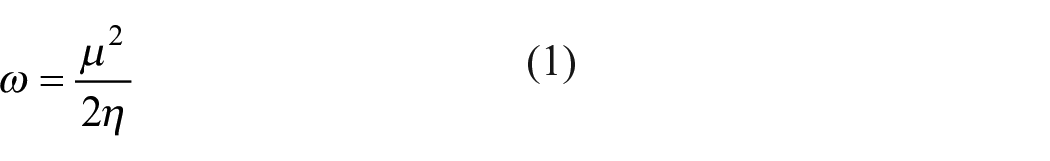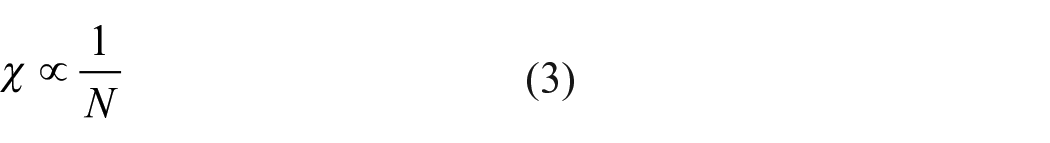Abstract
Electrophilicity index is a well-known Conceptual Density Functional Theory–based reactivity descriptor to explain a wide range of physicochemical behaviours. In the present work, an attempt is made to evaluate a new electrophilicity index scale in terms of nucleophilicity index relying on Gordy’s electronegativity scale. The computation is performed for 103 elements of the periodic table invoking regression analysis. The new set of electrophilicity index satisfies the sine qua non of a standard scale. Electrophilicity Equalization Principle is also validated by our computed data. It is put forward that the new scale will be useful in understanding various physicochemical properties and related phenomenon.
Keywords

Introduction
Ingold 1 defined the concept of electrophile and nucleophile for the first time describing electron deficient and electron-rich atoms or molecules. As we know, electrophiles are electron-deficient species and hence prefer to accept electrons and form bonds with nucleophiles. However, nucleophiles tend to be electron rich and thus prefer to donate electrons. Since then numerous attempts have been made to obtain a quantitative measure of atomic electrophilicity. Legon,2–4 Pérez et al., 5 Ritchie 6 and Mayr and Patz 7 have proposed empirical scales of electrophilicity. The quantitative definition of electrophilicity was proposed by Parr and co-workers 8 following the work of Maynard et al. 9 In mathematical form, it is represented as
In equation (1), μ represents chemical potential and η is the chemical hardness. Recently, Cárdenas et al. 10 have evaluated precise values of chemical potential as well as chemical hardness for atoms and ions and these can be used to calculate electrophilicity using equation (1).
Electrophilicity index is described as the capacity of a chemical species to accept an arbitrary number of electrons. 11 It is an intrinsic property of atoms which signifies the process of energy lowering by accepting electrons from donors. The electrophilicity index measures energy stabilization when the system gets an additional electronic charge from the environment. Electrophilicity index is one of the most important conceptual density functional theory (CDFT) descriptors,8,12–15 which allow understanding as well as predicting a plethora of physicochemical behaviours. Following Parr and his group’s work, 8 a great deal of research was carried out on electrophilicity scales which included global, local as well as molecular relationships.16–25 Recently, a force-concept-based electrophilicity index was also proposed for the first time. 26 This new scale is found to be efficient in computation of numerous real-world descriptors, for instance, internuclear bond distance. 26
As transparent, numerous studies have been performed for assessing electrophilicity index scales, however, a universal scale in terms of energy concept is still lacking. Although Parr et al’.s 8 definition is the standard approach to approximate change in energy related to the saturation of electrons (in view of a condensed electronic energy’s Taylor expansion of a non-interacting system at zero temperature), it presents certain intricate problems in this index. 27 Thus, to overcome this issue, we are proposing a simple and potent energy-based ansatz for electrophilicity index using inverse of nucleophilicity index.
Method of computation
As we know, Gordy 28 defined electronegativity (χ) of an atom as the electrostatic potential felt by one of its valence electrons. He suggested an ansatz for it which is expressed as
Here, Zeff is the effective nuclear charge and r is the covalent radius of an atom. e is the charge of an electron which is nearly equal to one.
Another important atomic descriptor is nucleophilicity index (N) and it is defined as the electron donating power of a chemical species. As per Tandon et al., 29 electronegativity is inversely related to nucleophilicity index
Also, electrophilicity index (ω) is the inverse of nucleophilicity index as proposed by Chattaraj and Maiti 30
Now by combining equations (2), (3) and (4), we get the following expression
Thus, equation (5) is proposed as the ansatz for evaluating atomic electrophilicity index expressed as inverse of nucleophilicity index (1/N) in terms of two descriptors, that is, effective nuclear charge (Zeff) and absolute radius (r) of atoms. We have used absolute radius in this formula since it is the true size descriptor of atomic property.
For the computation of atomic electrophilicity index (1/N) for 103 elements of the periodic table, equation (5) has been used. Regression analysis is performed employing inverse of nucleophilicity index as the dependent variable. Effective nuclear charge and absolute radii serve as the independent variables. Data for all the three parameters have been taken from previous works.31 ,32,33 e is approximately equal to 1 for each atom. Particular attention has been kept to compute electrophilicity index in proper unit (au).
In order to test the applicability of our computed electrophilicity indices, molecular electrophilicities have been computed invoking the electrophilicity equalization principle (EEP). 34 According to this principle, ‘the electrophilicity gets equalized during molecule formation, like the electronegativity and the hardness. The final electrophilicity is roughly given by the geometric mean of the corresponding isolated atom values’. 34 Mathematically, the molecular electrophilicity index (ωGM) is represented as
Here, Z is the total atoms present in a molecule, whereas ωi indicates the electrophilicity index of the ith atom (i = 1, 2, 3, . . ., Z). Relying upon this principle, we have calculated molecular electrophilicity indices for some diatomic molecules and compared them with the reported values. 34
In addition, for a variety of molecules, including inorganic, aliphatic and aromatic, we have computed frontier orbital energies (EH and EL) at DFT-B3LYP/LanL2DZ level of theory using Gaussian 03 35 computational package. These energies have been used to compute molecular electrophilicity indices using equation (7)
Furthermore, for the same set of molecules, molecular electrophilicity indices have been calculated empirically using our present work atomic data. The obtained results of both computational and empirical approach are then compared and correlated to establish the efficiency of the present work as well as to validate the EEP.
Result and discussion
The computed atomic electrophilicity index values for 103 elements in atomic unit (au) are shown in the form of a periodic chart in Table 1. We have also plotted the computed electrophilicity indices as a function of atomic number to test the periodicity (Figure 1). It can be seen from Table 1 that the computed electrophilicity indices of 103 elements of periodic table present periodic behaviour nicely. We have observed that the value of electrophilicity increases from left to right in a period and becomes maximum for noble gases. Elements of boron family present a slight deviation from the regular trend. Their values abruptly decrease within a period. Furthermore, electrophilicity index value for Pd also departs from the standard trend and shows an increase in the value. This may be accounted by the fully filled shell structure of Pd. Moreover, the sizes of d- and f-block elements undergo a slow and constant reduction and this established fact is adequately noted from our computed data. As apparent, transition metal elements show very less difference in their electrophilicity values. The property of deformation is also related to smaller electrophilicity and the electrophilicity index values of transition metals support this fact. Furthermore, computed values for lanthanides also follow the accepted variation trend. The new scale of electrophilicity index conforms to the sine qua non of the electrophilicity scale.
Periodic chart of computed atomic electrophilicity index (1/N) values for 103 elements (approximated to third decimal place).


Plot of electrophilicity indices (1/N; in au) as a function of atomic number (He atom is excluded from the figure owing to its considerably high electrophilicity index value).
The effectiveness of our evaluated data is checked by comparing with electrophilicity index scale of Parr et al. based on ground-state parabola model 8 and Tandon et al. 26 Figure 2 represents the trend between the three model values. As evident, all the values run parallel to each other.

Furthermore, we have tested the validity of our computed electrophilicity data by calculating molecular electrophilicity indices for some diatomics using equation (6) and comparing with reported 34 values for the same molecules (Figure 3). An excellent correlation (R2 = 0.798) is found to exist between the computed and reported sets. In addition, a similar comparison is carried out between our calculated and computationally evaluated molecular electrophilicity indices for a variety of molecules. It is evident from Figure 4 that there is a difference between the magnitudes of both the electrophilicity indices; however, they are similar qualitatively. The obtained results run hand-in-hand (R2 = 0.848) displaying the efficiency of the computed electrophilicity index data and validating the suitability of EEP in calculating molecular values. Thus, our computed electrophilicity index follows EEP appreciably and can be used to predict other properties as well as phenomenon.

Comparison of our calculated molecular electrophilicity index (ωGM) values with reported 34 values for some diatomic molecules (in au).

Comparison of our calculated molecular electrophilicity index (ωGM) values with computationally evaluated values at DFT-B3LYP/LanL2DZ level of theory for a variety of molecules (in au).
Conclusion
In the present study, we have derived a scale for evaluating atomic electrophilicity indices of 103 elements in the form of inverse of nucleophilicity index. The new scale is based on two important atomic descriptors – effective nuclear charge and absolute radius. All the sine qua non of a descriptor is fulfilled by the proposed scale. Furthermore, periodicity is nicely followed by the computed data. A good correlation is achieved with the standard existing electrophilicity index scales as well. Finally, principle of electrophilicity equalization is validated by the present computation. Hence, it is suggested that the new scale is efficient and valuable for determining other physicochemical properties and related phenomenon.
Footnotes
Acknowledgements
The authors are thankful to Manipal University Jaipur, Jaipur and Presidency University, Bengaluru for providing computational resources and research facility.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.