Abstract
An electroactive nitrogen-doping tin dioxide nanorod array (N-SnO2 NRA) is designed as an effective energy-storage electrode material for supercapacitor applications. N-SnO2 supported on a carbon fiber substrate is prepared using SnCl4 as a precursor through hydrolysis, hydrothermal growth, and an NH3-nitriding process. Electroactive N-SnO2 is formed by an N-doping reaction between Sn(OH)4 and NH3, revealing a high nitrogen-doping level of 12.5% in N-SnO2. N-SnO2/carbon fiber reveals a lower ohmic resistance and charge transfer resistance than SnO2/carbon fiber, which is consistent with its higher current response and lower voltage drop in electrochemical measurements. N-SnO2 NRA has an independent nanoarray structure and a small side length of a quadrangular nanorod, contributing to a more accessible interspace, reactive sites, and feasible electrolyte ion diffusion. The N-SnO2/carbon fiber NRA electrode shows higher specific capacitance (105.4 F g−1 at 0.5 A g−1) and rate capacitance retention (45.0% from 0.5 to 5 A g−1) than a SnO2/carbon fiber NRA electrode (58.6 F g−1, 38.4%). Significantly, the cycling capacitance retention after 2000 cycles increases from 78.1% of SnO2/carbon fiber to 98.8% of N-SnO2/carbon fiber, presenting a superior electrochemical cycling stability. The N-SnO2 supercapacitor maintains stable power working at an output voltage of 1.6 V. The specific capacitance decreases from 75.2 to 55.1 F g−1 when the current density increases from 1 to 10 A g−1. The corresponding energy density decreases from 24.23 to 9.81 Wh kg−1, presenting a reasonable rate capability. So, the prepared N-SnO2 nanorod array demonstrates superior capacitance performance for energy-storage applications.
An electroactive nitrogen-doping SnO2 nanorod array is supported on carbon fiber to form N-SnO2/carbon fiber nanorod array electrode, which exhibits high electrochemical energy-storage performance.

Introduction
A supercapacitor as an effective energy-storage device is usually classified as an electric double-layer capacitor and a pseudocapacitor.1–3 Carbon fiber (CF) has attracted significant attention due to its high stiffness, high tensile strength, low weight, and high chemical inertness.4,5 However, one challenging aspect of a CF electrode material is how to increase the active sites for driving electrochemical reaction since the electron transfer process occurs only at the interface between the solid electrode and the aqueous or gel electrolyte.6,7 Surface modification would be an efficient strategy to improve the electrochemical properties of CF.8–11 Pseudocapacitive materials such as metal oxides and conductive polymers are well introduced into CF.12–16 Electroactive materials are capable of rapid reversible redox reactions occurring on the CF surface, which could well enhance the capacitive performance of CF composite electrode.17–19 Transition metal compounds have been investigated in detail as electroactive supercapacitor electrode materials.20–22 Tin dioxide (SnO2) is an n-type semiconductor with a wide band gap of 3.6 eV and a low electron affinity. SnO2 has a high theoretical capacity of 780 mAh g−1, attracting considerable attention in supercapacitor applications.23,24 However, the weak electronic conductivity of SnO2 has restrained its applications in high-performance energy-storage systems.25,26 SnO2 composites using conductive materials usually show improved capacitance performance.27,28 SnO2/CFs have been studied in detail as electroactive electrode material of lithium-ion batteries.29,30 Doping of SnO2 with appropriate elements can directly change its electronic structure and effectively modulate its electronic properties, accordingly improving the capacitance properties for supercapacitor applications.31,32 Nitrogen has been proposed as a good p-type dopant to improve the electronic conductivity of SnO2 and enhance the electrochemical capacitance contributed by reversible Faradaic reactions. It is reported that N-doped SnO2 subsequently has higher electrochemical performance than pristine SnO2.33–35 Experimental and theoretical investigations on N-doped SnO2 also demonstrate consistent results for the improvement of electrochemical properties. 36
In this study, nitrogen-doping tin dioxide nanorod array (N-SnO2 NRA) is designed, fabricated, and supported on a CF substrate, forming a novel N-SnO2/CF NRA electrode.28,37 The N-SnO2/CF NRA electrode with high nitrogen-doping and an independent nanoarray structure improves the conductivity and electroactivity. The N-SnO2/CF NRA electrode is expected to have high electrochemical performance. This N-SnO2/CF NRA electrode is also used to construct an N-SnO2 supercapacitor, while maintaining high compatibility with the proton acid polymer gel electrolyte in solid-state supercapacitor applications for effective energy storage.
Results and discussion
Preparation process
Figure 1(a) shows the schematic illustrating the preparation process of N-SnO2/CF electrode. The N-SnO2 NRA supported on CF substrate is prepared using SnCl4 as a precursor through a hydrolysis process, a hydrothermal process, and an NH3-nitriding process. Figure 1(b) shows a photograph of the SnO2/CF and N-SnO2/CF NRA electrodes. Both the SnO2/CF and N-SnO2/CF NRA electrodes present a colorful appearance when they are grown on CF through the hydrothermal process. Comparatively, the N-SnO2/CF NRA electrode presents slighter gray appearance compared to the SnO2/CF NRA electrode, which is related to the nitriding treatment of SnO2 NRA. A solid-state asymmetric N-SnO2 supercapacitor was then constructed using the N-SnO2/CF NRA electrode as a positive electrode, TiN/CF as a negative electrode, and H2SO4–polyvinyl alcohol as a gel polymer electrolyte.

(a) Schematic illustrating the process for the preparation of the N-SnO2/CF nanorod array electrode and (b) a photograph of the SnO2/CF and N-SnO2/CF electrodes.
Morphology and structure characterization
Figure 2 shows scanning electron microscope (SEM) images of SnO2 NRA and N-SnO2 NRA. Both SnO2 and N-SnO2 adopt a uniform nanorod array with quadrangular prism shape. The side length of the quadrangular nanorod is 50–60 nm for SnO2 and 20–30 nm for N-SnO2. Comparatively, N-SnO2 shows a bigger interspace of neighboring nanorods than SnO2, accordingly promoting the electrolyte ion diffusion in the electrochemical reaction.

SEM images of (a and b) SnO2 NRA and (c and d) N-SnO2 NRA.
Figure 3 shows the energy diffraction X-ray spectra of SnO2 and the N-SnO2 nanorod array. Table 1 lists the elemental analysis of SnO2 and the N-SnO2 nanorod array. SnO2 comprises tin (21.4 at %) and oxygen (78.6 at %). The atomic ratio of O/Sn is much larger than 2:1, which is ascribed to the surface hydroxylation group of SnO2. The nitrogen, oxygen, and tin elements are determined in N-SnO2, which indicates that the nitrogen has been doped into SnO2. A high oxygen atomic percent of 72.3% is also due to the surface hydroxyl group. The relative nitrogen atomic percentage is 12.5%, indicating a high nitrogen-doping level in N-SnO2 NRA. Usually, Sn(OH)4 is formed by hydrolysis of the SnCl4 precursor which is shown as the reaction formula (1). SnO2 is formed by dehydration annealing treatment of Sn(OH)4 which is shown as the reaction formula (2). N-SnO2 is prepared by nitriding treatment of SnO2 which is shown as the reaction formula (4). Comparatively, Sn(OH)4 has a higher reactivity than SnO2 with NH3 due to the superior interfacial proton acid-base affinity. Accordingly, a high nitrogen-doping level is achieved by nitriding treatment of Sn(OH)4 which is shown as the reaction formula (3). Sufficient nitrogen-doping could lead to improving the conductivity and the reactivity of N-SnO2. It should be noted that the N-doping content is not optimized in N-SnO2 for supercapacitor applications. The formation of N-SnO2 (SnO2 − xNx) involves the following reactions
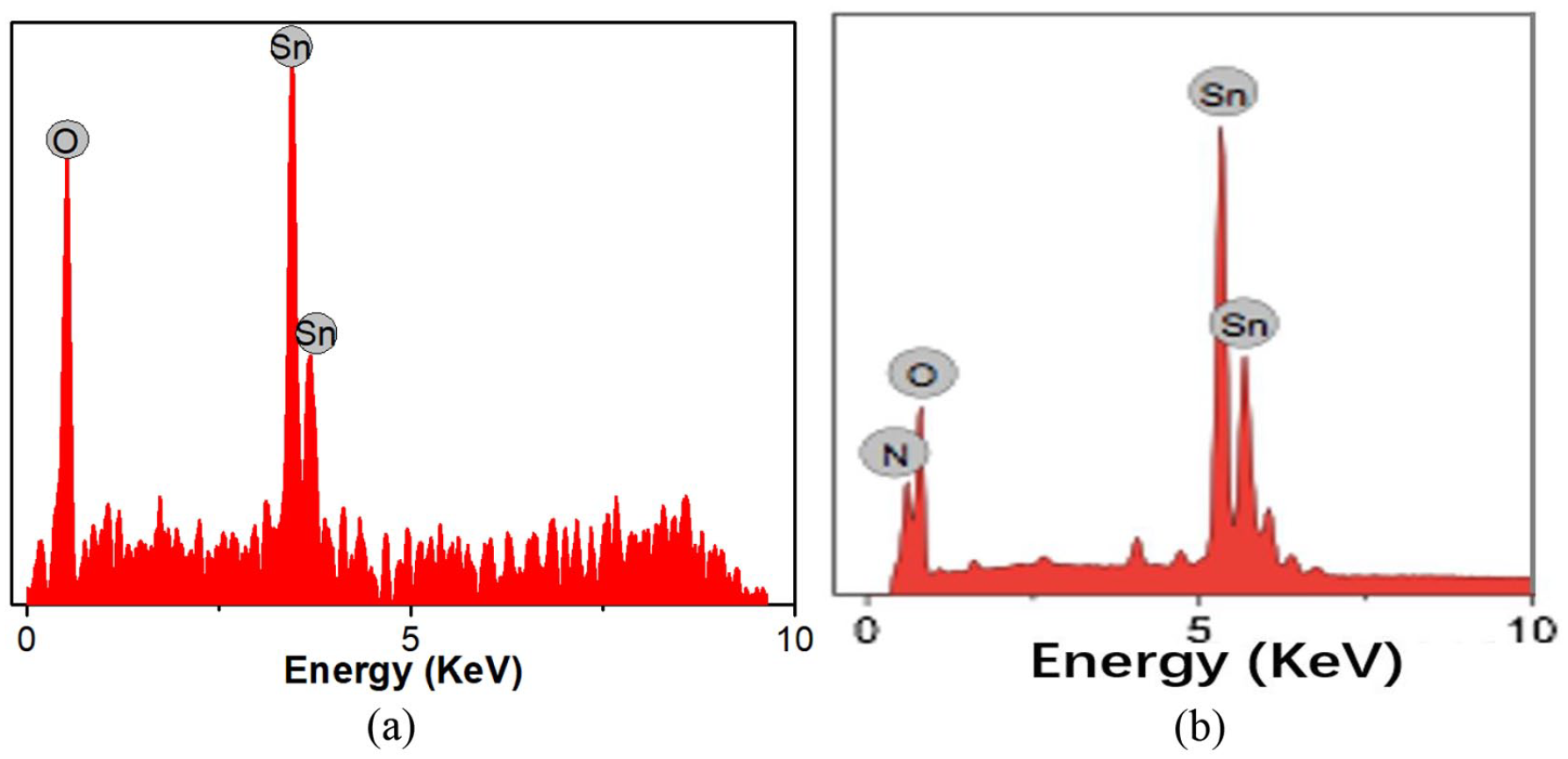
Energy diffraction X-ray spectra of (a) SnO2 and (b) N-SnO2.
Elemental analysis of SnO2 and N-SnO2 nanorod array.

Figure 4 shows the X-ray diffraction (XRD) patterns of SnO2/CF and N-SnO2/CF. Concerning the CF substrate, SnO2/CF and N-SnO2/CF exhibit a strong XRD peaks at around 2θ = 25°, corresponding to the (002) crystal plane of graphitic carbon. Both samples show similar characteristic diffraction peaks at 26.6°, 33.8°, and 38.0°, which could be indexed to the (110), (101), and (200) crystal planes of tetragonal rutile SnO2 (JCPDS card no. 41-1445). N-SnO2/CF does not show other new characteristic diffraction peaks. So, N-SnO2 and SnO2 exhibit similar crystal phase.

XRD patterns of SnO2/CF and N-SnO2/CF NRA electrodes.
Figure 5(a) shows the survey X-ray photoelectron spectroscopy (XPS) spectra of SnO2 and N-SnO2. As common points, the characteristic peaks of Sn
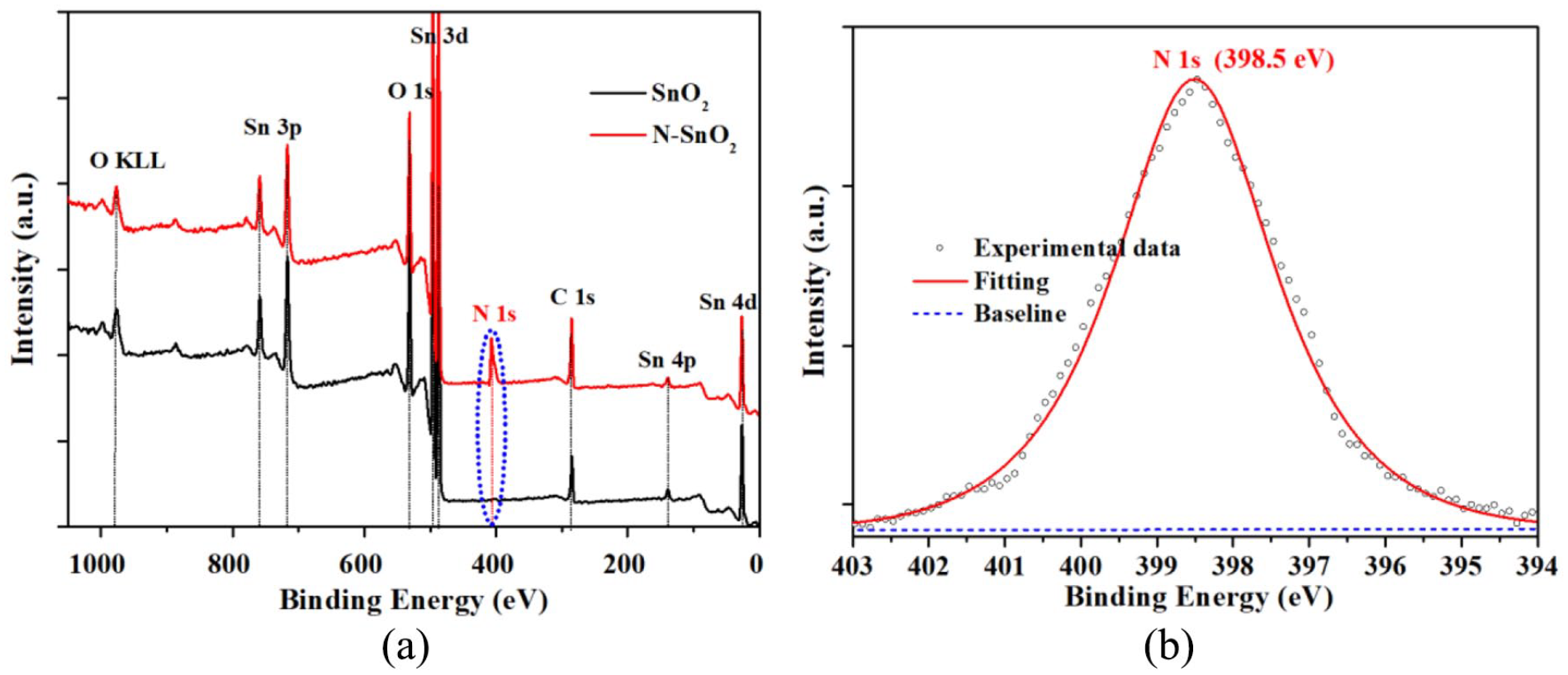
(a) Survey XPS spectra of the SnO2/CF and N-SnO2/CF NRA electrodes and (b) N
Electrochemical properties
Figure 6 shows the electrochemical impedance spectra of the SnO2/CF and N-SnO2/CF NRA electrodes and the corresponding equivalent circuit. The equivalent circuit elements include ohm resistance (Ro), charge transfer resistance (Rct), and the constant phase element (CFE) and the Warburg impedance element (Wo). The ohm resistance can be obtained from the intercept of the impedance curve on the real axis in high-frequency region. Table 2 lists the fitting parameters of the equivalent circuit of the SnO2/CF and N-SnO2/CF NRA electrodes. The Ro value decreases from 2.774 Ω for SnO2 NRA to 2.141 Ω for N-SnO2 NRA, indicating that the improved conductivity of N-SnO2 contributed to by nitrogen-doping. The charge transfer resistance can be estimated from the semicircle diameter in high-frequency region. The Rct value also obviously decreases from 1.24 Ω for SnO2 NRA to 0.85 Ω for N-SnO2 NRA, indicating the improved charge transfer of N-SnO2 contributed to by the high interspace of the nanorods. The straight line in the low-frequency region is related to the diffusion-controlled Warburg impedance. The N-SnO2 NRA shows a larger slope than the SnO2 NRA, indicating better approaching an ideal capacitor performance. The CFE can be defined by the capacitance (CFE-T) and the constant phase element exponent (CFE-P). N-SnO2 NRA shows a higher CFE-T (1.968) than SnO2 NRA (1.312), indicating its more porous structure. N-SnO2 NRA shows a relatively higher CFE-P (0.963) than SnO2 NRA (0.894), indicating that it is closer to being an ideal capacitor. The Wo value can be defined by the Warburg phase element exponent (Wo-P), the time constant (Wo-T), and the Warburg resistance (Wo-R). Both SnO2 and N-SnO2 NRA show similar Wo-P values of around 0.5, indicating the finite-length Warburg diffusion. N-SnO2 NRA shows a much lower Wo-R (0.719) than SnO2 NRA (3.164), indicating that it is more suitable for electrolyte ion diffusion. N-SnO2 NRA shows a relatively lower Wo-T (0.020) than SnO2 NRA (0.023), indicating a higher effective diffusion coefficient.

(a and b) Electrochemical impedance spectra of the SnO2/CF and the N-SnO2/CF NRA electrodes, and (c) the corresponding equivalent circuit.
The fitting parameters of the equivalent circuit of the SnO2/CF and N-SnO2/CF NRA electrodes.

CFE: constant phase element; CFE-T: constant phase element capacitance; CFE-P: constant phase element exponent.
Figure 7(a) and (b) shows the cyclic voltammetry (CV) curves of the SnO2/CF and N-SnO2/CF NRA electrodes at different scan rates. The CV curves of both electrodes maintain a similar shape without obvious distortion at different scan rates from 5 to 200 mV s−1. Figure 7(c) shows the response current curves at different scan rates. Herein, the average response current density is defined as

(a and b) CV curves at different scan rates. (c) The corresponding current response curve. (d and e) GCD curves at different current densities. (f) Rate capacitance retention curves and (g) cycling capacitance retention curves of the SnO2/CF and (g) N-SnO2/CF NRA electrodes.
Figure 8(a) and (b) shows the potential-extended CV curves at 5 mV s−1 and the potential-extended GCD curves at 1 A g−1 for N-SnO2 supercapacitor. When the window potential is extended from 0.8 to 1.6 V, the CV and GCD curves of the N-SnO2 supercapacitor mostly keep a similar shape without any obvious distortion or deformation. The polarization effect becomes more and more obvious at a higher potential above 1.6 V. Hence, subsequent electrochemical measurements of the N-SnO2 supercapacitor were controlled at 1.6 V. Figure 8(c) and (d) shows the CV curves at scan rates of 5–200 mV s−1 and GCD curves at current densities of 1–10 A g−1 for the N-SnO2 supercapacitor. The CV curves maintain a similar rectangle shape without any obvious distortions from 5 to 200 mV s−1. Also, the GCD curves remain in a rectangular shape without any obvious distortions from 1 to 10 A g−1. The specific capacitance decreases from 75.2 to 55.1 F g−1, while keeping the high rate capacitance retention of 73.3%. This means that the N-SnO2 supercapacitor can conduct a stable and reversible charge–discharge process at an output voltage of 1.6 V. Figure 8(e) shows the Ragone plot of the N-SnO2 supercapacitor. When the current density increases from 1 to 10 A g−1, the power density increases from 800 to 8000 W kg−1. Meanwhile the energy density decreases from 24.23 to 9.81 Wh kg−1, while maintaining the high-performance capacity in comparison with other reported SnO2 supercapacitors. 42 Moreover, the N-SnO2 supercapacitor also demonstrates higher energy density than an as-reported different SnO2 supercapacitor. 43 Figure 8(f) shows photographs of the N-SnO2 supercapacitor powering electric devices. It should be noted that the N-SnO2 supercapacitor is composed of a single unit without any serial or parallel connection. It can well power a light-emitting diode (LED) light bulb with a nominal voltage of 1.8 V and drive an electric fan with a nominal power of 1.0 W, indicating the high performance the N-SnO2/CF NRA electrode and its effective interaction with the H2SO4–PVA gel electrolyte. Hence, the N-SnO2 NRA demonstrates superior energy-storage capability.
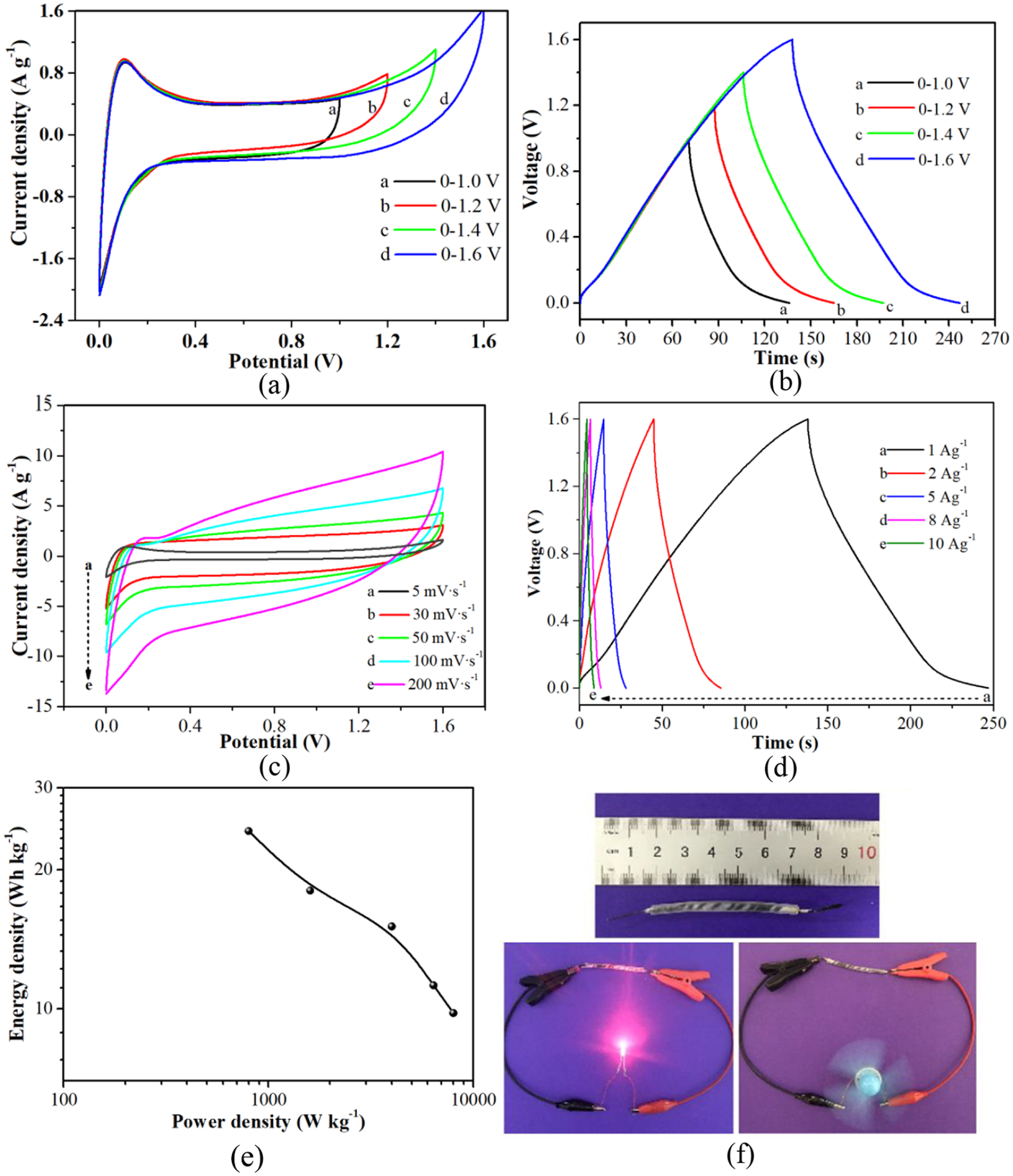
Electrochemical performance of the N-SnO2 supercapacitor: (a) potential-extended CV curves at 5 mV s−1, (b) potential-extended GCD curves at 1 A g−1, (c) CV curves at scan rates of 5–200 mV s−1, (d) GCD curves at current densities of 1–10 A g−1, (e) the Ragone plot at current densities of 1–10 A g−1, and (f) photographs of the N-SnO2 supercapacitor powering an LED and driving an electric fan.
Conclusion
An electroactive N-SnO2 NRA is designed and fabricated as an effective energy-storage electrode material for supercapacitor applications. N-SnO2 supported on a CF substrate is prepared using SnCl4 as the precursor through hydrolysis, hydrothermal growth, and an NH3-nitriding process. N-SnO2 has a high nitrogen-doping level of 12.5% which exhibits highly improved conductivity. The N-SnO2/CF NRA electrode shows higher specific capacitance and rate capacitance retention (105.4 F g−1 at 0.5 A g−1 and 45.0% from 0.5 to 5 A g−1) than an SnO2/CF NRA electrode (58.6 F g−1 and 38.4%). Moreover, the cycling capacitance retention after 2000 cycles increases from 78.1% for SnO2/CF to 98.8% for N-SnO2/CF, presenting superior electrochemical cycling stability. The N-SnO2 supercapacitor demonstrates stable and reversible charge–discharge operation at an output voltage of 1.6 V. When the current density increases from 1 to 10 A g−1, the specific capacitance decreases from 75.2 to 55.1 F g−1 and the energy density decreases from 24.23 to 9.81 Wh kg−1. Hence, N-SnO2 NRA demonstrates the superior capacitance performance.
Experimental
Preparation of the N-SnO2/CF electrode and N-SnO2 supercapacitor
The CF was immersed in 30 mL of 0.055 M SnCl4 solution. 30 mL of 0.11 M NaOH solution was added dropwise in SnCl4 solution and kept for 3 h at room temperature. Sn(OH)4 colloidal particles were prepared through a hydrolysis process and were fully adsorbed onto the CF substrate, forming Sn(OH)4/CF nanoparticles. A tin precursor solution was prepared from 30 mL of 0.0125 M SnCl4 and 0.25 M NaOH aqueous solutions. The Sn(OH)4/CF and this precursor solution were placed into a Teflon-lined stainless-steel autoclave. The hydrothermal process was conducted at 180 °C for 12 h. The obtained product was rinsed thoroughly with distilled water and dried in a vacuum oven at 60 °C to give the Sn(OH)4/CF nanorod array. The Sn(OH)4/CF was annealed in a tubular furnace at 400 °C for 4 h under a continuous ammonia vapor flow of 10 standard-state cubic centimeter per minute (sccm). An argon flow was used to cool down the sample, thus forming the N-SnO2/CF nanorod array. For comparison, the obtained Sn(OH)4/CF was subsequently converted into crystalline SnO2/CF by annealing at 320 °C for 2 h in a Muffle furnace. The mass loading of SnO2 and N-SnO2 was 4.2 and 3.8 mg cm−1 in the CF substrate, respectively.
Characterization and measurement
The surface microstructures and morphologies of SnO2 and N-SnO2 were characterized using an SEM (Zeiss Ultra Plus) under different magnifications. EDX (Oxford ISIS 310) was used to identify the element composition of N-SnO2. XPS (ESCALAB 250) was used to analyze the elements and the chemical states of the materials. The XPS peaks were calibrated against the C



where I is the current density (A g−1), Δt is the discharge time (s), ΔV is the potential window (V), and m is the mass of the electroactive material in the electrode.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Fundamental Research Funds for the Central Universities (2242018K41024) and the Priority Academic Program Development of Jiangsu Higher Education Institutions.




