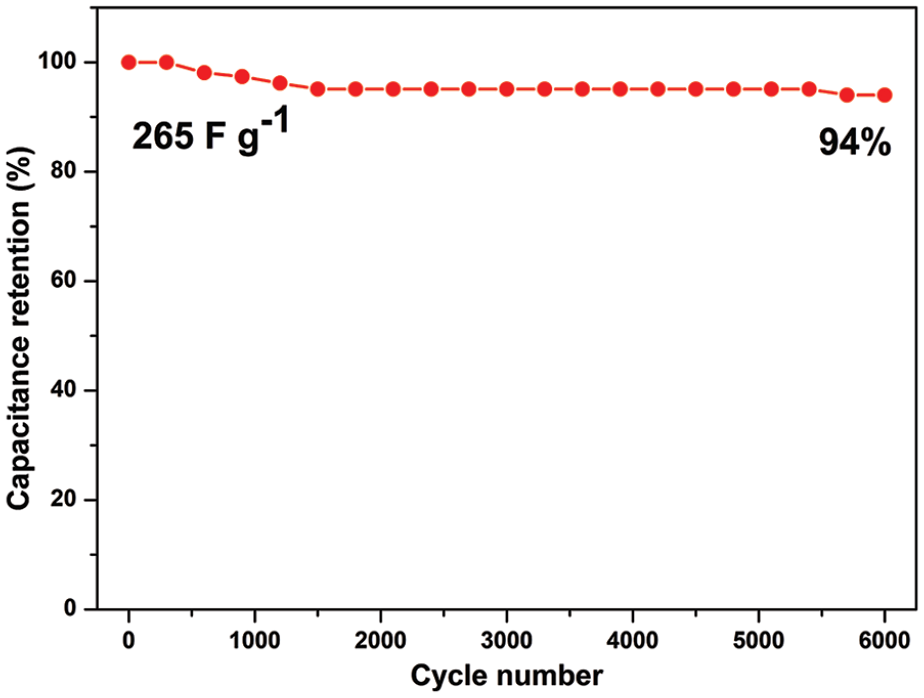
Long cycling performance of ADCDC-650 at 1 A g−1 over 6000 cycles.
Comparison of the cycle life of electrodes derived from different carbon precursors.
Conclusion
In summary, nitrogen-doped porous carbon materials have been synthesized by a facile method of mixed solutions based on the strong coordination ability of dipyridine and chemical activation using KOH at different annealing temperatures in this paper. ADCDC-650 has a large SSA, abundant pore structures, and nitrogen-doping induced defects, which facilitates more storage and a shorter transport distance for electrolyte ions. Applied as an SC, it exhibits a high specific capacitance of 305 F g−1 at 0.5 A g−1 and a stable cycling performance (94% retention after 6000 cycles). This research offers a facile, fast, and feasible route for the synthesis of a porous activated carbon derived from nitrogen-containing heterocyclic complexes and describes its application as an electrode material in SC.
Experimental
Chemicals
All chemicals were purchased as follows: 2,2’-bipyridyl (99%, Aladdin Chemical Reagent, Co., Ltd, Shanghai, China), dimethylformamide (DMF, 99%, Sinopharm Chemical Reagent Co., Ltd, Shanghai, China), zinc nitrate hexahydrate (AR, Aladdin Chemical Reagent Co., Ltd, Shanghai, China), potassium hydroxide (AR, Hubei University Chemical Factory, Wuhan), and absolute ethanol (AR, Sinopharm Chemical Reagent Co., Ltd, Shanghai, China).
Preparation of ADCDCs
2,2’-Bipyridyl (0.0036 mol, 0.5622 g) was dissolved in DMF (10 mL) under constant stirring at room temperature. Next, 0.12 M Zn(NO3)2·6H2O aqueous solution (10 mL) was added dropwise to the above solution. After 30 min, 1 M KOH solution (30 mL) was added to the above solution and continued stirring was for 30 min. The solution was heated until the solvent had evaporated. After that, the obtained solid mixture was annealed in a tubular furnace from room temperature to 650 °C at a heating rate of 5 °C min−1 and maintained at 650 °C for 2 h under an Ar atmosphere. After cooling to room temperature, the mixture was washed with HCl, DI water, and absolute ethanol (three times each). After 12 h of drying, the activated dipyridine compound derived carbon was obtained and named ADCDC-650. For comparison, three other materials were synthesized by an identical method and named ADCDC-n650 (non-activated by KOH), ADCDC-600, and ADCDC-700, respectively.
Characterization
The microscopic morphology of the carbon materials was studied using an SEM (JSM-6510LV, Japan) and a TEM (FEI-Tecnai-G20, Hillsboro, OR, USA). XRD patterns were collected using a Bruker D8 Advance diffractometer with Cu-Kα radiation. Nitrogen adsorption and desorption isotherms were measured on a Micromeritics ASAP-2020 apparatus. XPS spectra were recorded on an ESCALAB 250Xi spectrometer using monochromatic Al Ka radiation. The Brunauer–Emmett–Teller (BET) plot of the nitrogen adsorption isotherm and pore size distribution curves were recorded with a Quantachrome Instruments (Autosorb-iQ, Boynton Beach, FL, USA).
Electrochemical measurement
All eletrochemical measurements including CV curves, GCD curves, and EIS were obtained from an electrochemical working station (CHI 660E; Chenhua, Shanghai, China) using a typical three-electrode cell at room temperature. The working electrode was fabricated by mixing carbon material, acetylene, and polytetrafluoroethylene binder based on the ratio of 8:1:1, and then pressed onto Ni foam (about 1 cm2) under 10 MPa. A platinum wire and Hg/HgO electrode were used as the counter and reference electrodes, respectively. 6 M KOH solution was used as the electrolyte for the system.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

